QC Basics - Error Erasers
- Definition: System ensuring molecular test reliability, accuracy, and reproducibility. Essential for valid patient results.
- Goal: Detect & minimize errors across all test phases (pre-analytical, analytical, post-analytical).
- Error Types:
- Pre-analytical: Sample issues (collection, transport, storage). Most frequent.
- Analytical: Assay performance (reagents, equipment, technique).
- Post-analytical: Data handling, result interpretation & reporting.
- Key Metrics:
- Accuracy: Closeness to true value.
- Precision (Reproducibility): Consistency of results.
- Sensitivity & Specificity: Diagnostic power.
- Tools: Controls (monitor test runs), calibrators (standardize assays), proficiency testing (external assessment).
⭐ Pre-analytical errors constitute the largest source of laboratory errors, often exceeding 60-70%.
Pre-Analysis QC - Specimen Sentinels
- Critical first step; "Garbage In, Garbage Out" (GIGO) principle applies. Errors here are often irreversible, impacting downstream molecular assays.
- Core Areas:
- Patient Identification: At least 2 unique identifiers (e.g., name, DOB, MRN). Barcoding preferred.
- Specimen Collection: Appropriate site, technique, correct tube (e.g., EDTA for DNA; PAXgene/RNAprotect for RNA). Prevent contamination (e.g., microbial, cross-sample).
- Sample Volume & Quality: Sufficient quantity for planned tests. Check for hemolysis, clots, lipemia.
- Labeling: Accurate, legible, securely attached to primary container. Must match requisition form details.
- Transport & Handling: Maintain sample integrity. Adhere to time limits (e.g., RNA processing within 1-2 hours at Room Temperature or stabilize immediately). Strict temperature control (ambient, refrigerated, frozen as per analyte requirements).
⭐ Pre-analytical phase errors account for up to 70% of laboratory errors, significantly impacting molecular diagnostic accuracy.
- RNA Specifics: Extremely labile due to RNases. Use RNase-free consumables. Immediate processing or stabilization (e.g., TRIzol, PAXgene tubes) is vital.

Analysis QC - Assay Avengers
- Goal: Ensure reliable, accurate molecular test results for patient care.
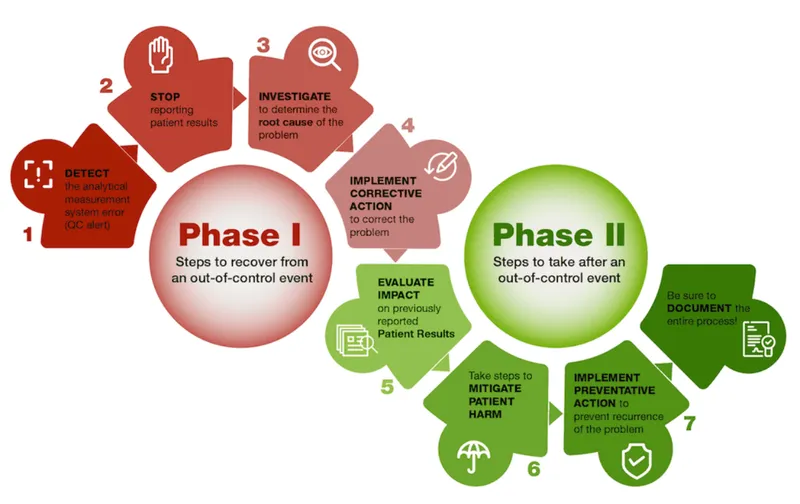
- Internal Quality Control (IQC):
- Run with each batch: Positive, Negative, Quantitative controls.
- Tracks precision, detects systematic/random errors.
- LJ charts + Westgard rules (e.g., 13s, 22s, R4s) for run validation and error detection.
- 📌 Westgard's Alerts Reveal Needy Systems (WARNS).
- External Quality Assessment (EQA) / Proficiency Testing (PT):
- Blind sample testing by external agency.
- Evaluates lab's accuracy and comparability vs peers.
- Calibration: Ensures instrument accuracy using calibrators/standards.
- Key Metrics for assay performance: Analytical Sensitivity (LoD), Specificity, Accuracy, Precision (CV%).

⭐ EQA participation is mandatory for NABL accreditation and reflects inter-laboratory performance.
Post-Analysis QC - Result Guardians
- Final Check: Ensures result accuracy, clinical relevance, clear & unambiguous communication.
- Key Steps: Meticulous interpretation, standardized reporting, secure archiving.
- Interpretation Process:
- Use current databases (ClinVar, OMIM, HGMD).
- ACMG/AMP guidelines for variant pathogenicity classification.
- Correlate molecular findings with detailed clinical phenotype.
- Reporting Standards:
- Clear, concise, unambiguous reports; standardized HGVS nomenclature.
- Include: Pt/Sample ID, method, results, interpretation, limitations, recommendations.
- Timely protocol for critical/actionable results.
- Documentation & Archiving:
- Retain all records (raw data, analysis files, reports) per NABL/CAP.
- LIMS for complete traceability, data integrity, audit trail.
⭐ VUS reporting: Clearly state uncertainty. Recommend genetic counseling, familial/functional studies if indicated. Avoid definitive diagnostic claims.
High‑Yield Points - ⚡ Biggest Takeaways
- Internal controls (e.g., housekeeping genes) ensure sample quality and detect PCR inhibitors.
- External controls (positive, negative) validate the entire analytical process and reagent integrity.
- Mandatory Proficiency Testing (PT/EQAS) assesses laboratory performance and ensures comparability.
- Strict adherence to SOPs is crucial for consistent and reliable results.
- Lot-to-lot reagent validation prevents performance variations and ensures accuracy.
- Monitor Analytical Sensitivity (LoD), Specificity, and Turnaround Time (TAT).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

