Principles of Molecular Pathology - Gene & Genome Basics
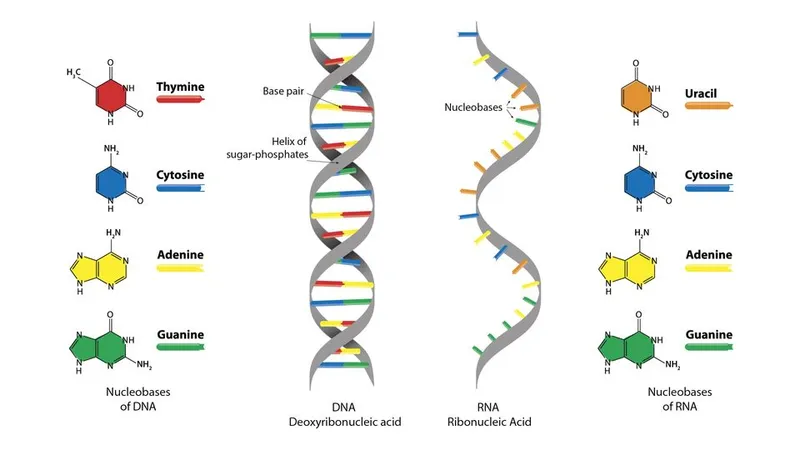
- DNA vs. RNA Structure:
- DNA: Deoxyribose sugar, Thymine base, Double helix.
- RNA: Ribose sugar, Uracil base, Single strand.
- Gene: Basic hereditary unit; a DNA segment coding for a functional product (RNA/protein).
- Genome: An organism’s complete set of DNA.
- Chromosome: Structure within cells containing genetic material (DNA).
- Central Dogma of Molecular Biology:
- Mutations: Permanent alterations in DNA sequence.
- Point Mutation: Change in a single nucleotide.
- Silent: No change in amino acid.
- Missense: Results in a different amino acid.
- Nonsense: Creates a premature stop codon.
- Frameshift Mutation: Insertion or deletion of nucleotides (not in multiples of three); alters the reading frame.
- Point Mutation: Change in a single nucleotide.
- Polymorphism: Common variation in DNA sequence within a population.
⭐ Single Nucleotide Polymorphisms (SNPs) are the most common type of genetic variation in the human genome.
- Allele: Variant form of a given gene.
- Genotype: An individual's collection of genes.
- Phenotype: Observable characteristics resulting from genotype-environment interaction.
Molecular Techniques - Lab Sleuth Kit
- Polymerase Chain Reaction (PCR): Amplifies specific DNA segments.
- Principle: Exponential DNA amplification.
- Steps: Denaturation, Annealing, Extension.
- Types: RT-PCR (RNA template), qPCR (quantitative real-time).
-
Blotting Techniques: 📌 SNoW DRoP mnemonic:
- Southern: DNA detection.
- Northern: RNA detection.
- Western: Protein detection.
-
Hybridization: Probes (labeled DNA/RNA) bind to target sequences.
- In Situ Hybridization (ISH): Detects sequences in cells/tissues.
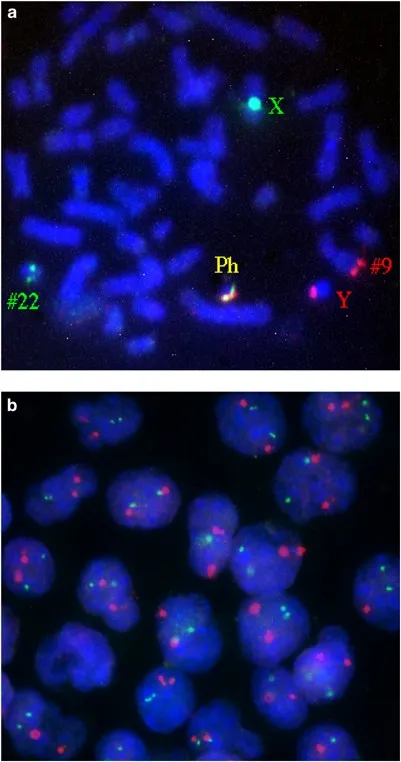
- Fluorescent ISH (FISH): Visualizes genes; detects gene amplification (e.g., HER2 in breast cancer), translocations (e.g., BCR-ABL in CML).
-
Microarrays: Analyze expression of thousands of genes simultaneously.
-
Sequencing:
- Sanger Sequencing: Dideoxy chain termination method for determining nucleotide sequence.
- Next-Generation Sequencing (NGS): High-throughput, parallel sequencing; faster & cost-effective for large genomes/panels.
⭐ Polymerase Chain Reaction (PCR) can amplify a single copy of DNA by millions of times, enabling detection of minute quantities of genetic material.
Clinical Applications - Disease Detectives
Molecular pathology identifies disease mechanisms:
- Genetic Disorders:
- Monogenic: Detects CFTR mutations (Cystic Fibrosis), HBB gene variants (Sickle Cell Anemia).
- Chromosomal: Identifies Trisomy 21 (Down Syndrome).

- Trinucleotide Repeats: Diagnoses Huntington's Disease (expanded HTT gene repeats).
- Cancer:
- Oncogenes (RAS, MYC) & Tumor Suppressors (TP53, RB1) for diagnosis/prognosis.
- Therapeutic Markers: EGFR mutations (lung cancer), HER2 amplification (breast cancer).
- Fusion Genes: BCR-ABL (Chronic Myeloid Leukemia - CML).
- Infectious Diseases:
- Pathogen ID & Load: HIV viral load monitoring, HPV typing for cancer risk.
- Antimicrobial Resistance: Detects genes like mecA (MRSA), NDM-1.
⭐ The Philadelphia chromosome, resulting from the t(9;22) translocation creating the BCR-ABL fusion gene, is a hallmark of Chronic Myeloid Leukemia (CML).
Emerging Concepts - Pathology's Future
- Epigenetics: Heritable changes not altering DNA sequence.
- DNA Methylation: Gene silencing; aberrant in cancer.
- Histone Modification: Alters chromatin accessibility.

- Pharmacogenomics: Tailoring drug therapy to genetic makeup.
- E.g., CYP enzyme variants (warfarin sensitivity).
⭐ Pharmacogenomics aims to tailor drug therapy based on an individual's genetic makeup, improving efficacy and reducing adverse drug reactions.
- Liquid Biopsy: Non-invasive tumor monitoring via circulating tumor DNA (ctDNA).
- Applications: Early detection, monitoring response, resistance.
- Gene Therapy: Introducing genetic material to treat disease.
- Viral vectors common for delivery.
- CRISPR-Cas9: Genome editing tool for precise DNA modification (brief mention).
High‑Yield Points - ⚡ Biggest Takeaways
- Central Dogma (DNA → RNA → Protein) underpins all molecular processes.
- PCR enables DNA amplification for diagnostics; RT-PCR for RNA.
- Blotting: Southern (DNA), Northern (RNA), Western (Protein) detect specific macromolecules.
- FISH visualizes gene loci and chromosomal aberrations directly in cells.
- NGS provides high-throughput DNA/RNA sequencing for comprehensive genomic analysis.
- Monoclonal antibodies offer specific targeting in diagnostics and therapeutics.
- Epigenetics (e.g., DNA methylation) regulates gene expression without altering DNA sequence.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

