Neuro-Ophthalmology

On this page
👁️ Neuro-Ophthalmology: The Neural Vision Command Center
Vision begins not in the eye but in the brain's intricate command networks that orchestrate every aspect of sight-from pupillary reflexes to visual field processing. You'll master how neural pathways translate light into perception, identify the diagnostic fingerprints of optic neuropathies and cranial nerve palsies, and recognize vision-threatening emergencies that demand immediate intervention. This lesson builds your clinical reasoning from anatomical precision through therapeutic decision-making, transforming complex neuro-ophthalmology into a systematic framework you can confidently apply at the bedside.
👁️ Neuro-Ophthalmology: The Neural Vision Command Center
🧠 Visual System Architecture: The Brain's Precision Network
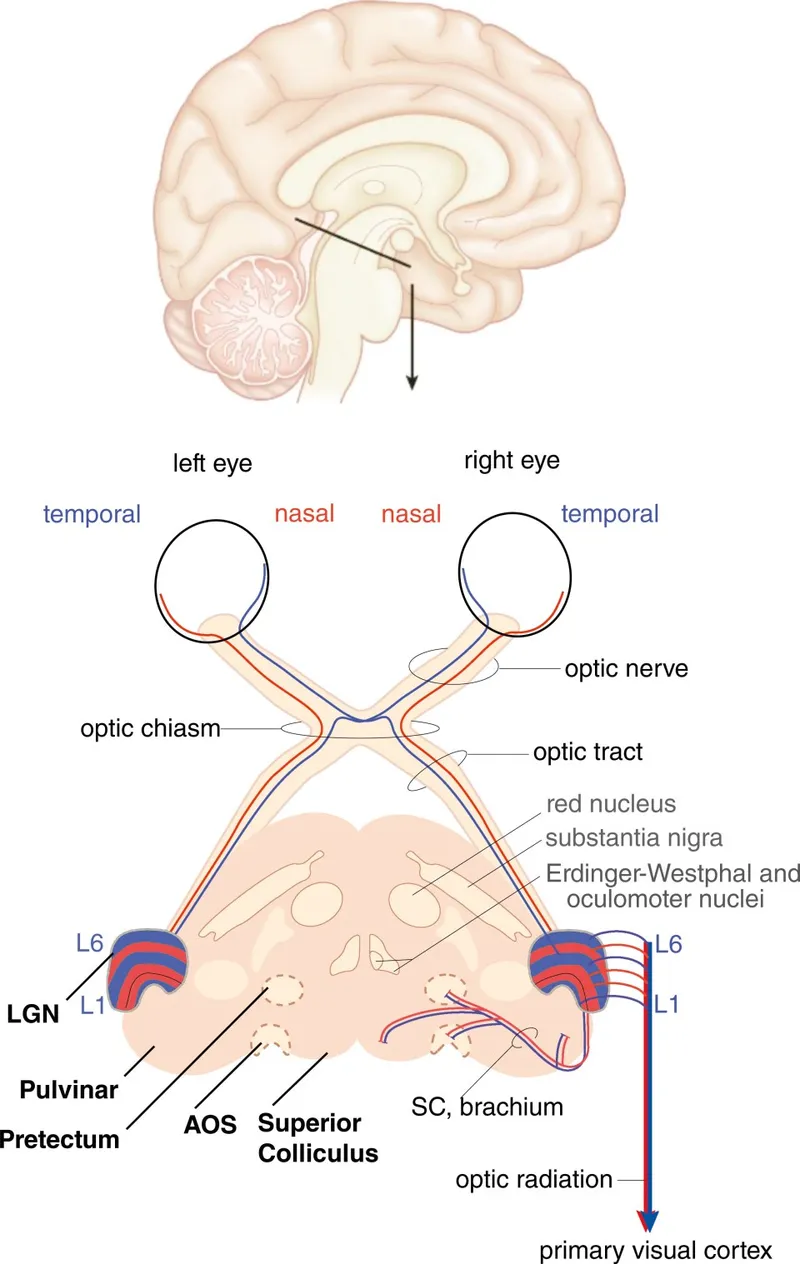
📌 Remember: RETINA - Retinal ganglion cells, Exit via optic disc, Temple fibers cross, Inferior radiations loop, Nasal fibers stay, Arrival at striate cortex
The visual pathway begins with 1.2 million retinal ganglion cells whose axons form the optic nerve, creating a 1.5mm diameter bundle containing precise retinotopic organization. Temporal retinal fibers (53% of total) remain ipsilateral at the chiasm, while nasal fibers (47%) decussate completely, establishing the foundation for binocular vision processing.
- Retinal Organization
- Foveal representation: 50% of visual cortex despite 2° visual field
- Ganglion cell density: 35,000 cells/mm² at fovea, 5,000 cells/mm² peripherally
- Magnocellular pathway: 10% of ganglion cells, motion/luminance detection
- Parvocellular pathway: 80% of ganglion cells, color/fine detail processing
- Koniocellular pathway: 10% of ganglion cells, blue-yellow color processing
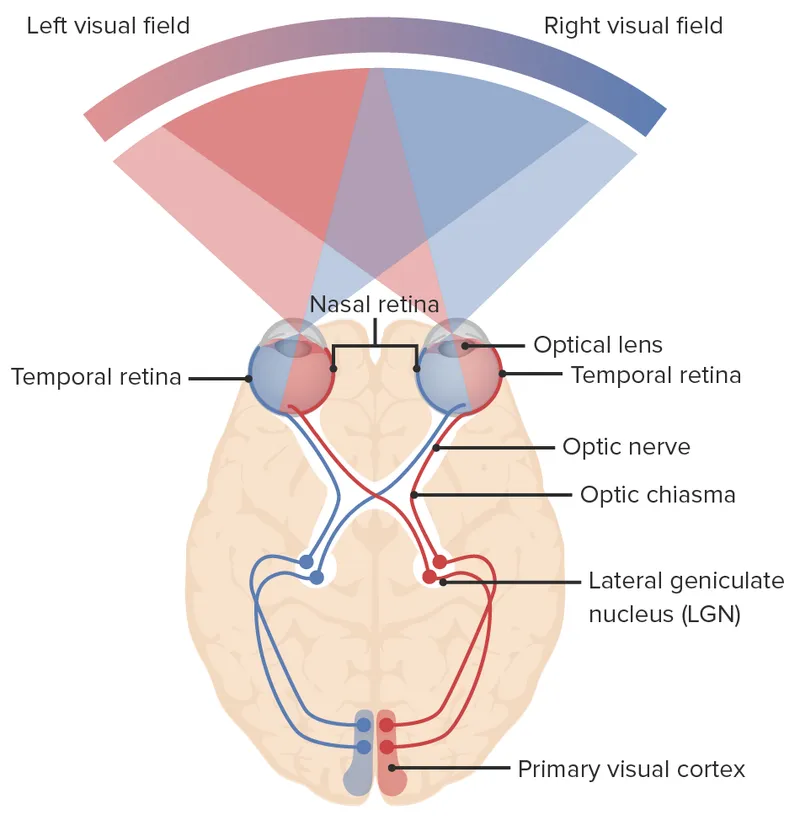
⭐ Clinical Pearl: Chiasmal lesions create bitemporal hemianopia in 85% of cases, with superior temporal quadrants affected first due to inferior chiasmal fiber vulnerability (p<0.001)
| Pathway Level | Fiber Count | Lesion Pattern | Field Defect | Pupil Response |
|---|---|---|---|---|
| Optic Nerve | 1.2 million | Unilateral | Monocular | RAPD positive |
| Optic Chiasm | 2.4 million | Central crossing | Bitemporal | RAPD variable |
| Optic Tract | 1.2 million | Homonymous | Incongruous | RAPD contralateral |
| LGN | 1.2 million | Homonymous | Congruous | Normal |
| Optic Radiations | 1.2 million | Homonymous | Congruous | Normal |
Understanding this architectural precision transforms pattern recognition, as every 2mm of pathway damage creates predictable visual field signatures that guide localization and diagnosis.
🧠 Visual System Architecture: The Brain's Precision Network
⚡ Pupillary Control Systems: The Autonomic Command Network
📌 Remember: PUPILS - Pretectal processes, Unilateral stimulation, Produces bilateral, Involves parasympathetic, Light and near, Sympathetic dilates
- Afferent Pupillary Pathway
- Retinal ganglion cells: 1-3% dedicated to pupillary function
- Pretectal nucleus: Bilateral projections to Edinger-Westphal nuclei
- Light response: Melanopsin-containing retinal ganglion cells
- Spectral sensitivity: Peak at 480nm (blue light)
- Conduction velocity: 2-8 m/sec via unmyelinated fibers
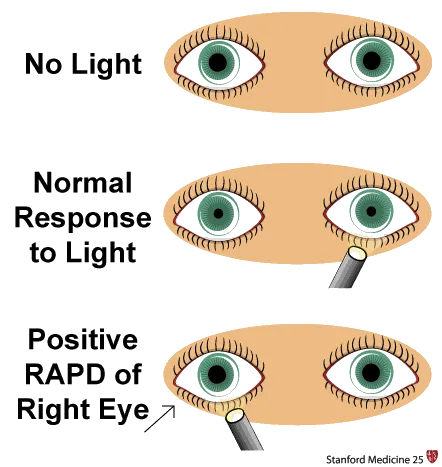
⭐ Clinical Pearl: RAPD detection requires ≥0.3 log units difference between eyes, corresponding to 20% retinal ganglion cell loss or equivalent optic nerve damage
- Efferent Pupillary Control
- Parasympathetic: Cranial nerve III, constriction via ciliary ganglion
- Sympathetic: 3-neuron pathway from hypothalamus to iris dilator
- First-order: Hypothalamus to C8-T2 spinal cord
- Second-order: T1 sympathetic chain to superior cervical ganglion
- Third-order: Carotid plexus to long ciliary nerves
| Pupillary Syndrome | Pupil Size | Light Response | Near Response | Pharmacologic Test |
|---|---|---|---|---|
| Adie's Tonic | Large (4-6mm) | Absent/sluggish | Present/tonic | Pilocarpine 0.1% positive |
| Horner's | Small (1-2mm) | Normal | Normal | Cocaine 4% positive |
| CN III Palsy | Large (6-8mm) | Absent | Absent | Pilocarpine 1% negative |
| Argyll Robertson | Small (1-3mm) | Absent | Present | Pilocarpine resistant |
| Pharmacologic | Variable | Absent | Absent | Specific to agent |
The near response involves convergence, accommodation, and miosis through separate neural pathways, explaining light-near dissociation patterns that distinguish between pretectal and parasympathetic lesions with 95% diagnostic accuracy.
⚡ Pupillary Control Systems: The Autonomic Command Network
🎯 Visual Field Mapping: The Diagnostic Fingerprint System
📌 Remember: FIELDS - Foveal sparing needs 1°, Incongruous suggests tract, Exact congruity means radiations, Lateral geniculate creates sectors, Double homonymous means bilateral, Scotomas central suggest macula
- Pathway-Specific Field Patterns
- Pre-chiasmal: Monocular defects respecting horizontal meridian
- Chiasmal: Bitemporal defects, superior quadrants first (85% cases)
- Central scotomas: Macular crossing fibers affected
- Junctional scotoma: Wilbrand's knee involvement (controversial)
- Temporal crescents: Monocular temporal field (30° zone)

⭐ Clinical Pearl: Incongruous homonymous defects indicate optic tract lesions with 90% accuracy, while congruous defects suggest occipital cortex involvement in 95% of cases
- Post-chiasmal Localization Rules
- Optic tract: Incongruous homonymous defects with RAPD
- LGN: Sectoral defects (rare, <1% of homonymous hemianopias)
- Lateral choroidal: Upper and lower quadrants
- Anterior choroidal: Lower quadrant with mild hemiparesis
- Temporal radiations: Superior quadrantanopia ("pie in the sky")
- Parietal radiations: Inferior quadrantanopia with neglect
- Occipital cortex: Congruous defects with macular sparing
| Lesion Location | Field Pattern | Congruity | Macular Involvement | Associated Signs |
|---|---|---|---|---|
| Optic Nerve | Monocular | N/A | Central scotoma | RAPD, disc pallor |
| Chiasm | Bitemporal | Variable | Central if large | Endocrine dysfunction |
| Optic Tract | Homonymous | Incongruous | Rare | Contralateral RAPD |
| Temporal Lobe | Superior quad | Congruous | Spared | Memory, language |
| Parietal Lobe | Inferior quad | Congruous | Involved | Neglect, apraxia |
| Occipital Lobe | Complete/partial | Congruous | Often spared | Isolated field defect |
Understanding progression patterns reveals etiology: Glaucoma creates nasal steps and arcuate scotomas, while neurologic lesions produce geographic defects respecting anatomical boundaries with sharp margins.
🎯 Visual Field Mapping: The Diagnostic Fingerprint System
🔍 Optic Neuropathy Classification: The Damage Assessment Matrix
📌 Remember: DISC SWAP - Demyelinating (neuritis), Ischemic (NAION/AION), Space-occupying (compression), Congenital (hereditary), Systemic (nutritional), Wallerian (secondary), Autoimmune (GCA), Papilledema (raised ICP)
- Acute Optic Neuropathies
- Optic neuritis: Peak age 20-40, female predominance 3:1
- Visual loss: Hours to days, pain with eye movement 90%
- Recovery: Spontaneous improvement begins 2-4 weeks
- MS association: 50% develop MS within 15 years
- NAION: Peak age >50, male predominance in younger patients
- Risk factors: Diabetes 25%, hypertension 50%, sleep apnea 70%
- Disc at risk: Small cup-to-disc ratio <0.3
- Fellow eye involvement: 15-20% within 5 years
- Optic neuritis: Peak age 20-40, female predominance 3:1
⭐ Clinical Pearl: Pain with eye movement occurs in 90% of optic neuritis cases but only 10% of NAION, providing 95% specificity for inflammatory etiology when combined with age <50 years
- Chronic Optic Neuropathies
- Compressive: Gradual onset, painless, progressive visual loss
- Meningioma: Optociliary shunt vessels in 30% of cases
- Pituitary adenoma: Bitemporal defects with superior quadrant onset
- Thyroid orbitopathy: Apical crowding with color vision loss
- Hereditary: Bilateral, central scotomas, family history
- Leber's: Males 85%, mitochondrial inheritance, age 15-35
- Dominant optic atrophy: Autosomal dominant, insidious onset
- Compressive: Gradual onset, painless, progressive visual loss
| Optic Neuropathy | Age Peak | Onset | Pain | Disc Appearance | Recovery |
|---|---|---|---|---|---|
| Optic Neuritis | 20-40 years | Hours-days | 90% present | Normal/swollen | Excellent |
| NAION | >50 years | Sudden | 10% present | Swollen/pale | Poor |
| AION (GCA) | >70 years | Sudden | Headache | Pale swollen | None |
| Compressive | Variable | Months | Rare | Normal/pale | Variable |
| Leber's | 15-35 years | Weeks | None | Hyperemic | Poor |
| Toxic/Nutritional | Variable | Months | None | Normal/pale | Good if early |
Color vision testing using Ishihara plates detects red-green deficiency in 95% of optic neuropathies, often preceding visual field changes by weeks to months in compressive and toxic etiologies.
🔍 Optic Neuropathy Classification: The Damage Assessment Matrix
⚖️ Cranial Nerve Palsy Management: The Therapeutic Decision Tree
📌 Remember: PALSY RULE - Pupil involvement suggests aneurysm, Acute onset needs imaging, Lasting >3 months needs MRI, Systemic symptoms need workup, Young patients need full evaluation, Recurrent episodes suggest myasthenia, Unilateral suggests nerve, Lateral gaze palsy suggests VI, Elevation loss suggests IV
- CN III Palsy Evaluation
- Pupil-involving: Aneurysm risk 25-30% in patients <50 years
- Posterior communicating artery: Most common aneurysmal cause
- Urgent imaging: CTA or MRA within 6 hours of onset
- Surgical consultation: Neurosurgery for confirmed aneurysm
- Pupil-sparing: Microvascular etiology in 85% if age >50 + diabetes
- Recovery timeline: 6-12 weeks for complete resolution
- Imaging indicated: No improvement by 3 months
- Pupil-involving: Aneurysm risk 25-30% in patients <50 years

⭐ Clinical Pearl: Pupil-sparing CN III palsy in diabetic patients >50 has 95% probability of microvascular etiology, while pupil involvement in patients <50 requires urgent aneurysm evaluation with CTA/MRA
- CN VI Palsy Management
- Most common: Microvascular in adults, increased ICP in children
- Bilateral CN VI: Raised ICP until proven otherwise
- Papilledema present: 85% have increased ICP
- Lumbar puncture: Opening pressure >25 cmH2O diagnostic
- Recovery: 80% resolve within 6 months if microvascular
| Cranial Nerve | Primary Action | Common Etiology | Associated Signs | Recovery Rate |
|---|---|---|---|---|
| CN III | Elevation, adduction | Microvascular 60% | Ptosis, pupil | 85% by 6 months |
| CN IV | Intorsion, depression | Trauma 40% | Head tilt | 75% by 12 months |
| CN VI | Abduction | Microvascular 50% | Horizontal diplopia | 80% by 6 months |
| Multiple | Variable | Cavernous sinus | Trigeminal numbness | Variable |
| Bilateral VI | Bilateral abduction | Increased ICP | Papilledema | Depends on cause |
Treatment algorithms prioritize prism glasses for stable deviations <15 prism diopters, botulinum toxin for acute large deviations, and surgical correction for persistent diplopia >6 months with stable measurements for 3 consecutive months.
⚖️ Cranial Nerve Palsy Management: The Therapeutic Decision Tree
🔗 Neuro-Ophthalmic Emergency Recognition: The Critical Decision Matrix
📌 Remember: EMERGENCY - ESR >50 suggests GCA, Mydriasis with CN III needs CTA, Edema bilateral suggests ICP, RAPD with normal disc needs MRI, Gradual loss needs imaging, Eye pain with vision loss urgent, New diplopia needs workup, Cortical blindness needs stroke protocol, Young with neuritis needs LP
- Vision-Threatening Emergencies
- Giant Cell Arteritis: Age >50, ESR >50, jaw claudication
- Visual loss: Sudden, painless, irreversible in 95%
- Temporal artery: Tender, enlarged, absent pulse in 60%
- Treatment: IV methylprednisolone 1g daily × 3 days immediately
- Biopsy: Within 7 days of steroid initiation, positive in 85%
- Pituitary apoplexy: Sudden headache, bitemporal hemianopia, diplopia
- Endocrine crisis: Hypotension, hyponatremia, hypoglycemia
- Imaging: MRI shows hemorrhage/infarction in enlarged sella
- Treatment: Hydrocortisone 100mg q8h, urgent neurosurgery consultation
- Giant Cell Arteritis: Age >50, ESR >50, jaw claudication
⭐ Clinical Pearl: Bilateral visual loss with headache and altered mental status suggests posterior circulation stroke requiring immediate stroke protocol activation within 4.5-hour window for thrombolytic therapy
- Life-Threatening Presentations
- Aneurysmal CN III palsy: Pupil involvement + severe headache
- Subarachnoid hemorrhage: Thunderclap headache in 85%
- Imaging: CTA sensitivity 95% for aneurysms >3mm
- Intervention: Endovascular coiling or surgical clipping within 24 hours
- Cavernous sinus thrombosis: Proptosis, chemosis, multiple CN palsies
- Infection source: Sphenoid sinusitis in 70%, facial cellulitis in 20%
- Treatment: IV antibiotics + anticoagulation + surgical drainage
- Aneurysmal CN III palsy: Pupil involvement + severe headache
| Emergency Condition | Key Features | Diagnostic Test | Treatment Window | Intervention |
|---|---|---|---|---|
| Giant Cell Arteritis | ESR >50, jaw claudication | Temporal artery biopsy | <24 hours | IV steroids |
| Aneurysmal CN III | Pupil involved, headache | CTA/MRA | <6 hours | Neurosurgery |
| Pituitary Apoplexy | Bitemporal VF, endocrine | MRI pituitary | <24 hours | Steroids + surgery |
| Cavernous Thrombosis | Multiple CN, proptosis | MRI/MRV | <12 hours | Antibiotics + anticoagulation |
| Posterior Stroke | Cortical blindness | MRI DWI | <4.5 hours | Thrombolytics |
| Papilledema | Bilateral disc swelling | LP opening pressure | <24 hours | ICP reduction |
Triage protocols emphasize ABC assessment followed by rapid visual acuity, pupillary examination, and fundoscopy within 5 minutes of presentation, enabling risk stratification that guides immediate versus urgent versus routine management pathways.
🔗 Neuro-Ophthalmic Emergency Recognition: The Critical Decision Matrix
🎯 Neuro-Ophthalmic Mastery: The Clinical Excellence Arsenal
📌 Essential Numbers Arsenal: ESR >50 (GCA screening), 0.3 log units (RAPD threshold), 53:47 (temporal:nasal fiber ratio), 15-20% (fellow eye NAION risk), 90% (pain with movement in neuritis), 95% (pupil-sparing CN III microvascular if diabetic >50)
- Rapid Assessment Protocol
- Visual acuity: Snellen or counting fingers bilateral
- Pupillary examination: RAPD testing with swinging flashlight
- Visual fields: Confrontation or formal perimetry if indicated
- Fundoscopy: Optic disc assessment for swelling/pallor/cupping
- Motility: Extraocular movements and diplopia evaluation
| Clinical Scenario | Key Discriminator | Urgent Action | Diagnostic Yield |
|---|---|---|---|
| Sudden visual loss + jaw pain | ESR >50, age >70 | IV steroids + biopsy | 90% GCA detection |
| CN III + pupil involvement | Age <50, headache | CTA within 6 hours | 95% aneurysm detection |
| Bilateral disc swelling | Normal vision | LP opening pressure | 85% ICP elevation |
| Homonymous hemianopia | Acute onset | MRI DWI stroke protocol | 95% stroke detection |
| Progressive bitemporal VF | Endocrine symptoms | MRI pituitary | 90% adenoma detection |
💡 Master This: Neuro-ophthalmic excellence combines anatomical precision with clinical urgency - every visual complaint demands systematic evaluation that can save vision and save lives through early recognition of treatable conditions
Understanding these integrated frameworks enables confident management of complex presentations while minimizing diagnostic delays that lead to irreversible visual loss in time-sensitive conditions.
🎯 Neuro-Ophthalmic Mastery: The Clinical Excellence Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























