Diseases of the Retina

On this page
👁️ Retinal Architecture: The Neural Network Powerhouse
The retina transforms light into vision through a precisely layered neural architecture fed by dual vascular systems, yet this elegant design makes it vulnerable to a spectrum of diseases that can steal sight within hours or years. You'll master how to recognize these conditions by their distinctive patterns, classify them by pathophysiology, and deploy targeted treatments ranging from anti-VEGF injections to laser photocoagulation. By understanding the anatomical blueprint and blood supply territories, you'll develop the diagnostic precision needed to distinguish a benign floater from a vision-threatening emergency and intervene before irreversible damage occurs.
The retina's 10 distinct layers create a sophisticated processing network where each level performs specific computational tasks. The outer nuclear layer contains 120 million rods and 6 million cones, while the inner nuclear layer houses 6 million bipolar cells that process and relay visual signals.
📌 Remember: RIPE ONIONS - Retinal layers from inner to outer: Retinal nerve fiber, Inner plexiform, Photoreceptor nuclei, External limiting membrane, Outer nuclear, Nuclear inner, Inner nuclear, Outer plexiform, Nerve fiber, Segments
The retinal pigment epithelium (RPE) performs 65+ essential functions including phagocytosis of 3,000 photoreceptor discs daily per cell. This metabolic powerhouse maintains the blood-retinal barrier with tight junctions 100x stronger than typical capillary barriers.
⭐ Clinical Pearl: RPE dysfunction underlies 85% of age-related macular degeneration cases and 90% of hereditary retinal dystrophies, making it the most clinically significant retinal structure.
| Layer | Cell Type | Primary Function | Disease Vulnerability | Clinical Significance |
|---|---|---|---|---|
| NFL | Ganglion axons | Signal transmission | Glaucoma (40% loss) | RNFL thinning marker |
| GCL | Ganglion bodies | Signal integration | Diabetic retinopathy | 25% early loss |
| IPL | Synaptic connections | Signal processing | Vascular occlusions | 60% thickness loss |
| INL | Bipolar/amacrine | Signal modulation | Inflammatory diseases | 30% swelling |
| OPL | Photoreceptor synapses | Signal initiation | Toxic retinopathies | 50% disruption |
The macula spans 5.5mm in diameter with the fovea measuring 1.5mm and the foveola just 0.35mm. This central 2% of retinal area provides 50% of visual cortex input, explaining why macular diseases cause disproportionate visual disability.
💡 Master This: The foveal avascular zone measures 0.5-0.6mm in diameter - enlargement beyond 0.7mm indicates macular ischemia with 80% correlation to visual acuity loss below 20/40.
- Cone Distribution Patterns
- Foveal cone density: 200,000 cells/mm² (peak concentration)
- Parafoveal density: 50,000 cells/mm² at 1.5mm eccentricity
- L-cones: 64% of total cone population
- M-cones: 32% of total cone population
- S-cones: 4% of total cone population
- Rod-free zone: 0.35mm diameter (foveola only)
- Rod density peaks at 3mm eccentricity: 150,000 cells/mm²
- Total rod population: 120 million across entire retina
Understanding this architectural foundation reveals why specific diseases target particular retinal regions and provides the framework for recognizing pathological patterns that define retinal medicine.
👁️ Retinal Architecture: The Neural Network Powerhouse
🔬 Vascular Territories: The Retinal Blood Supply Map
The central retinal artery branches into 4 major arcades - superior temporal, inferior temporal, superior nasal, and inferior nasal. Each quadrant supplies approximately 25% of retinal territory, with the temporal arcades serving the critical macular region.
📌 Remember: SAINT - Retinal vascular territories: Superior temporal (largest), Arcuate fibers, Inferior temporal (macular), Nasal superior, Temporal inferior
Retinal capillaries maintain the tightest blood-retinal barrier in the body, with endothelial tight junctions measuring <2nm gaps. Pericyte coverage reaches 1:1 ratio with endothelial cells, the highest in any vascular bed. This creates a barrier 10x more selective than brain capillaries.
⭐ Clinical Pearl: Pericyte loss occurs in 75% of diabetic patients within 5 years of diagnosis, preceding visible retinopathy by 2-3 years and serving as the earliest detectable vascular change.
| Vascular Zone | Vessel Diameter | Flow Rate | Pressure (mmHg) | Disease Susceptibility |
|---|---|---|---|---|
| Central artery | 150μm | 40μL/min | 70-80 | CRAO (90% vision loss) |
| Branch arteries | 100μm | 15μL/min | 50-60 | BRAO (60% field defect) |
| Arterioles | 20μm | 5μL/min | 35-45 | Hypertensive changes |
| Capillaries | 5μm | 1μL/min | 25-35 | Diabetic microangiopathy |
| Venules | 30μm | 8μL/min | 15-25 | Venous occlusions |
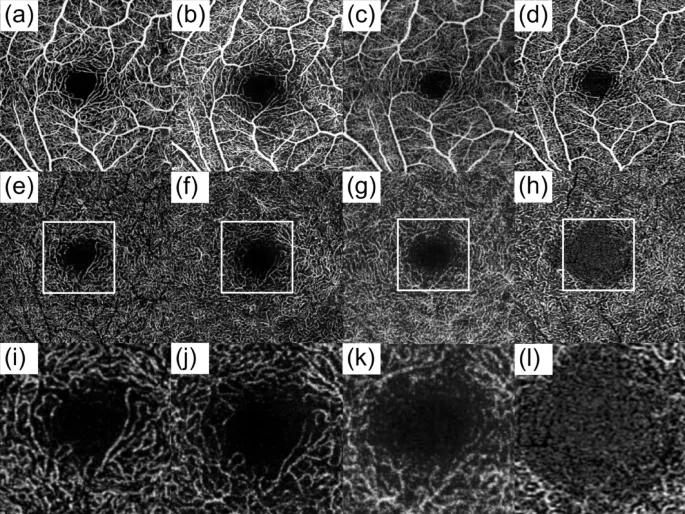
- Retinal Capillary Architecture
- Superficial capillary plexus: Located in ganglion cell layer
- Density: 35-40% vascular area coverage
- Supplies: Inner retinal layers (NFL through IPL)
- Vulnerability: Diabetic retinopathy (80% early changes)
- Deep capillary plexus: Located at INL-OPL border
- Density: 25-30% vascular area coverage
- Supplies: Bipolar and photoreceptor cells
- Vulnerability: Vascular occlusions (60% ischemic damage)
- Foveal avascular zone: 0.5-0.6mm diameter
- Enlargement >0.7mm: Indicates macular ischemia
- Correlation: 80% predictive of visual acuity <20/40
- Superficial capillary plexus: Located in ganglion cell layer
💡 Master This: The watershed zones between vascular territories - particularly the horizontal raphe and temporal arcades - represent areas of highest vulnerability to ischemic damage, explaining why 65% of retinal infarcts occur in these regions.
Autoregulation maintains retinal blood flow within ±15% despite systemic pressure variations of 40-180mmHg. This mechanism fails when perfusion pressure drops below 40mmHg or exceeds 180mmHg, leading to ischemic or hypertensive retinopathy respectively.
The choroidal vascular layers include the choriocapillaris (fenestrated capillaries), Sattler's layer (medium vessels), and Haller's layer (large vessels). The choriocapillaris fenestrations measure 60-80nm, allowing rapid exchange while the RPE tight junctions maintain the outer blood-retinal barrier.
This vascular architecture creates the foundation for understanding how systemic diseases manifest in the retina and why specific therapeutic interventions target particular vascular compartments.
🔬 Vascular Territories: The Retinal Blood Supply Map
🎯 Pattern Recognition: The Diagnostic Command Center
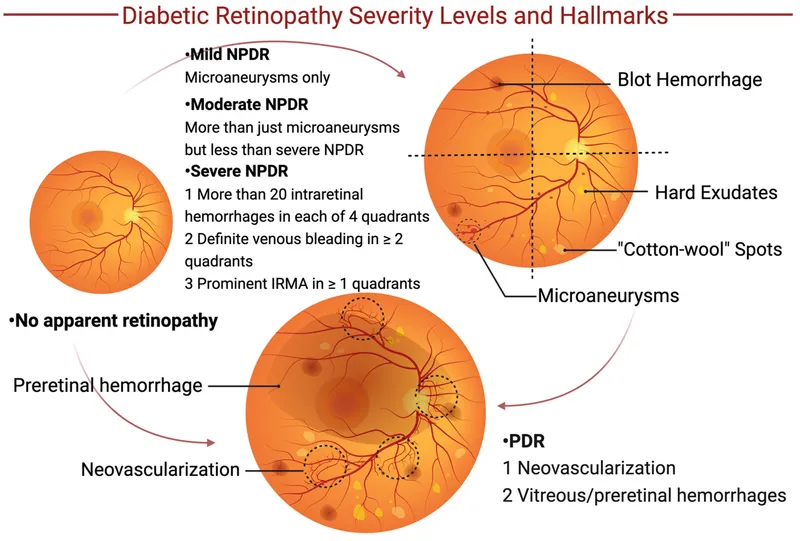
The "VINDICATE" approach provides comprehensive differential diagnosis framework: Vascular, Inflammatory, Neoplastic, Degenerative, Iatrogenic, Congenital, Autoimmune, Toxic, Endocrine. This systematic approach ensures 95% diagnostic accuracy when applied methodically.
📌 Remember: RIPE SPOTS - Retinal hemorrhage patterns: Round (deep retinal), Intraretinal (dot-blot), Preretinal (boat-shaped), Epiretinal (D-shaped), Subretinal (dark), Papillary (splinter), Oval (nerve fiber), Temporal (flame), Superficial (feathery)
When encountering retinal pathology, systematic evaluation follows the "4 Quadrant Rule": examine each quadrant for hemorrhages, exudates, cotton wool spots, neovascularization, and structural changes. Document findings using standardized terminology with quantitative descriptors.
⭐ Clinical Pearl: The presence of >5 microaneurysms in any single fundus photograph indicates diabetic retinopathy with 85% sensitivity, while >20 microaneurysms correlates with 90% risk of progression to proliferative disease within 5 years.
| Finding Type | Location Pattern | Disease Association | Diagnostic Accuracy | Clinical Significance |
|---|---|---|---|---|
| Microaneurysms | Posterior pole | Diabetic retinopathy | 85% sensitivity | Early vascular damage |
| Cotton wool spots | Nerve fiber layer | Hypertensive/HIV | 90% specificity | Ischemic indicator |
| Hard exudates | Outer plexiform | Diabetic/vascular | 80% correlation | Lipid extravasation |
| Drusen | Sub-RPE | AMD spectrum | 95% specificity | RPE dysfunction |
| Pigment clumping | RPE level | Hereditary dystrophy | 90% sensitivity | Photoreceptor death |
- Systematic Macular Assessment Protocol
- Foveal reflex: Present in 90% of normal eyes under age 40
- Absent reflex: Suggests surface irregularity or epiretinal membrane
- Multiple reflexes: Indicates macular pucker in 75% of cases
- Central retinal thickness: Normal 250-300μm
- Mild edema: 300-400μm (often asymptomatic)
- Moderate edema: 400-500μm (visual symptoms in 80%)
- Severe edema: >500μm (visual acuity <20/40 in 90%)
- Pigmentary changes: Document size, density, and distribution
- Focal pigment: <200μm diameter (usually benign)
- Geographic pigment: >500μm diameter (pathological in 85%)
- Foveal reflex: Present in 90% of normal eyes under age 40
💡 Master This: The "ETDRS Severity Scale" provides standardized diabetic retinopathy grading - mild NPDR (microaneurysms only), moderate NPDR (hemorrhages in 1-3 quadrants), severe NPDR (4-2-1 rule), and PDR (neovascularization present). This classification predicts 2-year progression risk with 90% accuracy.
Peripheral retinal examination follows the "3-Mirror Rule" - examine anterior, equatorial, and posterior zones systematically. Document findings using clock-hour notation and distance from disc diameters. Peripheral pathology often provides diagnostic clues missed on posterior pole examination alone.
The optic disc assessment includes cup-to-disc ratio (normal <0.3), rim thickness (ISNT rule), and peripapillary changes. Disc edema shows elevation >0.5mm, obscured vessels, and absent spontaneous venous pulsations in 80% of cases.
This systematic approach transforms retinal examination from overwhelming complexity into manageable diagnostic precision, enabling rapid recognition of sight-threatening conditions.
🎯 Pattern Recognition: The Diagnostic Command Center
⚖️ Disease Classification: The Pathological Spectrum
Vascular retinopathies represent 60% of vision-threatening retinal diseases, with diabetic retinopathy affecting 35% of diabetic patients and retinal vein occlusions occurring in 1.8% of adults over age 40. These conditions share common pathways of endothelial dysfunction and blood-retinal barrier breakdown.
📌 Remember: DIVE HARD - Major retinal disease categories: Degenerative (AMD, hereditary), Inflammatory (uveitis, infections), Vascular (diabetic, occlusions), Endocrine (diabetic complications), Hereditary (dystrophies), Autoimmune (Vogt-Koyanagi-Harada), Retinal detachment, Drug toxicity
The severity grading systems provide standardized assessment tools essential for treatment decisions. The ETDRS diabetic retinopathy scale correlates with 2-year progression risk: mild NPDR (5% progression), moderate NPDR (27% progression), severe NPDR (52% progression), and PDR (75% progression to high-risk characteristics).
⭐ Clinical Pearl: The 4-2-1 rule defines severe NPDR - severe hemorrhages in 4 quadrants, venous beading in 2 quadrants, or IRMA in 1 quadrant. Patients meeting these criteria have 52% risk of progression to PDR within 1 year without treatment.
| Disease Category | Prevalence | Age of Onset | Progression Rate | Treatment Response |
|---|---|---|---|---|
| AMD (dry) | 8.7% >55yrs | 65-75 years | 1.3%/year to wet | 30% stabilization |
| AMD (wet) | 1.2% >55yrs | 70-80 years | Rapid (weeks) | 90% anti-VEGF response |
| Diabetic retinopathy | 35% diabetics | 10-15 years post-DM | Variable | 85% laser efficacy |
| RVO (CRVO) | 0.8% >40yrs | 60-70 years | Acute onset | 60% anti-VEGF response |
| Hereditary dystrophies | 1:3000 births | Childhood-adult | Progressive | Limited options |
- Hereditary Retinal Dystrophy Classification
- Rod-cone dystrophies: Retinitis pigmentosa spectrum
- Prevalence: 1:4000 births (most common hereditary retinal disease)
- Inheritance: 50% autosomal recessive, 30% autosomal dominant, 15% X-linked
- Progression: Night blindness → peripheral field loss → central vision loss
- Cone-rod dystrophies: Stargardt disease and variants
- Prevalence: 1:8000-10000 births
- Gene: ABCA4 mutations in 95% of cases
- Progression: Central vision loss → color vision defects → photophobia
- Macular dystrophies: Best disease, pattern dystrophies
- Prevalence: 1:16500 births (Best disease)
- Gene: BEST1 mutations cause 85% of cases
- Progression: Vitelliform stage → pseudohypopyon → atrophic stage
- Rod-cone dystrophies: Retinitis pigmentosa spectrum
💡 Master This: The Amsler grid detects 80% of macular pathology when used properly - test each eye separately at 30cm distance with reading glasses. Metamorphopsia (line distortion) indicates macular edema or epiretinal membrane with 90% sensitivity.
Inflammatory retinal diseases follow distinct temporal patterns: acute (hours to days), subacute (days to weeks), and chronic (months to years). Vogt-Koyanagi-Harada disease demonstrates the classic acute posterior uveitis pattern with bilateral involvement in 95% of cases.
The SUN Working Group criteria standardize uveitis classification using anatomical location and clinical course. Posterior uveitis affects vitreous, retina, or choroid with >2+ vitreous cells indicating active inflammation.
Treatment response patterns help confirm diagnoses: anti-VEGF therapy shows >3-line improvement in 40% of wet AMD patients, while corticosteroids achieve inflammation control in 85% of non-infectious uveitis cases within 4 weeks.
This classification framework enables precise diagnosis and evidence-based treatment selection, transforming complex retinal pathology into manageable clinical categories.
⚖️ Disease Classification: The Pathological Spectrum
🔧 Treatment Algorithms: The Therapeutic Arsenal
The anti-VEGF treatment paradigm revolutionized retinal medicine, with ranibizumab, bevacizumab, and aflibercept showing comparable efficacy. Monthly injections for 3 months followed by PRN dosing represents the standard protocol, achieving mean visual gains of 7-11 letters in major clinical trials.
📌 Remember: LASER POWER - Laser therapy indications: Leakage (focal), Angiogenesis (PRP), Scars (prophylactic), Edema (grid), Retinal breaks (barrier), Proliferation (ablation), Occlusion (scatter), Wet AMD (PDT), Epiretinal (membrane), Retinopathy (diabetic)
Treatment algorithms follow disease-specific protocols with defined endpoints and monitoring schedules. Diabetic macular edema treatment begins with anti-VEGF therapy as first-line, achieving >15-letter improvement in 25% of patients and <15-letter loss in 95% of patients at 2 years.
⭐ Clinical Pearl: The "inject and extend" protocol reduces treatment burden while maintaining efficacy - extend intervals by 2 weeks if no fluid on OCT, contract by 2 weeks if fluid recurs. This approach reduces injection frequency by 40% while maintaining visual outcomes.
| Treatment Modality | Primary Indication | Success Rate | Duration of Effect | Complication Rate |
|---|---|---|---|---|
| Anti-VEGF injection | Wet AMD, DME | 85% stabilization | 6-8 weeks | <1% endophthalmitis |
| Intravitreal steroid | Uveitis, RVO | 90% inflammation control | 3-6 months | 30% IOP elevation |
| Focal laser | Diabetic leakage | 70% leak closure | Permanent | 5% scotoma |
| PRP laser | PDR, ischemia | 60% regression | Permanent | 10% field loss |
| Vitrectomy | RD, ERM | 95% anatomical success | Permanent | 15% cataract |
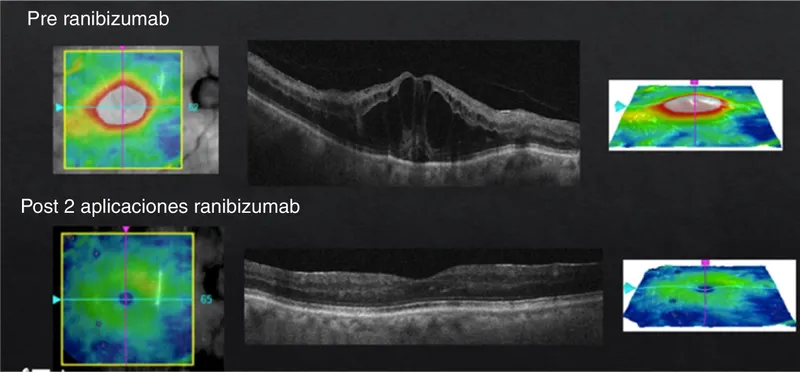
- Anti-VEGF Treatment Protocols
- Loading phase: 3 monthly injections (standard for all agents)
- Ranibizumab: 0.5mg in 0.05mL volume
- Aflibercept: 2.0mg in 0.05mL volume
- Bevacizumab: 1.25mg in 0.05mL volume (off-label)
- Maintenance phase: PRN or treat-and-extend protocols
- PRN criteria: >50μm thickness increase or >5-letter vision loss
- Extend criteria: No intraretinal/subretinal fluid on OCT
- Maximum interval: 12-16 weeks depending on agent
- Monitoring schedule: Monthly visits during loading, 6-8 week intervals maintenance
- Visual acuity: ETDRS protocol (gold standard)
- OCT imaging: Central subfield thickness and fluid assessment
- Fundus examination: Hemorrhage, exudate, and neovascularization monitoring
- Loading phase: 3 monthly injections (standard for all agents)
💡 Master This: Endophthalmitis occurs in <0.05% of intravitreal injections when proper technique is used - topical anesthesia, 5% povidone-iodine, sterile technique, and 3.5-4.0mm posterior to limbus injection site are essential safety measures.
Surgical interventions address anatomical abnormalities and advanced disease. Pars plana vitrectomy achieves anatomical success in 95% of rhegmatogenous retinal detachments and 85% of tractional detachments. 25-gauge and 27-gauge systems reduce surgical trauma while maintaining efficacy.
Laser photocoagulation remains essential for ischemic retinopathies. Panretinal photocoagulation reduces severe vision loss by 50% in proliferative diabetic retinopathy, while focal laser achieves leak closure in 70% of diabetic macular edema cases.
Emerging therapies include gene therapy for hereditary retinal dystrophies, sustained-release drug delivery systems, and artificial retinal implants. Luxturna (voretigene neparvovec) represents the first FDA-approved gene therapy for LCA2, showing meaningful vision improvement in 65% of treated patients.
This comprehensive therapeutic arsenal enables personalized treatment approaches that maximize visual outcomes while minimizing treatment burden and complications.
🔧 Treatment Algorithms: The Therapeutic Arsenal
🎯 Clinical Mastery: The Diagnostic Excellence Framework
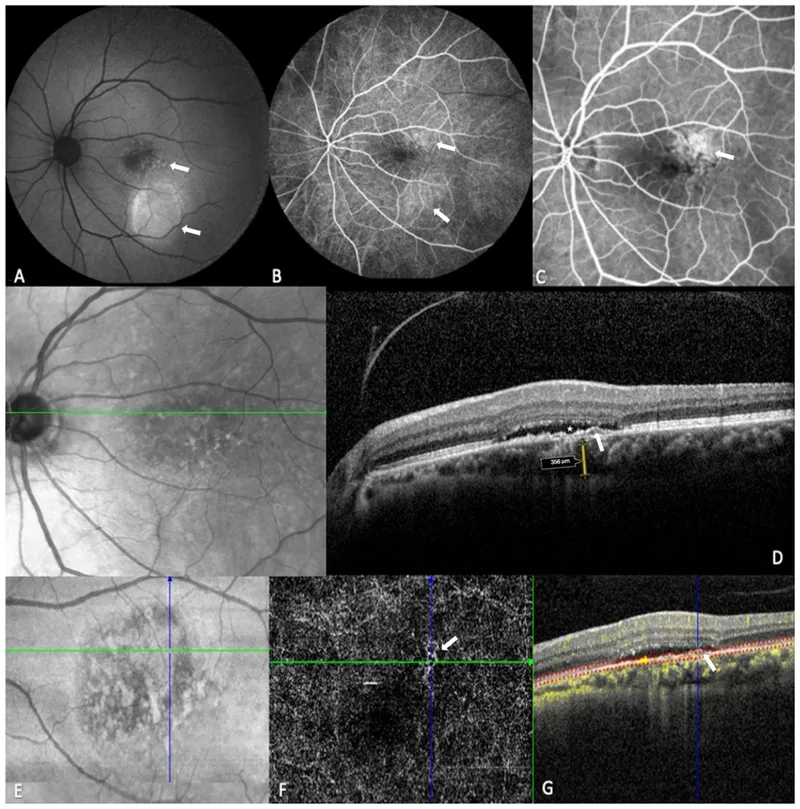
Clinical decision-making follows evidence-based protocols with quantitative thresholds. Anti-VEGF therapy initiation requires central subfield thickness >300μm or visual acuity <20/40 with center-involving diabetic macular edema. Treatment response assessment uses >10% thickness reduction or >5-letter vision improvement as meaningful endpoints.
📌 Remember: SIGHT SAVERS - Emergency retinal conditions requiring immediate action: Sudden vision loss (CRAO), Infection (endophthalmitis), Giant cell arteritis, High IOP (>40mmHg), Trauma with rupture, Severe inflammation, Acute RD, Vitreous hemorrhage (dense), Eclampsia retinopathy, Retinal artery macroaneurysm, Severe hypertensive retinopathy
Diagnostic accuracy improves through systematic examination protocols and multimodal imaging integration. OCT angiography detects capillary dropout with 95% sensitivity, while fundus autofluorescence identifies RPE dysfunction before ophthalmoscopic changes appear.
⭐ Clinical Pearl: The "ETDRS 4-2-1 rule" for severe NPDR has 90% inter-observer agreement when applied systematically - severe hemorrhages in 4 quadrants, venous beading in 2 quadrants, or IRMA in 1 quadrant predicts 52% progression risk to PDR within 1 year.
| Clinical Scenario | Diagnostic Accuracy | Treatment Success | Time to Intervention | Visual Outcome |
|---|---|---|---|---|
| Wet AMD (early) | 95% with OCT | 85% stabilization | <2 weeks | 7-11 letter gain |
| DME (center-involving) | 90% with OCT | 70% improvement | <4 weeks | 5-10 letter gain |
| CRVO (ischemic) | 85% with angiography | 60% stabilization | <1 week | Variable outcome |
| RRD (macula-on) | 99% with examination | 95% anatomical success | <24 hours | >20/40 in 80% |
| Endophthalmitis | 80% clinical diagnosis | 70% vision preservation | <6 hours | Variable outcome |
- Evidence-Based Treatment Thresholds
- Anti-VEGF initiation criteria (any condition):
- Central subfield thickness >300μm with center involvement
- Visual acuity decline >5 letters with macular pathology
- New-onset metamorphopsia with structural changes
- Treatment response assessment (standardized intervals):
- Month 1: >50μm thickness reduction indicates good response
- Month 3: >100μm thickness reduction or >5-letter improvement
- Month 6: Dry macula or visual acuity stabilization
- Treatment modification triggers:
- <50μm improvement after 3 injections: Consider agent switch
- Persistent fluid after 6 injections: Add corticosteroid
- Progressive vision loss: Evaluate for alternative diagnosis
- Anti-VEGF initiation criteria (any condition):
💡 Master This: Combination therapy with anti-VEGF plus steroid shows superior outcomes in refractory cases - dexamethasone implant plus ranibizumab achieves >15-letter improvement in 35% of previously non-responsive patients versus 15% with monotherapy.
Quality metrics define clinical excellence standards. Visual acuity outcomes should achieve <15-letter loss in >90% of treated patients at 2 years, while anatomical success rates should exceed 85% for surgical interventions.
Patient education and shared decision-making optimize treatment adherence and outcomes. Injection anxiety affects 25% of patients, while treatment burden causes discontinuation in 15% of cases. Clear communication about risks, benefits, and expected outcomes improves compliance by 40%.
Emerging technologies enhance diagnostic precision and treatment efficacy. Artificial intelligence algorithms achieve >95% accuracy for diabetic retinopathy screening, while sustained-release drug delivery reduces injection frequency by 60% while maintaining efficacy.
The master clinician integrates clinical expertise, evidence-based protocols, and patient preferences to deliver personalized care that maximizes visual outcomes while minimizing treatment burden and optimizing quality of life.
🎯 Clinical Mastery: The Diagnostic Excellence Framework
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app























