Hematopathology

On this page
🗺️ Hematopathology Foundations: The Marrow Mastery Blueprint
Hematopathology demands precision-every blast percentage, every cytogenetic abnormality, every immunophenotype pattern translates directly into diagnosis, prognosis, and treatment. Master the bone marrow's cellular architecture and you unlock the logic behind every peripheral smear abnormality, every lymph node biopsy, and every coagulation cascade failure. This lesson builds your diagnostic framework from normal hematopoiesis through neoplastic transformation, connecting morphology to molecular mechanisms and clinical outcomes.
The bone marrow microenvironment orchestrates hematopoiesis through a complex interplay of stem cells, stromal support, and growth factor signaling. Understanding this architecture reveals why certain neoplasms arise, how they progress, and where therapeutic interventions succeed.
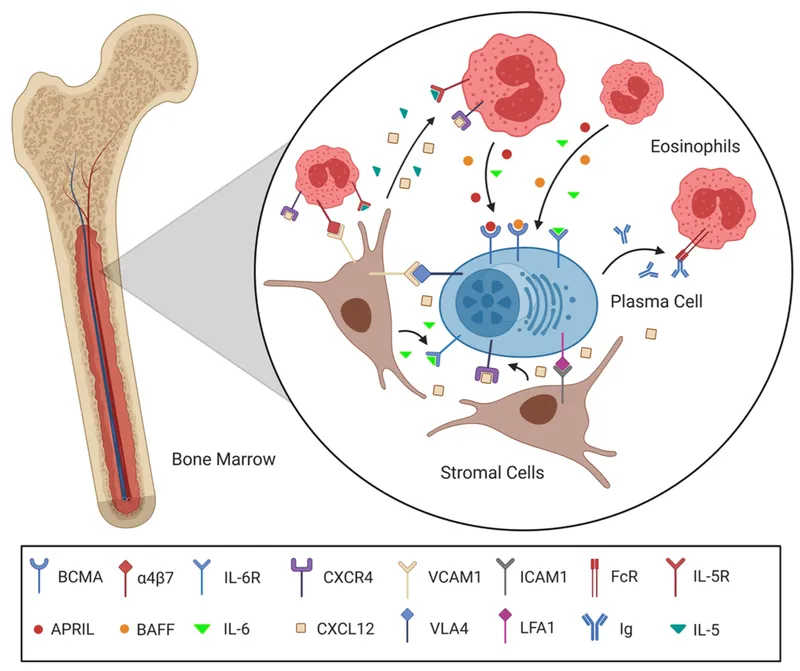
Hematopoietic Hierarchy & Compartments
-
Stem Cell Niche Architecture
- Endosteal niche: Quiescent HSC maintenance, <5% cycling rate
- Vascular niche: Active proliferation zone, 15-20% cycling HSCs
- CXCL12/CXCR4 axis: Critical retention signal
- SCF/c-KIT: Proliferation driver, mutated in 80% mastocytosis
- Mesenchymal stromal cells: Cytokine factory producing >20 growth factors
- TPO: Megakaryopoiesis driver, 30-50 pg/mL normal range
- G-CSF: Granulopoiesis stimulator, <100 pg/mL baseline
-
Maturation Compartments by Lineage
- Erythroid: 18-25% normal marrow cellularity
- Pronormoblast → basophilic → polychromatic → orthochromatic
- Enucleation occurs at orthochromatic stage, <24 hours terminal maturation
- Myeloid: 50-60% normal marrow cellularity
- Myeloblast → promyelocyte → myelocyte → metamyelocyte → band → neutrophil
- Maturation time: 10-14 days from blast to segmented neutrophil
- Megakaryocytic: <1% marrow cellularity but 2000-4000 platelets per cell
- Endomitosis produces polyploid nuclei, 8-64N DNA content
- Proplatelet formation: 1000-3000 μm cytoplasmic extensions
- Erythroid: 18-25% normal marrow cellularity
📌 Remember: M:E Ratio Mastery - Normal myeloid:erythroid ratio is 2-4:1. Inverted ratio (<2:1) suggests erythroid hyperplasia (hemolysis, hemorrhage) or myeloid hypoplasia (aplastic anemia). Elevated ratio (>4:1) indicates myeloid proliferation (infection, CML) or erythroid hypoplasia (pure red cell aplasia).
Neoplastic Transformation Patterns
The WHO classification organizes hematologic malignancies by cell lineage, maturation stage, and genetic drivers-a framework essential for diagnosis and risk stratification.
| Category | Maturation | Key Blast % | Prototype | Genetic Driver | 5-Year OS |
|---|---|---|---|---|---|
| Acute Leukemias | Arrested early | ≥20% blasts | AML, ALL | NPM1, FLT3, BCR-ABL1 | 25-70% |
| Myelodysplastic | Ineffective | <20% blasts | MDS-MLD | SF3B1, TET2, del(5q) | 40-80% |
| Myeloproliferative | Effective excess | <10% blasts | CML, PV, ET | BCR-ABL1, JAK2, CALR | 60-95% |
| Lymphomas | Mature lymphoid | Variable | DLBCL, FL | BCL2, BCL6, MYC | 30-90% |
| Plasma Cell | Terminal B-cell | Plasma cells | Multiple myeloma | CCND1, MAF, hyperdiploidy | 50-60% |
⭐ Clinical Pearl: The 20% blast threshold distinguishes acute leukemia from MDS-but context matters. AML with t(8;21), inv(16), or t(15;17) qualifies regardless of blast percentage due to favorable molecular biology. Similarly, therapy-related MDS carries poor prognosis even with <5% blasts due to complex karyotype patterns in >80% of cases.
Diagnostic Integration Framework
-
Morphology Foundation
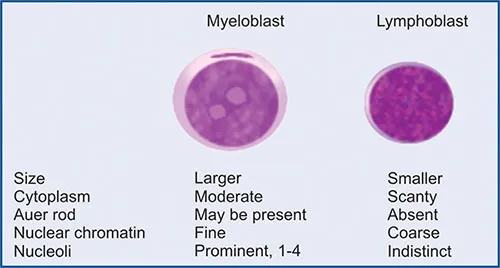
- Peripheral smear: Screen for circulating blasts, dysplasia, abnormal cells
- Blast identification: High N:C ratio, fine chromatin, 1-3 nucleoli
- Dysplasia markers: Pseudo-Pelger-Huët, hypogranular neutrophils, micromegakaryocytes
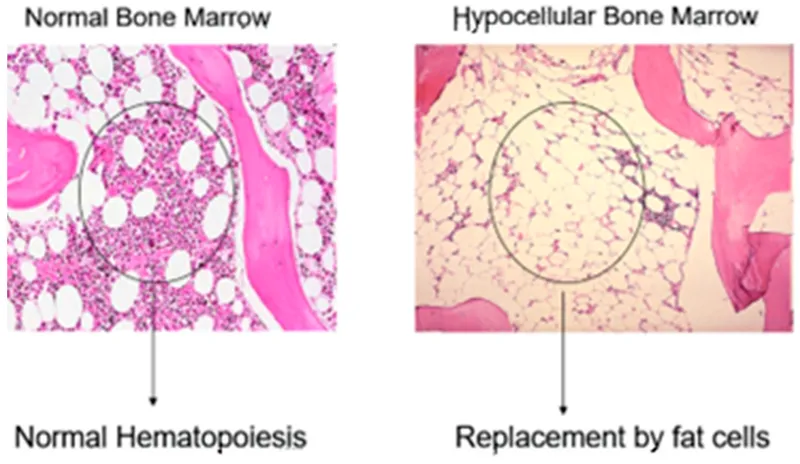
- Bone marrow aspirate: Assess cellularity (40-60% age-adjusted normal), M:E ratio, blast %
- Adequate sample: >10 spicules with >50 evaluable cells per lineage
- Core biopsy: Architecture, fibrosis (reticulin grade 0-1 normal), infiltration patterns
- Length requirement: ≥1.5 cm for adequate assessment
- Peripheral smear: Screen for circulating blasts, dysplasia, abnormal cells
-
Immunophenotyping Precision
- Flow cytometry: 4-10 color panels, ≥100,000 events for rare populations
- Myeloid markers: CD13, CD33, CD117, MPO
- B-lymphoid: CD19, CD20, CD22, surface immunoglobulin
- T-lymphoid: CD3, CD4, CD8, CD5, CD7
- Stem/progenitor: CD34, CD117, HLA-DR
- Aberrant expression: Loss of normal antigens or gain of lineage-inappropriate markers
- AML: CD7+ in 30%, CD19+ in 10-15%
- ALL: Myeloid antigen expression in 25-30%
- Flow cytometry: 4-10 color panels, ≥100,000 events for rare populations
💡 Master This: Immunophenotyping detects minimal residual disease (MRD) at 0.01% sensitivity-100-fold better than morphology. MRD positivity post-induction predicts relapse in 60-80% of AML patients versus <20% in MRD-negative cases. This quantitative assessment transforms prognostication and treatment intensity decisions.
Cytogenetic & Molecular Architecture
-
Chromosomal Abnormalities
- Translocations: Fusion genes drive >50% of acute leukemias
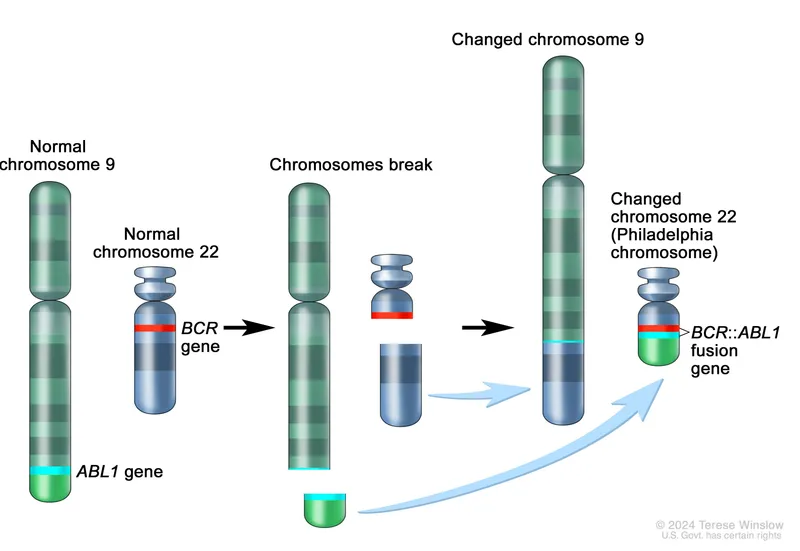
- BCR-ABL1 t(9;22): 95% CML, 25% adult ALL, 3% pediatric ALL
- PML-RARA t(15;17): 98% acute promyelocytic leukemia, ATRA-responsive
- RUNX1-RUNX1T1 t(8;21): 5-10% AML, favorable prognosis, 65% 5-year OS
- Deletions: Tumor suppressor loss
- del(5q): 10-15% MDS, lenalidomide-responsive in isolated del(5q)
- del(17p): TP53 locus, 5-10% MDS/AML, chemoresistance, <10% 2-year OS
- Numerical changes: Hyperdiploidy (favorable in ALL), monosomy 7 (poor in MDS/AML)
- Translocations: Fusion genes drive >50% of acute leukemias
-
Point Mutations & Small Variants
- NPM1: 30-35% AML, favorable if FLT3-ITD negative, 60% 5-year OS
- FLT3-ITD: 25-30% AML, high-risk, allelic ratio >0.5 predicts relapse
- JAK2 V617F: 95% PV, 55% ET, 65% PMF-diagnostic for MPN
- CALR: 25% ET, 35% PMF when JAK2 negative, better prognosis than JAK2+
- IDH1/IDH2: 15-20% AML, targetable with ivosidenib/enasidenib
📌 Remember: ELN Risk Stratification - European LeukemiaNet groups AML into 3 tiers: Favorable (NPM1-mutated without FLT3-ITD-high, core-binding factor), Intermediate (normal karyotype without favorable mutations), Adverse (complex ≥3 abnormalities, monosomy 5/7, TP53-mutated). This classification predicts 5-year OS ranging from 60% (favorable) to <10% (adverse).
Connect these foundational concepts through to understand how marrow failure manifests peripherally, then advance to where blast expansion disrupts this architecture.
🗺️ Hematopathology Foundations: The Marrow Mastery Blueprint
🔬 Clonal Evolution: The Neoplastic Transformation Engine
Hematologic malignancies arise through stepwise accumulation of mutations in hematopoietic stem and progenitor cells, creating clonal populations with proliferative advantage, impaired differentiation, and resistance to apoptosis. Understanding this multi-hit model explains disease heterogeneity, progression patterns, and therapeutic resistance mechanisms.
The Multi-Hit Transformation Model
Leukemogenesis requires 2-4 cooperating mutations across functional classes-a principle that predicts which genetic combinations drive malignancy and which remain pre-malignant.
-
Class I Mutations: Proliferation Drivers
- Tyrosine kinase activation: Constitutive signaling through RAS/MAPK and PI3K/AKT pathways
- FLT3-ITD: Internal tandem duplication in 25-30% AML, length matters-longer duplications (>40 bp) confer worse prognosis
- KIT mutations: 20-25% core-binding factor AML, D816V most common, predicts relapse risk
- BCR-ABL1: p210 fusion in CML (95%), p190 in ALL (25% adults)
- RAS pathway: NRAS, KRAS mutations in 10-15% AML, 30% juvenile myelomonocytic leukemia
- Downstream MEK activation increases proliferation 3-5 fold
- JAK-STAT activation: JAK2 V617F in 95% PV, drives erythrocytosis through EPO-independent signaling
- Tyrosine kinase activation: Constitutive signaling through RAS/MAPK and PI3K/AKT pathways
-
Class II Mutations: Differentiation Block
- Transcription factor disruption: Arrest maturation at blast stage
- RUNX1-RUNX1T1 t(8;21): Blocks myeloid maturation, requires 2-3 additional mutations for AML
- PML-RARA t(15;17): Sequesters retinoic acid receptors, APL differentiation block reversed by ATRA
- CBFB-MYH11 inv(16): Core-binding factor disruption, myelomonocytic differentiation arrest
- Epigenetic modifiers: Alter chromatin accessibility and gene expression
- DNMT3A: 20-25% AML, early mutation in pre-leukemic clones, persists in remission
- TET2: 10-15% AML, impairs DNA demethylation, associated with monocytic differentiation
- IDH1/IDH2: 15-20% AML, produce 2-hydroxyglutarate oncometabolite, blocks TET2 function
- Transcription factor disruption: Arrest maturation at blast stage
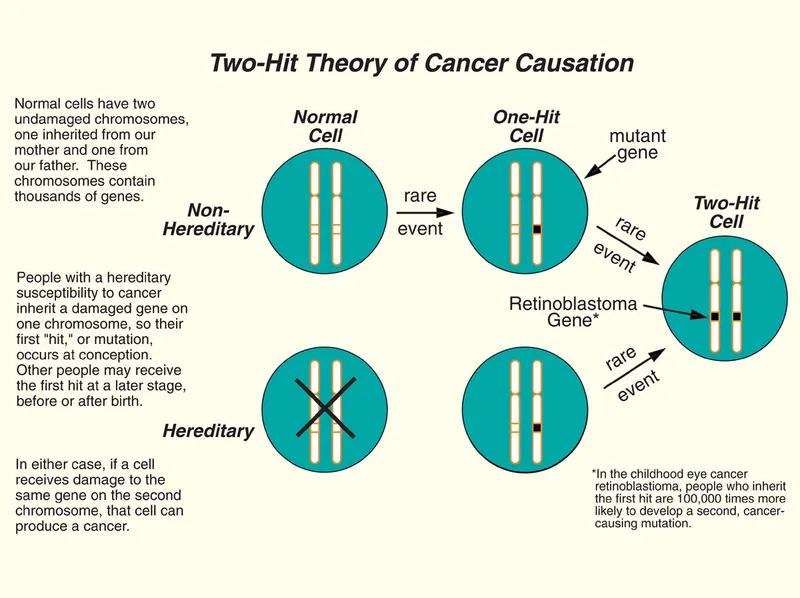
💡 Master This: The two-hit model explains why isolated FLT3-ITD causes myeloproliferative disease but not acute leukemia-proliferation alone is insufficient. Add NPM1 mutation (differentiation block) and you generate AML. This cooperation principle guides targeted therapy: single-agent FLT3 inhibitors fail (<15% remission rate), but combination with chemotherapy achieves 70% remission in FLT3-mutated AML.
Clonal Architecture & Evolution Dynamics
-
Pre-Leukemic Hematopoiesis
- Clonal hematopoiesis of indeterminate potential (CHIP): Age-related clonal expansion
- Present in 10% age 70+, 20% age 90+
- DNMT3A, TET2, ASXL1 most common (70% of CHIP)
- Progression to AML: 0.5-1% per year, 10-fold increased risk versus age-matched controls
- Clonal cytopenia of undetermined significance (CCUS): CHIP with cytopenias
- AML progression: 2-3% per year, 20-fold increased risk
- Mutation burden matters: Variant allele frequency >10% predicts progression
- Clonal hematopoiesis of indeterminate potential (CHIP): Age-related clonal expansion
-
Leukemic Clone Dynamics
- Founding clone: Carries initiating mutations, generates subclones
- Typically 60-90% of leukemic cells at diagnosis
- Subclonal evolution: Branching architecture with 3-8 detectable subclones
- Minor subclones (<5%) harbor resistance mutations pre-treatment
- Chemotherapy selects resistant clones, which expand at relapse
- Relapse patterns: 50-70% relapses derive from minor diagnostic subclones, not founding clone
- Founding clone: Carries initiating mutations, generates subclones
⭐ Clinical Pearl: Next-generation sequencing at diagnosis detects subclones carrying TP53 mutations at 1-5% frequency-below morphologic detection but sufficient to cause relapse. Patients with subclonal TP53 have <20% 2-year OS despite initial remission, versus 50-60% without TP53. This explains why morphologic remission doesn't guarantee cure.
Microenvironment Complicity
The bone marrow niche doesn't just support normal hematopoiesis-it actively protects leukemic cells from therapy and immune surveillance.
| Niche Component | Protective Mechanism | Clinical Impact | Therapeutic Target | Evidence |
|---|---|---|---|---|
| Stromal cells | CXCL12 chemotaxis, adhesion | Drug resistance ↑3-10 fold | CXCR4 inhibitor plerixafor | 40% mobilization |
| Hypoxic zones | HIF1α stabilization, quiescence | Persist through chemotherapy | HIF1α inhibitors | Preclinical |
| Osteoblasts | Calcium sensing, dormancy signals | MRD sanctuary, late relapse | Investigational | 20% residual |
| Adipocytes | Fatty acid metabolism support | Obesity worsens outcomes 15% | Metabolic inhibitors | Observational |
| Regulatory T-cells | PD-L1/PD-1 immune evasion | Suppress anti-leukemic immunity | Checkpoint blockade | 10-30% response |
-
Adhesion-Mediated Drug Resistance
- VLA-4/VCAM-1 binding: Protects 40-60% of leukemic cells from cytarabine
- Integrin signaling activates survival pathways: BCL-2 ↑2-4 fold, MCL-1 stabilization
- Minimal residual disease (MRD) enriched in CD49d+ (VLA-4) cells: 5-10 fold higher frequency
-
Metabolic Sanctuary
- Hypoxic niches: <1% oxygen tension, 30-50 μm from vasculature
- Quiescent leukemic stem cells: <5% in cycle, chemoresistant
- Oxidative stress low: Reactive oxygen species 50% of normoxic zones, protects from anthracyclines
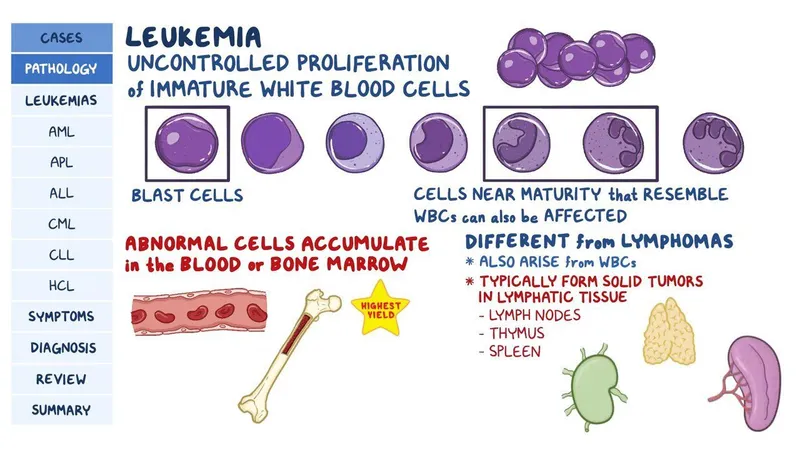
📌 Remember: SHIP Resistance - Stromal protection, Hypoxia, Immune evasion, Pathway activation-four mechanisms by which the marrow niche shields leukemia from therapy. Targeting CXCR4 with plerixafor mobilizes 40-60% of AML cells into circulation, where chemotherapy efficacy increases 3-fold. Combination strategies overcome niche-mediated resistance.
Epigenetic Dysregulation Mechanisms
-
DNA Methylation Abnormalities
- Hypermethylation: CpG island methylation silences tumor suppressors
- CDKN2A (p16): Methylated in 30-40% AML, loss of cell cycle control
- DAPK1: Methylated in 40% AML, apoptosis resistance
- Hypomethylation: Global demethylation drives genomic instability
- TET2 loss: Impairs 5-methylcytosine oxidation, blocks demethylation
- DNMT3A R882H: Dominant-negative effect, reduces methylation 40-60%
- Therapeutic implications: Hypomethylating agents (azacitidine, decitabine) achieve 40-50% response in MDS, 20-30% in AML
- Hypermethylation: CpG island methylation silences tumor suppressors
-
Histone Modification Disruption
- H3K27 methylation: EZH2 mutations in 10% MDS, loss-of-function reduces repressive marks
- H3K79 methylation: DOT1L recruited by MLL fusions, drives leukemogenesis in 5-10% AML
- Acetylation balance: CREBBP/EP300 mutations in 10% lymphomas, impair acetylation, reduce transcription
- BET proteins: Bromodomain readers (BRD4) maintain MYC expression, BET inhibitors show 30% response rates
💡 Master This: Epigenetic therapy works through differentiation induction, not cytotoxicity. Azacitidine at low doses (75 mg/m² × 7 days) incorporates into DNA and depletes DNMT1, causing passive demethylation over 3-4 cycles. This explains the median 3 months to response-slower than chemotherapy but durable in responders (median 18-24 months). High-dose azacitidine causes cytotoxicity without differentiation and loses efficacy.
Transition from these transformation mechanisms to their clinical manifestations through , where ineffective hematopoiesis demonstrates early clonal evolution, and , where full transformation arrests differentiation completely.
🔬 Clonal Evolution: The Neoplastic Transformation Engine
🧬 Diagnostic Integration: The Pattern Recognition Matrix
Hematopathology diagnosis synthesizes morphology, immunophenotype, cytogenetics, and molecular data into unified classification-each layer refines diagnostic precision and prognostic stratification. Master the integration algorithm and you transform raw laboratory data into actionable clinical intelligence.
Morphologic Pattern Recognition
Peripheral smear and bone marrow examination remain the diagnostic foundation, providing immediate clues to lineage, maturation stage, and neoplastic process.
- Blast Identification & Quantification
- Morphologic criteria: High nuclear:cytoplasmic ratio >3:1, fine chromatin, 1-3 prominent nucleoli
- Auer rods: Pathognomonic for AML, present in 30-50% cases, multiple rods suggest APL
- Cytoplasmic basophilia: Indicates ribosome content, prominent in lymphoblasts
- Blast enumeration standards
- Aspirate: Count 200-500 nucleated cells, report percentage
- Core biopsy: Estimate from multiple representative areas, correlate with aspirate
- Peripheral blood: Count 200 WBCs, include in WHO blast percentage if ≥20%
- Dysplasia assessment: Requires ≥10% of cells in lineage show abnormalities
- Neutrophils: Pseudo-Pelger-Huët (bilobed, >10%), hypogranularity, hyposegmentation
- Erythroid: Megaloblastic changes, nuclear budding, multinuclearity in >10%
- Megakaryocytes: Micromegakaryocytes, hypolobated nuclei, ≥3 abnormal forms
- Morphologic criteria: High nuclear:cytoplasmic ratio >3:1, fine chromatin, 1-3 prominent nucleoli
📌 Remember: The 10-20-30 Rule - MDS requires ≥10% dysplasia in a lineage, AML requires ≥20% blasts (except specific cytogenetics), and multi-lineage dysplasia means ≥2 lineages with ≥10% dysplasia each. Blast percentage at 10-19% defines MDS with excess blasts-2 (MDS-EB2), conferring median 9 months survival without treatment versus >5 years for low-grade MDS.
Immunophenotyping Decision Trees
- Lineage Assignment Hierarchy
- Myeloid: CD13, CD33, CD117, MPO (most specific)
- MPO positivity: ≥3% blasts by flow or cytochemistry defines myeloid
- Monocytic: CD14, CD64, CD11b, nonspecific esterase
- Megakaryocytic: CD41, CD61, CD42b-diagnose acute megakaryoblastic leukemia
- B-lymphoid: CD19 (required), CD22, CD79a, cytoplasmic CD22
- Precursor: CD34+, TdT+, surface Ig−
- Mature: Surface Ig+, CD20+, CD34−
- T-lymphoid: Cytoplasmic CD3 (most specific), surface CD3, CD7, CD5
- Precursor: CD34+, TdT+, variable CD4/CD8
- Mature: CD3+, CD4+ or CD8+, TdT−
- Myeloid: CD13, CD33, CD117, MPO (most specific)
| Leukemia Type | Key Positive | Key Negative | Aberrant Expression | Frequency | Prognostic Impact |
|---|---|---|---|---|---|
| AML with maturation | CD13, CD33, MPO, CD117 | CD34 (50%), HLA-DR (30%) | CD7 (30%), CD19 (10%) | 25-30% AML | CD7+ worse OS |
| Acute promyelocytic | CD33 bright, MPO bright | CD34, HLA-DR | CD2 (30%) | 10% AML | CD2+ ↑ relapse risk |
| Acute monocytic | CD14, CD64, CD11b, CD4 | CD34 (variable) | CD56 (20%) | 5-10% AML | CD56+ CNS risk ↑ |
| B-ALL | CD19, CD22, CD79a, TdT | MPO, surface Ig | Myeloid Ag (25-30%) | 75% ALL | Myeloid Ag+ neutral |
| T-ALL | cCD3, CD7, CD5, TdT | CD19, MPO | CD13/CD33 (30%) | 15-20% ALL | Early T-cell worse |
⭐ Clinical Pearl: Aberrant antigen expression creates leukemia-associated immunophenotypes (LAIPs) for minimal residual disease monitoring. A patient with AML co-expressing CD7 allows 0.01% sensitivity MRD detection by tracking CD34+/CD117+/CD7+ cells-absent in normal marrow. MRD ≥0.1% post-consolidation predicts 70% relapse risk versus <20% in MRD-negative patients.
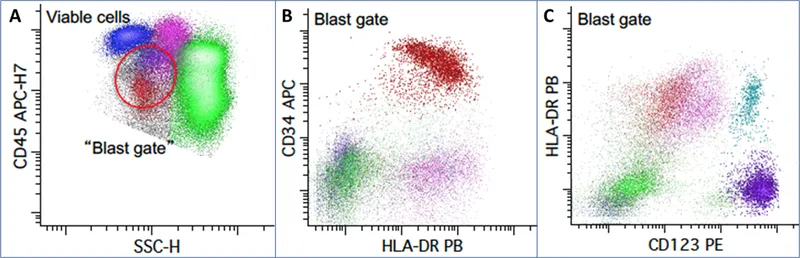
Cytogenetic Integration Framework
-
Recurrent Translocations
- Core-binding factor AML: Favorable prognosis, 60-70% 5-year OS
- t(8;21)(q22;q22) RUNX1-RUNX1T1: 5-10% AML, associated with Auer rods, granulocytic differentiation
- inv(16)(p13q22) or t(16;16) CBFB-MYH11: 5-8% AML, abnormal eosinophils, monocytic features
- Both require 2-3 consolidation cycles with high-dose cytarabine (3 g/m² q12h × 6 doses)
- Acute promyelocytic leukemia: Medical emergency, 90% cure rate with ATRA + arsenic
- t(15;17)(q24;q21) PML-RARA: 98% APL, diagnostic, ATRA-responsive
- Variant translocations (2%): ZBTB16-RARA, NPM1-RARA, STAT5B-RARA-ATRA-resistant
- KMT2A (MLL) rearrangements: Poor prognosis, <30% 5-year OS
- t(9;11)(p21;q23) MLLT3-KMT2A: Most common, 2-3% AML, monocytic features
-
100 partner genes described, infant ALL association (70-80%)
- Core-binding factor AML: Favorable prognosis, 60-70% 5-year OS
-
Adverse Cytogenetics
- Complex karyotype: ≥3 unrelated abnormalities, 10-15% AML, <10% 2-year OS
- Typically includes −5/del(5q), −7/del(7q), −17/del(17p)
- Associated with TP53 mutations in 70-80%
- Monosomy karyotypes: Chromosome 5 or 7 loss, chemoresistance
- Monosomy 7: 5% de novo AML, 30% therapy-related, <10% 5-year OS
- del(5q): 10% MDS (isolated), 30% AML (with other abnormalities)
- Specific translocations: Poor-risk despite balanced appearance
- t(6;9)(p23;q34) DEK-NUP214: 1-2% AML, basophilia, <20% 5-year OS
- inv(3)(q21q26) or t(3;3) GATA2,MECOM: 1-2% AML, dysplastic megakaryocytes, <15% 5-year OS
- Complex karyotype: ≥3 unrelated abnormalities, 10-15% AML, <10% 2-year OS
💡 Master This: Fluorescence in situ hybridization (FISH) detects cryptic abnormalities missed by karyotype in 5-10% of cases-particularly PML-RARA in APL with normal karyotype (10% of APL) and KMT2A rearrangements. FISH on peripheral blood can diagnose APL in 4-6 hours versus 3-5 days for karyotype, enabling immediate ATRA initiation to prevent fatal hemorrhage (occurs in 10-20% of untreated APL within 48 hours).
Molecular Diagnostic Algorithm
-
Tier 1: Diagnostic & Targetable Mutations (test at diagnosis)
- NPM1: 30-35% AML, favorable if FLT3-ITD negative
- FLT3-ITD and FLT3-TKD: 25-30% AML, midostaurin indication
- IDH1 and IDH2: 15-20% AML, ivosidenib/enasidenib targetable
- TP53: 5-10% AML, 70-80% complex karyotype, chemoresistance
- CEBPA: 5-10% AML, biallelic favorable (60% 5-year OS)
-
Tier 2: Prognostic Refinement (refine risk stratification)
- RUNX1: 10% AML, intermediate-adverse prognosis
- ASXL1: 10-15% AML, adverse prognosis
- KIT D816 mutations: 20-25% CBF-AML, predicts relapse
-
Tier 3: Emerging Targets (clinical trials)
- WT1: 10% AML, under investigation
- NRAS/KRAS: 10-15% AML, MEK inhibitor trials
📌 Remember: ELN 2022 Triple Risk - Favorable: NPM1 without FLT3-ITD-high OR CBF translocations OR biallelic CEBPA (35-40% of AML, 60-70% 5-year OS). Intermediate: Everything else without adverse features (35-40%, 40-50% OS). Adverse: Adverse cytogenetics OR TP53 OR RUNX1 OR ASXL1 OR FLT3-ITD-high without NPM1 (20-25%, <20% OS). This stratification determines transplant timing: adverse goes to transplant in CR1, favorable defers unless relapse.
Integrate these diagnostic layers through for blast crisis recognition and for mature neoplasm characterization. The next section builds differential diagnosis frameworks from these integrated patterns.
🧬 Diagnostic Integration: The Pattern Recognition Matrix
🏥 Differential Diagnosis Architecture: The Clinical Discriminator System
Hematopathology differentials hinge on quantitative discriminators-blast percentage, cytogenetic patterns, immunophenotypic profiles, and molecular signatures that distinguish entities with overlapping features. Master these decision points and you navigate diagnostic ambiguity with precision.
Blast Percentage Stratification
The blast count serves as the primary classification pivot, but context-lineage, cytogenetics, dysplasia-refines interpretation.
| Blast % | Diagnosis | Key Discriminators | Median Survival | Treatment | Progression Risk |
|---|---|---|---|---|---|
| <5% | MDS-MLD, MDS-RS-SLD | Dysplasia ≥10% in 1-2 lineages | >60 months | Supportive, EPO | 10-15% → AML |
| 5-9% | MDS-EB1 | Dysplasia + cytopenias | 18-24 months | HMA, transplant | 25-30% → AML |
| 10-19% | MDS-EB2 | High-risk cytogenetics common | 9-12 months | HMA, intensive if fit | 40-50% → AML |
| ≥20% | AML (most) | Lineage, cytogenetics, mutations | 12-18 months | Induction chemo | N/A (already AML) |
| Any % | AML with defining cytogenetics | t(8;21), inv(16), t(15;17) | >60 months | Targeted therapy | Relapse 20-30% |
| 20-90% | ALL (typical) | Lymphoid immunophenotype, BCR-ABL1 | 24-36 months | Multi-agent chemo | CNS relapse 5-10% |
⭐ Clinical Pearl: The 20% blast threshold is arbitrary-a patient with 19% blasts and complex karyotype has worse prognosis than one with 25% blasts and t(8;21). WHO 2022 acknowledges this by diagnosing AML regardless of blast count in core-binding factor and APL cases. Similarly, MDS-EB2 (10-19% blasts) behaves like AML and warrants intensive therapy in fit patients, achieving 40-50% complete remission versus <20% with hypomethylating agents alone.
Myeloid vs Lymphoid Lineage Discrimination
- Ambiguous Lineage Scenarios
- Mixed-phenotype acute leukemia (MPAL): 2-5% of acute leukemias
- Requires fulfilling criteria for ≥2 lineages on same blast population
- B/myeloid: CD19+ and MPO ≥3% or monocytic markers
- T/myeloid: cCD3+ or CD3+ and MPO ≥3% or monocytic markers
- Prognosis: Intermediate-poor, 30-40% 5-year OS, treat as ALL typically
- Acute leukemia with aberrant markers: Single lineage with inappropriate antigens
- AML with CD7 (30%), CD19 (10%), CD56 (15%)-still AML, not MPAL
- ALL with CD13/CD33 (25-30%)-still ALL, no prognostic impact
- Acute undifferentiated leukemia: Lacks lineage-defining markers (<1%)
- CD34+, HLA-DR+, TdT+ but no MPO, CD3, CD19
- Very poor prognosis, <20% 5-year OS
- Mixed-phenotype acute leukemia (MPAL): 2-5% of acute leukemias
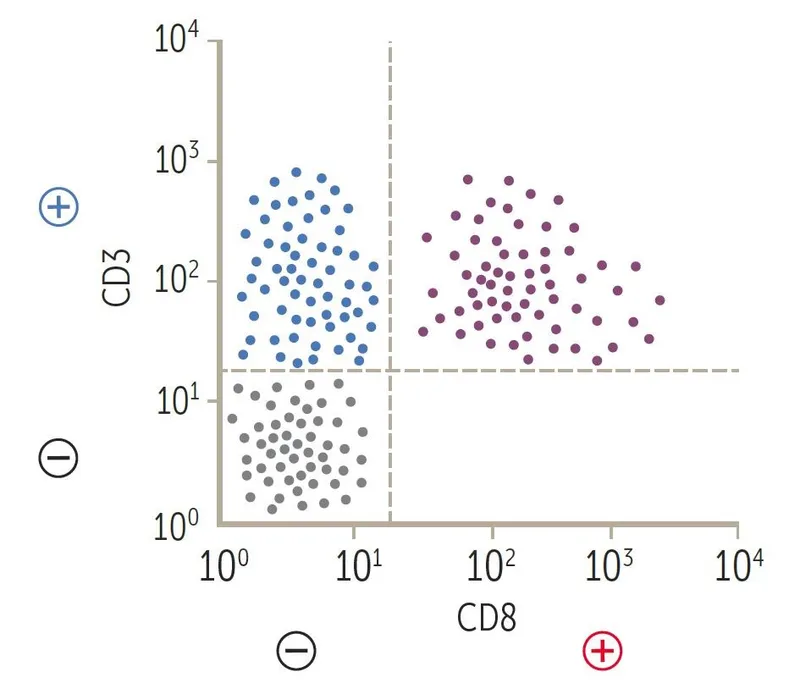
💡 Master This: The WHO lineage assignment algorithm prioritizes MPO for myeloid (≥3%), cytoplasmic CD3 for T-cell, and CD19 for B-cell. When blasts express CD19 and CD33 but MPO is <3%, diagnose B-ALL with aberrant CD33-not MPAL. If MPO reaches 3%, it's MPAL. This 3% MPO threshold determines treatment: ALL regimens for B-ALL (80-90% remission) versus AML regimens for MPAL (50-60% remission).
MDS vs AML vs MDS/MPN Overlap
-
Dysplasia with Increased Blasts
- MDS-EB2 (10-19% blasts): Treat as AML if fit
- Intensive chemotherapy: 40-50% CR rate, median 12-18 months OS
- Hypomethylating agents: 20-30% CR rate, median 18-24 months OS (better tolerability)
- Transplant in CR1: 40-50% 5-year OS
- AML with myelodysplasia-related changes (AML-MRC): ≥20% blasts plus dysplasia or MDS cytogenetics
- Worse prognosis than de novo AML: 20-30% 5-year OS versus 40-50%
- Requires ≥50% dysplasia in ≥2 lineages OR MDS-defining cytogenetics
- MDS-EB2 (10-19% blasts): Treat as AML if fit
-
Proliferative Dysplasia: MDS/MPN Overlap Syndromes
- Chronic myelomonocytic leukemia (CMML): Monocytes ≥1 × 10⁹/L and ≥10%, blasts <20%
- Dysplasia present in ≥1 lineage
- Median survival: 20-40 months, 15-30% AML transformation
- Proliferative (WBC >13 × 10⁹/L) worse than dysplastic subtype
- Atypical CML (aCML): Neutroph
- Chronic myelomonocytic leukemia (CMML): Monocytes ≥1 × 10⁹/L and ≥10%, blasts <20%
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app














