Cancer Genes - The Prime Suspects
- Proto-oncogenes (Accelerators):
- Promote normal cell growth/division.
- Mutation (gain-of-function) → Oncogenes (e.g., RAS, MYC, EGFR).
- Tumor Suppressor Genes (TSGs) (Brakes):
- Inhibit proliferation, induce apoptosis.
- Mutation (loss-of-function, Knudson's "two-hit" hypothesis).
- E.g., TP53, RB1, APC, BRCA1/2.
- Apoptosis Regulating Genes:
- Control programmed cell death.
- Imbalance (e.g., ↑BCL-2 anti-apoptotic, ↓BAX pro-apoptotic) → uncontrolled cell survival.
- DNA Repair Genes (Caretakers):
- Maintain genomic integrity.
- Defects → ↑mutation rates, genomic instability (e.g., MSH2, MLH1, BRCA).
⭐ TP53, "guardian of the genome," is the most commonly mutated gene, found in >50% of sporadic human cancers.
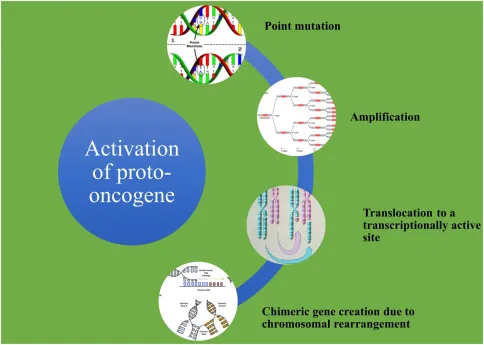
Oncogenes - Pedal to Metal
- Mutated or overexpressed versions of normal proto-oncogenes; they drive uncontrolled cell proliferation via gain-of-function mutations.
- Dominant: one mutated allele sufficient.
- Activation Mechanisms:
- Point Mutation: RAS (pancreatic, colon, lung Ca).
- Gene Amplification: N-MYC (neuroblastoma), ERBB2/HER2 (breast Ca).
- Chromosomal Translocation:
- BCR-ABL (t(9;22), Philadelphia chr.): CML.
- MYC (t(8;14)): Burkitt lymphoma.
- Examples & Roles:
- RAS: GTP-binding protein, signal transduction.
- MYC: Transcription factor, cell cycle progression.
- ERBB2 (HER2): Receptor tyrosine kinase.
- ABL: Non-receptor tyrosine kinase.
⭐ BCR-ABL fusion in CML results in constitutively active tyrosine kinase, a prime target for imatinib.
Tumor Suppressors - Guardians Down!
- Act as cellular "brakes"; normally prevent uncontrolled cell growth.
- Loss-of-function mutations promote cancer.
- Typically require inactivation of both alleles (Knudson's "two-hit" hypothesis).
- First hit: often germline (inherited).
- Second hit: somatic (acquired).
- Key Examples & Associations:
- RB1: Retinoblastoma, osteosarcoma (G1/S checkpoint).
- TP53: Li-Fraumeni syndrome ("Guardian of the Genome").
- APC: Familial adenomatous polyposis (FAP), colorectal cancer.
- BRCA1/BRCA2: Hereditary breast and ovarian cancer.
- NF1/NF2: Neurofibromatosis 1 & 2.
- WT1: Wilms tumor.

⭐ TP53 is the most frequently mutated gene in human cancers, implicated in over 50% of sporadic tumors.
System Sabotage - Repair, Death, Epigenetics
- DNA Repair Defects (Genomic Instability):
- Mismatch Repair (MMR): HNPCC/Lynch (MSH2, MLH1). Microsatellite Instability (MSI).
- Nucleotide Excision Repair (NER): Xeroderma Pigmentosum (XP). UV sensitivity, skin cancers.
- Homologous Recombination (HR): BRCA1/2 mutations. Breast, ovarian cancer risk. Fanconi Anemia.
- ATM gene: Ataxia-Telangiectasia. Defective DNA damage sensing.
- Evasion of Apoptosis (Resisting Cell Death):
- BCL-2 family: ↑Anti-apoptotic BCL-2 (follicular lymphoma t(14;18)).
- TP53 mutations: Loss of p53-mediated apoptosis.
- IAPs: Upregulation blocks caspases.
- Epigenetic Alterations (Gene expression changes without DNA sequence alteration):
- DNA Methylation: TSG promoter hypermethylation (CDKN2A, MLH1). Global hypomethylation.
- Histone Modification: Altered acetylation, methylation (EZH2).
- Non-coding RNAs: Dysregulated miRNAs (oncomiRs) & lncRNAs.
⭐ Follicular Lymphoma's t(14;18) translocation constitutively activates BCL-2 by placing it near the IgH enhancer, promoting cell survival via apoptosis evasion.
High‑Yield Points - ⚡ Biggest Takeaways
- Proto-oncogenes (e.g., RAS, MYC) drive cancer via gain-of-function mutations.
- Tumor suppressor genes (e.g., TP53, RB1) require loss-of-function (Knudson's two-hit hypothesis).
- TP53 is the most commonly mutated gene in human cancers, crucial for cell cycle control.
- Defects in DNA repair genes (e.g., BRCA1/2, MSH/MLH) lead to ↑ genomic instability.
- Epigenetic alterations like hypermethylation can silence tumor suppressor genes.
- Chromosomal translocations (e.g., BCR-ABL in CML) create oncogenic fusion proteins.
- Telomerase reactivation allows limitless replicative potential, a hallmark of cancer.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more