Glaucoma

On this page
🌊 The Aqueous Architecture: Fluid Factory to Drainage Destiny
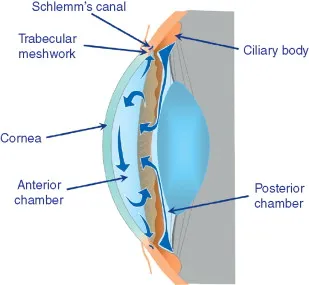
Glaucoma fundamentally represents a failure of aqueous humor balance-either overproduction, impaired outflow, or both. Understanding the 2.5 μL/min production rate and the two distinct drainage pathways unlocks every therapeutic target and explains why different glaucoma subtypes demand different approaches.
The Production Powerhouse: Ciliary Body Machinery
The non-pigmented ciliary epithelium generates aqueous through three mechanisms, with active secretion accounting for 80-90% of total production:
-
Active Secretion (dominant pathway)
- Na⁺-K⁺-ATPase pumps drive sodium into posterior chamber
- Water follows osmotic gradient via aquaporin channels
- Carbonic anhydrase generates bicarbonate to maintain pH
- Energy-dependent: blocked by carbonic anhydrase inhibitors (CAIs)
- Temperature-sensitive: decreases 2.5% per 1°C drop
-
Ultrafiltration (passive component)
- Driven by hydrostatic pressure gradient across capillaries
- Contributes 10-15% of total production
- Modified by blood pressure fluctuations
- Explains nocturnal IOP spikes during supine positioning
-
Diffusion (minimal contribution)
- Small molecules cross concentration gradients
- Clinically negligible in healthy eyes
- May increase with blood-aqueous barrier breakdown
📌 Production Mnemonic: "SAUD" - Secretion (active, 80%), ATPase-driven, Ultrafiltration (passive, 15%), Diffusion (negligible). Secretion is the surgical target (cyclodestruction), ultrafiltration explains positional IOP changes.
⭐ Clinical Pearl: Aqueous production follows a circadian rhythm with peak rates in early morning (2.4-2.8 μL/min) and trough at night (1.5-2.0 μL/min). This explains why IOP measurements vary 3-6 mmHg throughout the day, with highest values typically between 6 AM-10 AM in 60-70% of patients.
The Drainage Divide: Conventional vs Uveoscleral Routes
Aqueous exits via two anatomically and pharmacologically distinct pathways:
| Feature | Conventional (Trabecular) | Uveoscleral (Alternative) | Clinical Significance |
|---|---|---|---|
| Outflow % | 75-85% baseline | 15-25% baseline | Trabecular route is primary therapeutic target |
| Pathway | Trabecular meshwork → Schlemm canal → collector channels → episcleral veins | Through ciliary muscle interstices → suprachoroidal space → sclera | Uveoscleral enhanced by prostaglandin analogs |
| Pressure-sensitive | Yes-increases with higher IOP | Relatively pressure-independent | Explains why PGAs work even at low IOP |
| Age effect | Decreases 0.5%/year after age 20 | Decreases 50% by age 80 | Age is major POAG risk factor |
| Drug targets | Pilocarpine (↑ via trabecular stretch), Laser trabeculoplasty | Latanoprost (↑ by 50-100%), Atropine (↓ by relaxing ciliary muscle) | PGAs are first-line medical therapy |
| Resistance site | Juxtacanalicular meshwork (inner wall of Schlemm canal) | Ciliary muscle compactness | POAG pathology localizes to juxtacanalicular tissue |
💡 Master This: The juxtacanalicular meshwork (also called cribriform region) generates 75-90% of total outflow resistance despite comprising only the innermost 2-4 μm of trabecular tissue. This explains why laser trabeculoplasty targets this zone and why POAG pathology centers here-extracellular matrix accumulation and trabecular cell dysfunction specifically affect this critical bottleneck.
IOP Regulation: The Goldilocks Equation
Normal IOP (10-21 mmHg, mean 15.5 mmHg) reflects dynamic equilibrium:
$$\text{IOP} = \frac{\text{Aqueous Production Rate}}{\text{Outflow Facility}} + \text{Episcleral Venous Pressure}$$
-
Outflow facility (C): Normal 0.25-0.30 μL/min/mmHg
- Decreases to 0.10-0.15 in POAG (50% reduction)
- Each 0.01 decrease raises IOP by approximately 2-3 mmHg
-
Episcleral venous pressure (EVP): Baseline 8-10 mmHg
- Relatively constant in health
- Elevated in carotid-cavernous fistula (↑ to 15-20 mmHg)
- Cannot be pharmacologically reduced (surgical drainage only option)
⭐ Clinical Pearl: Episcleral venous pressure sets the floor for achievable IOP-medical therapy cannot lower IOP below EVP regardless of aqueous suppression. This explains why eyes with elevated EVP (Sturge-Weber syndrome, thyroid eye disease) require surgery, not just maximal medical therapy.
Pharmacological Pressure Points: Drug Target Map
-
Aqueous Suppressors (reduce production)
- Beta-blockers: ↓ secretion 20-30%, reduce IOP 20-25%
- Alpha-2 agonists: ↓ secretion 30-50%, ↑ uveoscleral outflow 30-40%
- Carbonic anhydrase inhibitors: ↓ secretion 40-60%, reduce IOP 15-25%
-
Outflow Enhancers
- Prostaglandin analogs: ↑ uveoscleral flow 50-100%, reduce IOP 25-35% (most potent class)
- Pilocarpine: ↑ trabecular outflow via ciliary muscle contraction, reduce IOP 15-25%
- Rho kinase inhibitors: ↓ trabecular resistance, reduce IOP 15-20%
📌 Drug Mnemonic: "PGA-1st BAC" - Prostaglandin analogs are 1st-line (most effective, once-daily), then Beta-blockers, Alpha agonists, Carbonic anhydrase inhibitors as adjuncts or alternatives.
🌊 The Aqueous Architecture: Fluid Factory to Drainage Destiny
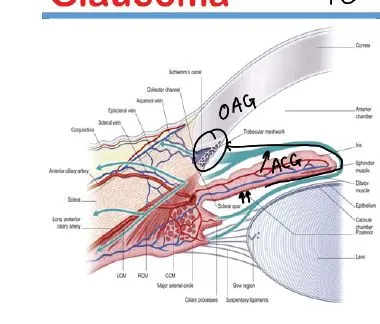
🔍 Primary Open-Angle Glaucoma: The Silent Optic Nerve Assassin
POAG represents 60-70% of all glaucoma cases globally and 50% in India, characterized by progressive optic neuropathy with open anterior chamber angles and no identifiable secondary cause. The disease remains asymptomatic until 40-50% of retinal ganglion cells are lost, making screening and early detection critical.
Pathophysiological Cascade: Pressure, Perfusion, and Cell Death
POAG results from multifactorial retinal ganglion cell (RGC) death centered at the lamina cribrosa:
-
Mechanical Theory (IOP-dependent damage)
- Elevated IOP compresses axons at lamina cribrosa
- Posterior bowing of lamina disrupts axoplasmic flow
- Axonal ischemia triggers RGC apoptosis
- Explains why IOP reduction slows progression in 100% of cases
-
Vascular Theory (perfusion-dependent damage)
- Reduced ocular perfusion pressure (OPP = MAP - IOP)
- OPP below 50 mmHg associated with 3× higher POAG risk
- Nocturnal hypotension during sleep ("dippers") shows faster progression
- Explains normal-tension glaucoma (NTG) with IOP <21 mmHg
-
Molecular Mechanisms
- Glutamate excitotoxicity from impaired clearance
- Oxidative stress and mitochondrial dysfunction
- Neuroinflammation with glial activation
- Explains therapeutic targets beyond IOP reduction
💡 Master This: POAG is fundamentally a lamina cribrosa disease-the sieve-like connective tissue at the optic nerve head is the primary site of axonal damage. Eyes with thinner lamina (<200 μm), larger cup-to-disc ratios, and deeper laminar insertion are at 2-4× higher risk, explaining why myopic eyes with tilted discs and peripapillary atrophy progress faster.
Risk Factor Stratification: Building Your Predictive Model
| Risk Factor | Magnitude | Mechanism | Clinical Action |
|---|---|---|---|
| Elevated IOP | Each 1 mmHg ↑ = 10-12% ↑ risk | Direct mechanical and vascular damage | Target ≥25% reduction from baseline |
| Age >40 years | Risk doubles every 10 years | Cumulative trabecular dysfunction | Screen every 1-2 years after age 40 |
| Family history | 4-9× higher risk with first-degree relative | Genetic susceptibility (MYOC, OPTN genes) | Screen relatives annually starting age 35 |
| African ancestry | 3-4× higher prevalence, earlier onset | Thinner corneas, larger cups, genetic factors | Start screening age 30-35 |
| Myopia >3 D | 2-3× higher risk | Thinner lamina, peripapillary atrophy, longer axial length | Annual dilated exams, OCT monitoring |
| Thin CCT <555 μm | Each 40 μm ↓ = 71% ↑ risk | Underestimates true IOP, weaker biomechanics | Adjust IOP targets downward 2-4 mmHg |
| Diabetes mellitus | 1.5× higher risk | Microvascular disease, impaired autoregulation | Optimize glycemic control, aggressive IOP targets |
⭐ Clinical Pearl: The Ocular Hypertension Treatment Study (OHTS) demonstrated that treating IOP >24 mmHg reduces 5-year conversion to POAG from 9.5% to 4.4% (54% risk reduction). However, 10% of glaucoma patients have IOP consistently <21 mmHg (normal-tension glaucoma), emphasizing that POAG is defined by optic nerve damage, not pressure thresholds.
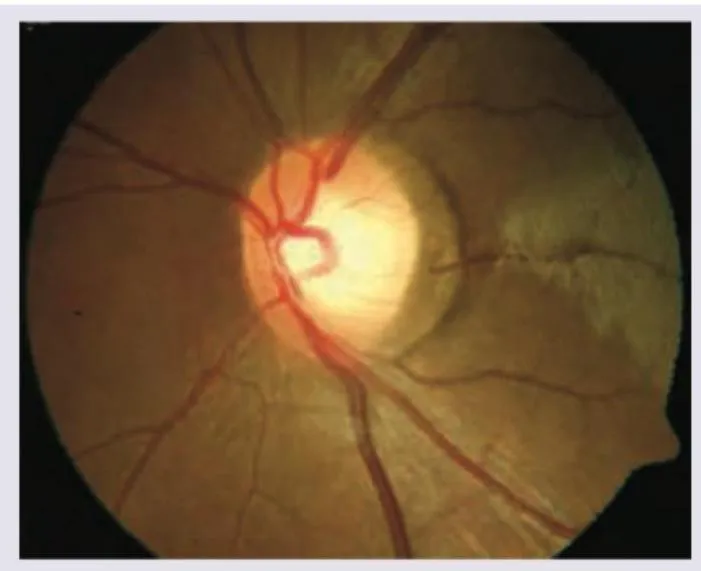
Diagnostic Triad: Structural, Functional, and Pressure Assessment
POAG diagnosis requires integration of three domains:
-
Optic Nerve Head (ONH) Assessment
- Vertical cup-to-disc ratio (VCDR) >0.6 or asymmetry >0.2 between eyes
- ISNT rule violation: Normal rim thickest Inferior > Superior > Nasal > Temporal
- Disc hemorrhages (Drance hemorrhages): Present in 4-7%, predict progression
- Peripapillary atrophy: Beta-zone (visible sclera/choroid) correlates with damage
- Bayonetting sign: Sharp angulation of vessels at cup margin
-
Retinal Nerve Fiber Layer (RNFL) Defects
- Wedge-shaped slit defects or diffuse thinning
- Best seen with red-free (green) light photography
- OCT quantifies thickness: Normal 90-110 μm, glaucoma often <70 μm
- Temporal-superior-nasal-inferior-temporal (TSNIT) curve shows peaks and valleys
-
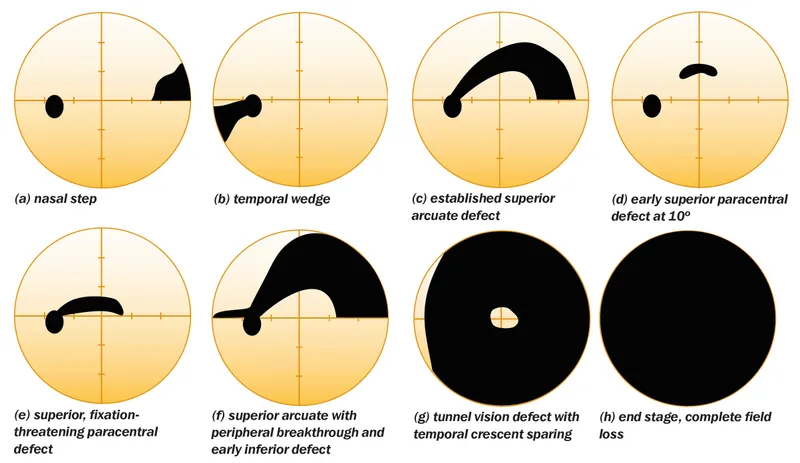
Visual Field Defects (perimetry)
- Paracentral scotomas within 10° of fixation (earliest)
- Arcuate (Bjerrum) scotomas respecting horizontal midline
- Nasal step (Roenne's): Superior and inferior arcuates at different eccentricities
- Temporal wedge: Extends from blind spot
- Advanced: Ring scotoma or central island with peripheral loss
📌 POAG Diagnosis Mnemonic: "FIND POAG" - Family history, IOP elevation (but not required), Nerve damage (cupping, hemorrhages), Defects in visual field, Progressive loss, Open angles on gonio, Absence of secondary causes, Glaucomatous RNFL thinning. All 4 core elements (nerve + field + open angle + no secondary cause) must be present.
Staging and Progression Monitoring
-
Hodapp-Parrish-Anderson Classification (visual field severity)
- Early: MD better than -6 dB, <18 points <5% probability
- Moderate: MD -6 to -12 dB, <37 points <5%, no central point <15 dB
- Advanced: MD worse than -12 dB, central points <0 dB, or only central island remaining
-
Progression Detection (requires ≥5 reliable fields over ≥2 years)
- Event-based: ≥3 points worsen by ≥5 dB on consecutive tests
- Trend-based: MD slope <-1 dB/year indicates fast progression
- OCT RNFL: Loss >4 μm/year or >2 μm/year in fast vs slow progressors
⭐ Clinical Pearl: Structure precedes function-OCT RNFL thinning typically detectable 2-4 years before reproducible visual field defects appear. However, once VF loss is established, functional progression (VF MD slope) often correlates better with clinical outcomes than structural changes, because floor effects limit OCT sensitivity in advanced disease.
🔍 Primary Open-Angle Glaucoma: The Silent Optic Nerve Assassin
⚡ Primary Angle-Closure Glaucoma: The Anatomical Ambush
PACG accounts for 50% of glaucoma blindness worldwide and shows 2-3× higher prevalence in Asian populations. Unlike POAG's gradual onset, PACG can present as an ophthalmic emergency with acute angle closure (AAC) causing irreversible damage within 24-48 hours if untreated.
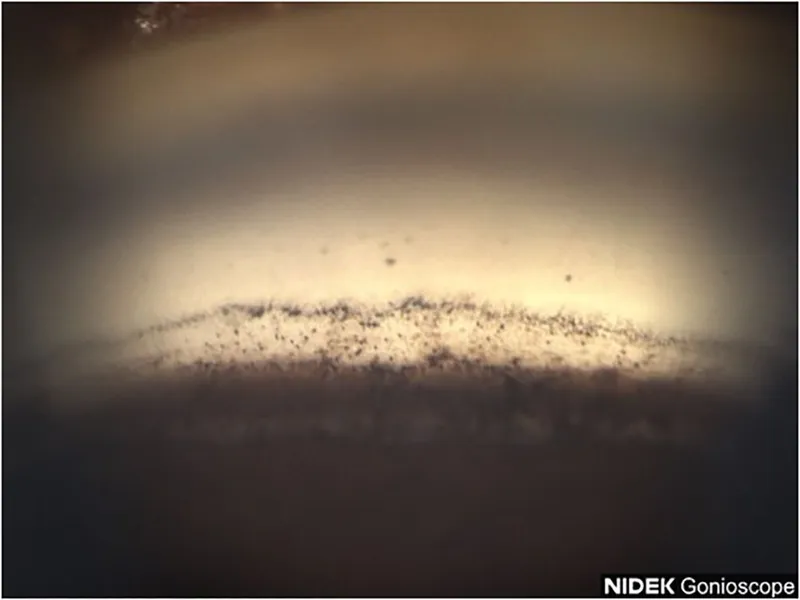
Anatomical Predisposition: The Crowded Anterior Segment
PACG results from anatomical crowding that blocks aqueous drainage through the trabecular meshwork:
-
Primary Anatomical Risk Factors
- Short axial length <22 mm (hyperopia): 5-10× higher risk
- Shallow anterior chamber depth <2.5 mm: 3-6× higher risk
- Thick lens with high lens/axial length ratio
- Small corneal diameter <11 mm
- Anterior lens position (relative to ciliary body)
-
Secondary Anatomical Contributors
- Plateau iris configuration: Anteriorly positioned ciliary processes
- Narrow angle recess with steep iris insertion
- Increased lens thickness with aging (0.02 mm/year)
-
Demographic Patterns
- Women > Men (2-3:1 ratio): Smaller eyes, shallower AC
- Age >50 years: Lens thickening narrows angle by 12-15° per decade
- East Asian ancestry: 3-4× higher prevalence vs Caucasians
📌 Angle-Closure Mnemonic: "PACG = Pupil Apposition Creates Glaucoma" - Pupillary block (90%), Anterior lens position, Ciliary process position (plateau iris), Growing lens thickness. The common pathway is iris-trabecular contact preventing aqueous drainage.
Mechanisms of Angle Closure: Four Pathways to Obstruction
-
Pupillary Block (most common, 90% of cases)
- Aqueous flow from posterior to anterior chamber blocked at pupil
- Pressure gradient pushes iris forward (iris bombé)
- Mid-dilated pupil (4-6 mm) creates maximal lens-iris contact
- Laser peripheral iridotomy (LPI) curative in >95%
-
Plateau Iris Configuration
- Anteriorly rotated ciliary processes mechanically support peripheral iris
- Angle remains narrow despite patent iridotomy
- Diagnosed by ultrasound biomicroscopy (UBM) or anterior segment OCT
- Requires laser iridoplasty or long-term pilocarpine
-
Phacomorphic Mechanism
- Intumescent (swollen) cataractous lens pushes iris forward
- Lens thickness >5 mm in 80% of cases
- Lens extraction is definitive treatment
-
Lens Subluxation
- Anteriorly dislocated lens (trauma, Marfan, homocystinuria)
- Creates reverse pupillary block if lens moves into anterior chamber
- Requires surgical lens removal
💡 Master This: The "dark room prone provocative test" exploits pupillary block physiology-having patient lie prone in darkness for 60 minutes causes pupil dilation and anterior lens shift. IOP rise >8 mmHg or angle closure on gonioscopy indicates high acute attack risk. However, sensitivity is only 50-60%, limiting clinical use; anterior segment OCT showing angle opening distance <500 μm is more reliable.
Clinical Presentations: From Silent to Catastrophic
| Stage | Symptoms | IOP | Angle Status | Management | Urgency |
|---|---|---|---|---|---|
| Primary Angle-Closure Suspect (PACS) | None | Normal | Occludable (≤180° contact) | LPI prophylaxis | Elective |
| Primary Angle-Closure (PAC) | Intermittent blurred vision, halos | Normal to elevated | Closed with PAS or IOP elevation | LPI + medical therapy | Semi-urgent |
| Primary Angle-Closure Glaucoma (PACG) | Progressive vision loss | Elevated | Closed with PAS | LPI + medications ± surgery | Urgent |
| Acute Angle-Closure (AAC) | Severe pain, N/V, vision loss | >30 mmHg (often 40-80) | Completely closed | Emergency: IOP lowering → LPI | EMERGENCY |
| Chronic Angle-Closure | Gradual vision loss | Persistently elevated | Synechial closure (PAS >270°) | Surgery (trabeculectomy/tube) | Urgent |
Acute Angle-Closure Crisis: The 6-Hour Window
AAC is an ophthalmic emergency requiring treatment within 6 hours to prevent permanent vision loss:
-
Classic Presentation (triad in >90%)
- Severe ocular pain with headache, nausea, vomiting
- Blurred vision with colored halos around lights
- Red eye with conjunctival injection, corneal edema
-
Examination Findings
- IOP 40-80 mmHg (often >60 mmHg)
- Mid-dilated, fixed pupil (4-6 mm) unreactive to light
- Corneal edema (microcystic epithelial edema)
- Shallow anterior chamber with closed angle on gonioscopy
- Ciliary injection (perilimbal flush)
-
Medical Management Protocol (sequential steps)
- Immediate IOP reduction:
- Acetazolamide 500 mg IV or 2× 250 mg PO
- Topical beta-blocker (timolol 0.5%) + alpha-agonist (brimonidine 0.2%)
- Hyperosmotic agent: Mannitol 1-2 g/kg IV over 45 min or oral glycerol 1-1.5 g/kg
- Break attack once IOP <30 mmHg:
- Pilocarpine 2% every 15 min × 2 doses (ineffective if IOP >40 mmHg-ischemic iris)
- Topical steroid (prednisolone 1%) every 1 hour
- Definitive treatment within 24-48 hours:
- Laser peripheral iridotomy (LPI) to both eyes (fellow eye 40-80% risk within 5 years)
- Immediate IOP reduction:
⭐ Clinical Pearl: Pilocarpine paradox-miotics are ineffective when IOP exceeds 40-50 mmHg because iris sphincter ischemia prevents contraction. Always reduce IOP below 30 mmHg with systemic and topical aqueous suppressants before instilling pilocarpine, or you risk worsening angle closure by increasing lens-iris contact without achieving pupil constriction.
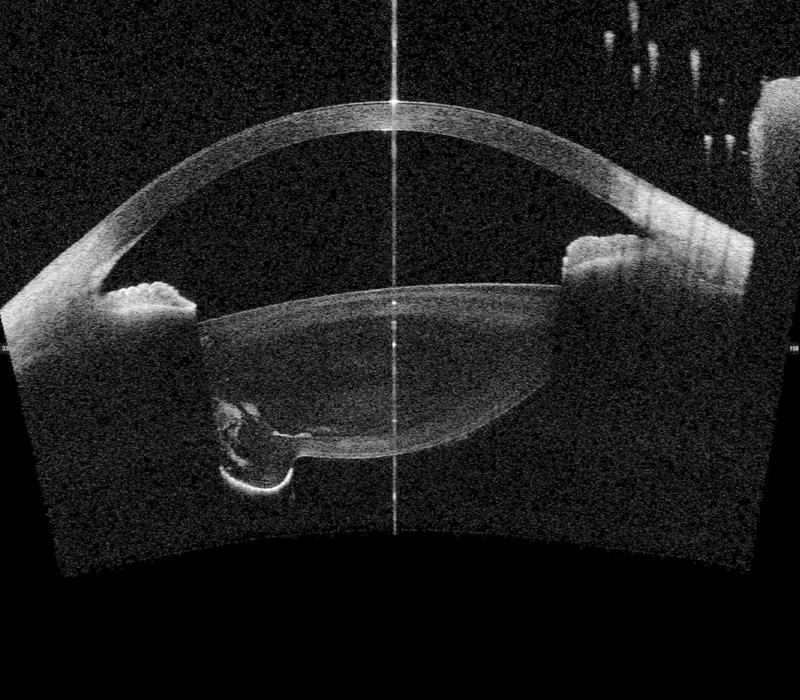
Laser Peripheral Iridotomy: Breaking the Block
LPI creates an alternative pathway for aqueous, eliminating pupillary block:
-
Technique Essentials
- Nd:YAG laser most common (2-10 mJ, 1-3 bursts)
- Location: Superior peripheral iris (11-1 o'clock), covered by upper lid
- Size: Patent opening 150-200 μm
- Endpoint: Pigment plume, visible lens capsule, deepening of AC
-
Success Rates
- >95% effective for pupillary block angle closure
- 50-60% effective for plateau iris (adjunct iridoplasty needed)
- Fellow eye prophylaxis reduces AAC risk from 50% to <5% over 5 years
-
Complications (usually transient)
- IOP spike (5-10 mmHg) in 20-30% at 1 hour (pre-treat with apraclonidine)
- Bleeding at iridotomy site (usually self-limited)
- Corneal burns (avoid excessive energy)
- Chronic glare/dysphotopsia in <5% (minimize iridotomy size)
📌 LPI Location Mnemonic: "SCUT" - Superior iris (11-1 o'clock), Covered by lid (avoids glare), Under iris crypt (thinner tissue), Temporal quadrant if superior not feasible. Avoid 3-9 o'clock (horizontal meridian) to prevent monocular diplopia.
⚡ Primary Angle-Closure Glaucoma: The Anatomical Ambush
🔬 Secondary Glaucomas: When Glaucoma Has a Cause
Secondary glaucomas result from identifiable ocular or systemic conditions that elevate IOP through distinct mechanisms. Recognizing the underlying cause is critical because treatment often targets the primary disease, not just IOP reduction.
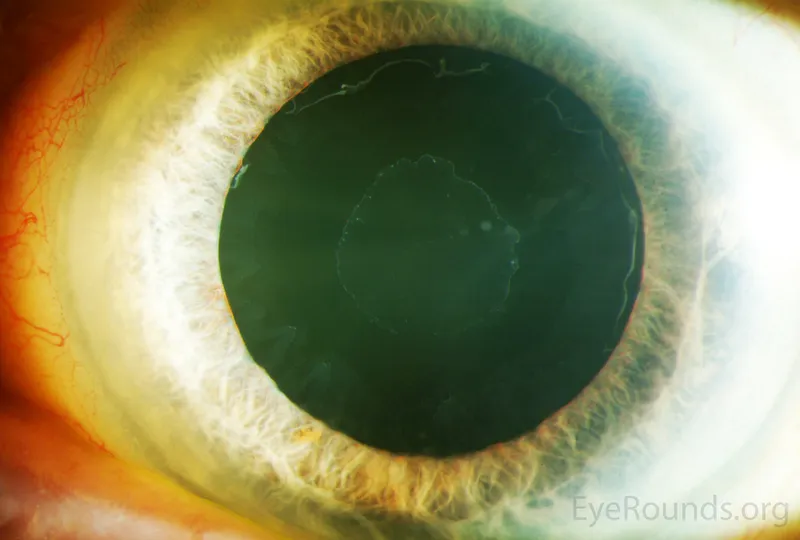
Secondary Open-Angle Glaucomas: Trabecular Obstruction
-
Pseudoexfoliation Glaucoma (most common secondary OAG)
- Prevalence: 20-30% of glaucoma in Scandinavia, 10-15% in India
- Mechanism: White flaky material (abnormal extracellular matrix) clogs trabecular meshwork
- Clinical signs:
- Target pattern on anterior lens capsule (central disc, clear zone, peripheral granular zone)
- Sampaolesi line: Pigment deposition anterior to Schwalbe line
- Poor pupil dilation, phacodonesis (lens instability)
- IOP characteristics: Higher baseline (often >25 mmHg), greater fluctuations (>10 mmHg diurnal variation)
- Prognosis: 2× faster progression than POAG, 3× higher surgical complication rates
- Management: More aggressive IOP targets (<15 mmHg), early surgery consideration
-
Pigment Dispersion Syndrome (PDS) / Pigmentary Glaucoma
- Demographics: Young myopic males (20-40 years), 80-90% bilateral
- Mechanism: Posterior iris bowing rubs against zonules, liberating pigment that clogs TM
- Classic triad:
- Krukenberg spindle: Vertical pigment on corneal endothelium
- Iris transillumination defects: Radial spoke-like pattern mid-periphery
- Dense trabecular pigmentation: 360° homogeneous dark band
- IOP spikes: After exercise, pupil dilation (pigment showers)
- Management: Pilocarpine prevents iris-lens contact, laser iridotomy controversial (30-50% benefit)
-
Steroid-Induced Glaucoma
- Prevalence: 30-40% of population are "steroid responders" (IOP ↑ >6 mmHg)
- Mechanism: Glucocorticoids ↓ trabecular outflow facility by ↑ extracellular matrix, ↓ phagocytosis
- Risk factors: POAG, diabetes, high myopia, connective tissue disorders
- Onset: 2-6 weeks after starting steroids (topical, periocular, systemic, inhaled)
- Management: Discontinue steroids if possible, switch to loteprednol or fluorometholone (weaker IOP effect), treat like POAG
-
Uveitic Glaucoma
- Mechanisms: Trabecular inflammation/debris, PAS formation, steroid-induced
- Anterior uveitis: Inflammatory cells/fibrin block TM, PAS with chronicity
- Fuchs heterochromic iridocyclitis: Chronic low-grade inflammation, 10-15% develop glaucoma
- Management: Control inflammation first (steroids), then IOP; avoid miotics (worsen inflammation)
💡 Master This: Pseudoexfoliation and pigmentary glaucoma both involve trabecular meshwork obstruction by particles, but their demographics and prognosis differ dramatically. PXF affects older patients (>60 years), progresses rapidly, and has poor surgical outcomes due to weak zonules and dense cataracts. PDS affects younger myopes (20-40 years), often burns out after age 50 as lens thickens and iris flattens, and responds well to surgery.
Secondary Angle-Closure Glaucomas: Pushing or Pulling the Iris
-
Neovascular Glaucoma (NVG) ("90-day glaucoma")
- Mechanism: Retinal ischemia → VEGF release → iris/angle neovascularization → fibrovascular membrane contracts → synechial angle closure
- Causes: Proliferative diabetic retinopathy (33%), central retinal vein occlusion (50% in ischemic type), ocular ischemic syndrome
- Stages:
- Pre-rubeosis: Retinal ischemia without iris NV
- Rubeosis iridis: Iris NV at pupil margin, open angle
- Angle closure: Fibrovascular membrane, PAS, IOP >40 mmHg
- Management: Pan-retinal photocoagulation (PRP) or anti-VEGF injections to regress NV, then glaucoma surgery (tube shunt preferred)
-
Phacomorphic Glaucoma
- Mechanism: Intumescent cataract pushes iris forward, closes angle
- Presentation: Acute or subacute angle closure in patient with known cataract
- Diagnosis: Lens thickness >5 mm, shallow AC, closed angle
- Management: Medical IOP control → cataract extraction (definitive treatment)
-
Lens Particle Glaucoma
- Mechanism: Lens cortex material released after trauma/surgery clogs trabecular meshwork
- Presentation: IOP elevation days to weeks after cataract surgery or trauma
- Diagnosis: White fluffy particles in AC, open angle with trabecular debris
- Management: Steroids, IOP control, remove retained lens material if persistent
-
Malignant Glaucoma (Aqueous Misdirection)
- Mechanism: Aqueous flows posteriorly into vitreous, causing forward lens-iris diaphragm shift
- Presentation: Flat AC with elevated IOP after intraocular surgery (especially angle-closure surgery)
- Diagnosis: Uniformly shallow AC (central and peripheral), no pupillary block
- Management: Cycloplegics (atropine), aqueous suppressants, YAG laser hyaloidotomy, vitrectomy if medical fails
⭐ Clinical Pearl: Neovascular glaucoma is a clinical emergency requiring immediate PRP or anti-VEGF injection before addressing IOP surgically. Performing glaucoma surgery in an eye with active rubeosis has >80% failure rate due to continued fibrovascular proliferation. Treat the ischemia first (PRP within 24-48 hours), then consider tube shunt (not trabeculectomy-high failure rate) once NV regresses.
| Secondary Glaucoma | Mechanism | Key Finding | IOP Pattern | First-Line Treatment |
|---|---|---|---|---|
| Pseudoexfoliation | TM obstruction by matrix material | Target sign on lens, Sampaolesi line | Very high, fluctuating | Aggressive medical Rx, early surgery |
| Pigmentary | TM obstruction by iris pigment | Krukenberg spindle, transillumination defects | Spikes with exercise/dilation | Pilocarpine, LPI controversial |
| Steroid-induced | ↓ TM outflow facility | History of steroid use | Gradual rise over 2-6 weeks | Stop steroids, switch to weak steroid |
| Uveitic | Inflammation, debris, PAS | Cells/flare in AC, KP | Variable, often elevated | Treat inflammation first, then IOP |
| Neovascular | Fibrovascular membrane, PAS | Rubeosis iridis, retinal ischemia | Very high (>40 mmHg), refractory | PRP or anti-VEGF, then tube shunt |
| Phacomorphic | Lens pushes iris forward | Intumescent cataract, shallow AC | Acute elevation | Cataract extraction (definitive) |
🔬 Secondary Glaucomas: When Glaucoma Has a Cause
🎯 Medical Management Mastery: The Pharmacological Arsenal
Medical therapy aims to reduce IOP to a target pressure that prevents further optic nerve damage. Target IOP is individualized based on baseline pressure, disease severity, and progression risk-typically 25-30% reduction from baseline, with lower targets (<15 mmHg) for advanced disease.

Prostaglandin Analogs: First-Line Uveoscleral Champions
PGAs are the most effective single agents, reducing IOP 25-35% with once-daily dosing and minimal systemic side effects:
-
Mechanism: Bind FP prostanoid receptors → ↑ matrix metalloproteinases → remodel ciliary muscle and sclera → ↑ uveoscleral outflow by 50-100%
-
Agents and Efficacy
- Latanoprost 0.005%: Prototype, 28-31% IOP reduction
- Travoprost 0.004%: Similar efficacy, 27-30% reduction
- Bimatoprost 0.01-0.03%: Prostamide (not true PG), 28-33% reduction, slightly more potent
- Tafluprost 0.0015%: Preservative-free, 26-28% reduction
-
Advantages
- Once-daily dosing (evening optimal-maximizes daytime IOP reduction)
- No systemic contraindications (safe in asthma, COPD, heart disease)
- Additive with all other classes
-
Adverse Effects
- Periorbital changes: Increased iris pigmentation (10-15%, permanent), eyelash growth, periorbital fat atrophy (enophthalmos, deepening of lid sulcus)
- Conjunctival hyperemia (20-40%, usually mild)
- Cystoid macular edema (1-2% in pseudophakic/aphakic eyes, rare in phakic)
- Reactivation of herpes simplex keratitis (avoid in active HSV)
📌 PGA Mnemonic: "LTTB-Evening" - Latanoprost, Travoprost, Tafluprost, Bimatoprost, all dosed Evening (once daily at bedtime). They Lower IOP by Taking the Trabecular Bypass (uveoscleral route).
Beta-Blockers: Aqueous Production Suppressors
Beta-blockers reduce aqueous production 20-30% by blocking β2-adrenergic receptors on ciliary epithelium:
-
Agents
- Timolol 0.25-0.5%: Prototype non-selective β1/β2 blocker, 20-25% IOP reduction
- Betaxolol 0.5%: Selective β1 blocker, 15-20% reduction (less potent but safer in asthma)
- Levobunolol 0.25-0.5%, Carteolol 1%: Non-selective alternatives
-
Advantages
- Twice-daily dosing (or once-daily gel formulation)
- Inexpensive
- Well-established safety profile
-
Contraindications and Adverse Effects
- Systemic: Bradycardia, bronchospasm (absolute contraindication in severe asthma/COPD), hypotension, fatigue, depression, erectile dysfunction
- Ocular: Superficial punctate keratitis, decreased corneal sensitivity
- Tachyphylaxis: 10-20% lose efficacy over months (short-term escape)
⚠️ Warning: Beta-blockers are contraindicated in patients with asthma, COPD, bradycardia <50 bpm, second/third-degree AV block, decompensated heart failure. Even topical administration achieves systemic absorption (80% via nasolacrimal duct), so screen for pulmonary and cardiac disease before prescribing.
Alpha-2 Agonists: Dual-Action IOP Reducers
Alpha agonists reduce IOP 15-25% via two mechanisms:
-
Mechanism
- ↓ Aqueous production (30-50% reduction) via α2 receptors on ciliary epithelium
- ↑ Uveoscleral outflow (30-40% increase)
-
Agents
- Brimonidine 0.1-0.2%: Selective α2 agonist, 18-25% IOP reduction, 2-3× daily
- Apraclonidine 0.5-1%: Less selective, used for acute IOP spikes (post-laser), high tachyphylaxis (30% at 3 months)
-
Advantages
- Neuroprotective effects (independent of IOP reduction-controversial)
- Useful adjunct to PGAs or beta-blockers
-
Adverse Effects
- Ocular allergy (10-15% with brimonidine-redness, follicular conjunctivitis)
- CNS depression in children <6 years (contraindicated-apnea
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app







