Cell Injury: Causes & Overview - Hurt Locker Cells
📌 VITAMIN C & D outlines causes:
- Vascular: Hypoxia (most common), Ischemia.
- Infective agents.
- Traumatic/Physical agents.
- Autoimmune reactions.
- Metabolic: Nutritional imbalances, genetic defects.
- Idiopathic/Iatrogenic.
- Neoplastic.
- Chemicals, Toxins, Drugs.
- Degenerative.
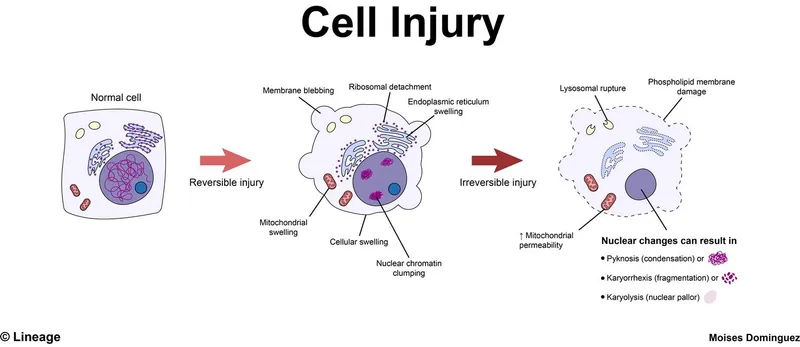
- Reversible Injury: Cellular swelling, fatty change; recovery possible if stimulus removed.
- Irreversible Injury: Leads to cell death (necrosis/apoptosis). Point of no return: severe mitochondrial dysfunction, profound membrane damage.
⭐ Hypoxia is the most common cause of cell injury.
Reversible Cell Injury - Cells on Pause
-
Key Mechanisms:
- ↓ATP (hypoxia, toxins): Impairs Na⁺-K⁺ pump, $Ca^{2+}$ homeostasis.
- Early Mitochondrial Damage: ↓ATP, ↑ROS.
- Influx of $Ca^{2+}$: Activates deleterious enzymes.
- Accumulation of Reactive Oxygen Species (ROS): Oxidative stress.
- Early Membrane Damage: Leakage of contents.
-
Morphological Features:
- Cellular Swelling (Hydropic Change/Vacuolar Degeneration): Due to Na⁺-K⁺ pump failure → water influx.
- Organs: Kidney, liver, heart, brain.
- Fatty Change (Steatosis): Abnormal triglyceride accumulation in parenchymal cells.
- Organs: Liver (classic), heart, muscle, kidney.
- Cellular Swelling (Hydropic Change/Vacuolar Degeneration): Due to Na⁺-K⁺ pump failure → water influx.
-
Ultrastructural Changes (EM):
- ER swelling, detachment of ribosomes (↓protein synthesis).
- Mitochondrial swelling with small amorphous densities.
- Plasma membrane blebs, blunting/loss of microvilli.
- Myelin figures (from damaged membranes).
⭐ Cellular swelling is the first manifestation of almost all forms of injury to cells.
Necrosis & Its Types - Cellular Demolition
- Irreversible cell injury: Point of no return. Severe mitochondrial vacuolization, extensive membrane damage (plasma & lysosomal), lysosomal rupture, nuclear changes (pyknosis → karyorrhexis → karyolysis).
- Necrosis: Spectrum of morphological changes following cell death in living tissue, largely from degradative action of enzymes on lethally injured cells.
- Key Mechanisms: Severe mitochondrial dysfunction (↓ATP, ↑ROS), profound membrane damage, denaturation of intracellular proteins, enzymatic digestion of cell (lysosomal enzymes).

- Morphological Types of Necrosis:
| Type | Key Features | Examples |
|---|---|---|
| Coagulative | Preserved cell outlines initially; anucleated, eosinophilic cells; firm texture | Myocardial infarction, kidney/spleen infarcts |
| Liquefactive | Viscous liquid mass; loss of tissue architecture; pus (if neutrophils) | Brain infarct, abscesses |
| Caseous | Cheese-like (friable white/yellow); granuloma; amorphous granular debris | Tuberculosis, fungal infections |
| Fat | Chalky white areas; saponification: $Ca^{2+} + \text{fatty acids} \rightarrow \text{calcium soaps}$ | Acute pancreatitis, trauma to breast |
| Fibrinoid | Eosinophilic, fibrin-like material in vessel walls; immune-mediated | Immune vasculitis, malignant hypertension |
| Gangrenous | Clinical term; often limb. Dry (coagulative), Wet (liquefactive + infection), Gas (Clostridial infection + gas bubbles) | Diabetic foot, frostbite |
⭐ Coagulative necrosis is the most common type, seen in hypoxic death of cells in all solid organs except the brain (where liquefactive necrosis occurs).
Apoptosis: Mechanisms & Morphology - Programmed Farewell
- Programmed cell death: energy-dependent, no inflammation (vs. necrosis).
- Examples:
- Physiological: Embryogenesis, endometrial shedding.
- Pathological: Viral infections (hepatitis), DNA damage, ER stress.
- Mechanisms:
- Intrinsic (Mitochondrial) Pathway:
- Triggered by cell stress.
- Bcl-2 family: Pro (BAX/BAK) vs. Anti (Bcl-2/Bcl-xL). 📌 BAX/BAK = Bad Actors Kill.
- ↑BAX/BAK → mitochondrial outer membrane permeabilization (MOMP) → Cytochrome c release.
- Apoptosome (Cyto c + Apaf-1) → Caspase-9 (initiator).
- Extrinsic (Death Receptor) Pathway:
- FASL/TNF-α binds FAS/CD95 or TNFR1.
- → Caspase-8 (initiator).
- Execution: Both activate Caspases-3, -6, -7 → cell dismantling.
- Intrinsic (Mitochondrial) Pathway:
- Morphology: Cell shrinkage; chromatin condensation (pyknosis), fragmentation (karyorrhexis); apoptotic bodies (membrane-bound); phagocytosis by macrophages (no significant inflammation).
⭐ Caspases (Cysteine proteases) are central executioners of apoptosis.

High‑Yield Points - ⚡ Biggest Takeaways
- Reversible injury: cellular swelling, fatty change.
- Irreversible injury: membrane damage, mitochondrial failure, nuclear dissolution (pyknosis, karyorrhexis, karyolysis).
- Apoptosis: programmed cell death, ATP-dependent, caspase-activated (intrinsic/extrinsic pathways), no inflammation.
- Necrosis: uncontrolled cell death, always pathological, triggers inflammation.
- Coagulative necrosis: common in ischemia (e.g., MI, kidney), except brain.
- Liquefactive necrosis: characteristic of brain infarcts and bacterial/fungal infections.
- Free radicals (ROS) cause significant oxidative stress and cell damage.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

