Menstrual Disorders

On this page
🩸 The Menstrual Maze: Decoding Cycle Chaos
Menstrual disorders affect nearly every woman you'll treat, yet their presentations range from subtle cycle shifts to life-altering hemorrhage. You'll master the hormonal choreography that orchestrates normal cycles, then learn to recognize when that rhythm breaks-distinguishing benign variations from pathology demanding intervention. Through pattern recognition, targeted diagnostics, and evidence-based therapeutics, you'll transform from observer to expert clinician who restores both physiologic balance and quality of life.
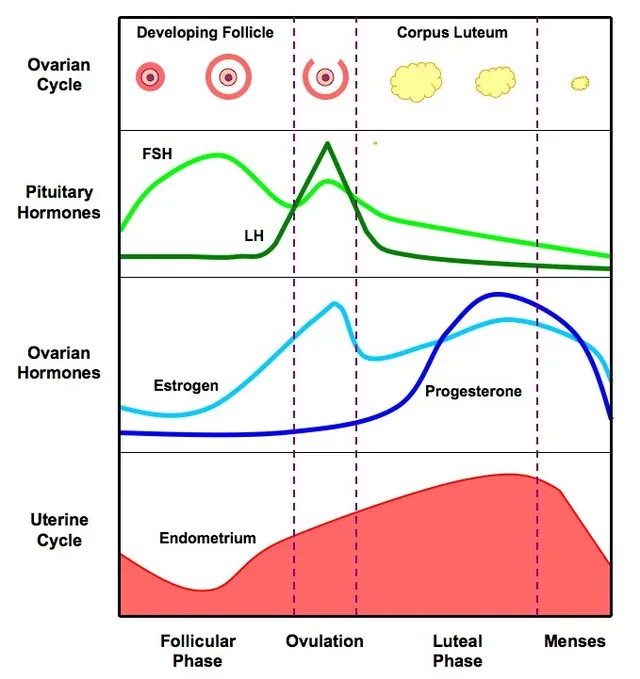
The menstrual cycle represents one of medicine's most elegant feedback systems. Normal cycle length ranges 21-35 days, with flow duration of 3-7 days and blood loss typically 5-40ml per cycle. These parameters serve as critical benchmarks for identifying pathology.
📌 Remember: FLOW mnemonic for normal menstrual parameters:
- Frequency: 21-35 days
- Length: 3-7 days
- Output: 5-40ml total
- Weight: <80ml considered normal
Menstrual Disorder Classification Framework
- Abnormal Uterine Bleeding (AUB)
- Heavy menstrual bleeding: >80ml per cycle or >7 days duration
- Intermenstrual bleeding: bleeding between regular cycles
- Spotting: <5ml light bleeding
- Breakthrough bleeding: >5ml significant bleeding
- Amenorrhea Spectrum
- Primary: no menarche by age 15 with normal development
- Secondary: absence of menses >3 months in previously cycling women
- Oligomenorrhea: cycles >35 days apart
- Polymenorrhea: cycles <21 days apart
| Parameter | Normal Range | Oligomenorrhea | Polymenorrhea | Amenorrhea | Clinical Significance |
|---|---|---|---|---|---|
| Cycle Length | 21-35 days | >35 days | <21 days | >90 days | Ovulation assessment |
| Flow Duration | 3-7 days | Variable | Variable | 0 days | Endometrial response |
| Blood Loss | 5-40ml | Variable | Variable | 0ml | Hemoglobin impact |
| Ovulation | Day 14±2 | Delayed/absent | Premature | Absent | Fertility implications |
| Luteal Phase | 12-14 days | Shortened | Shortened | Absent | Progesterone adequacy |

Pathophysiology Patterns
The hypothalamic-pituitary-ovarian (HPO) axis controls menstrual function through precise hormonal cascades. GnRH pulses every 90-120 minutes stimulate FSH/LH release, triggering folliculogenesis and steroidogenesis.
-
Structural Abnormalities (PALM)
- Polyps: 10-15% of AUB cases, malignancy risk 2-5%
- Adenomyosis: affects 20-30% of reproductive-age women
- Leiomyomas: present in 70% of women by age 50
- Malignancy: <1% in reproductive age, >10% postmenopausal
-
Non-Structural Causes (COEIN)
- Coagulopathy: von Willebrand disease in 13% of heavy bleeding
- Ovulatory dysfunction: 90% of adolescent AUB
- Endometrial: local hemostatic disorders
- Iatrogenic: breakthrough bleeding in 30% of hormonal users
- Not classified: idiopathic causes
💡 Master This: Anovulatory bleeding accounts for 90% of adolescent menstrual disorders due to immature HPO axis. Ovulatory cycles typically establish within 2 years of menarche, with luteal phase defects common initially.
⭐ Clinical Pearl: Thyroid dysfunction affects 15-20% of women with menstrual disorders. TSH >4.5 mIU/L can cause anovulation, while hyperthyroidism typically causes oligomenorrhea in 65% of cases.
Understanding these foundational patterns enables systematic approach to any menstrual complaint. The next section explores how hormonal dysregulation creates the specific syndrome presentations that define clinical practice.
🩸 The Menstrual Maze: Decoding Cycle Chaos
⚖️ Hormonal Havoc: The Endocrine Earthquake
PCOS: The Metabolic Menstrual Storm
PCOS represents the most common endocrine disorder in reproductive-age women, affecting 1 in 10 globally. The Rotterdam Criteria requires 2 of 3 features: oligoovulation, hyperandrogenism, and polycystic ovarian morphology.
📌 Remember: PCOS diagnostic criteria - HAIR mnemonic:
- Hyperandrogenism (clinical or biochemical)
- Anovulation/oligoovulation
- Imaging showing polycystic ovaries (≥12 follicles 2-9mm)
- Rule out other causes (thyroid, prolactin, CAH)
- Hormonal Dysregulation Cascade
- Insulin resistance: present in 65-70% of PCOS patients
- Hyperinsulinemia: stimulates ovarian androgen production
- Testosterone levels: typically >70ng/dl (normal <50ng/dl)
- Free androgen index: elevated >5 (normal <4.5)
- LH:FSH ratio: classically >2:1 but present in only 60%
- LH hypersecretion: disrupts follicular development
- Chronic anovulation: progesterone deficiency
| PCOS Phenotype | Hyperandrogenism | Ovulatory Dysfunction | PCO Morphology | Prevalence | Metabolic Risk |
|---|---|---|---|---|---|
| Classic | Present | Present | Present | 60-65% | Highest |
| Non-PCO | Present | Present | Absent | 15-20% | High |
| Ovulatory | Present | Absent | Present | 10-15% | Moderate |
| Normoandrogenic | Absent | Present | Present | 10-15% | Variable |
Thyroid-Reproductive Axis Disruption
Thyroid disorders create profound menstrual irregularities through multiple mechanisms. Hypothyroidism affects 2-4% of reproductive-age women, while hyperthyroidism occurs in 0.5-2%.
-
Hypothyroidism Effects
- Menorrhagia: occurs in 40-50% of hypothyroid women
- TRH elevation: increases prolactin secretion
- Prolactin levels: can reach >100ng/ml (normal <25ng/ml)
- Galactorrhea: present in 25% of cases
- Anovulation: due to disrupted GnRH pulsatility
- TSH >10 mIU/L: associated with infertility in 76%
-
Hyperthyroidism Manifestations
- Oligomenorrhea: most common pattern (65% of cases)
- Amenorrhea: occurs in 20-25% of severe cases
- SHBG elevation: reduces free testosterone availability
- SHBG levels: increase 2-3 fold above normal
- Free androgen index: typically <2 (normal 2-4.5)
💡 Master This: Subclinical hypothyroidism (TSH 4.5-10 mIU/L with normal T4) causes luteal phase defects in 64% of women, leading to irregular cycles and infertility. Treatment with levothyroxine normalizes cycles in 80% within 3 months.
Prolactin Disorders: The Lactation Disruption
Hyperprolactinemia affects 5-10% of women with menstrual disorders. Prolactin levels >25ng/ml suppress GnRH pulsatility, causing hypogonadotropic hypogonadism.
- Prolactinoma Classification
- Microprolactinomas: <10mm, prolactin 25-200ng/ml
- Macroprolactinomas: >10mm, prolactin >200ng/ml
- Visual field defects: occur when >20mm
- Headaches: present in 70% of macroadenomas
⭐ Clinical Pearl: Dopamine agonists (cabergoline, bromocriptine) normalize prolactin levels in 85-90% of patients and restore ovulation in 80-85%. Cabergoline is preferred due to twice-weekly dosing and lower side effect profile.
The intricate hormonal networks controlling menstruation create vulnerability to multiple disruptions. Understanding these mechanisms guides targeted therapeutic interventions that restore both hormonal balance and reproductive function.
⚖️ Hormonal Havoc: The Endocrine Earthquake
🔍 Pattern Recognition: The Clinical Detective's Toolkit
Age-Stratified Pattern Recognition
Age serves as the most powerful discriminator in menstrual disorders, with distinct pathophysiology patterns emerging across reproductive life stages.
📌 Remember: AGES pattern recognition framework:
- Adolescent (menarche-19): Anovulation dominates (90% of AUB)
- Growing (20-39): Structural lesions increase (fibroids 20%, polyps 10%)
- Established (40-45): Adenomyosis peaks (30% prevalence)
- Senescent (>45): Malignancy risk rises (endometrial cancer 5-10%)
-
Adolescent Patterns (Menarche-19 years)
- Anovulatory bleeding: 90% of presentations
- Coagulopathy screening: indicated if heavy bleeding since menarche
- von Willebrand disease: 13% of adolescent menorrhagia
- Platelet disorders: 5-7% of cases
- Normal cycle establishment: within 2 years of menarche
- Cycle length: may vary 21-45 days initially
- Anovulation: normal for first 12-18 months
-
Reproductive Age Patterns (20-39 years)
- Structural causes: increase with age
- Uterine fibroids: 20% at age 20, 40% at age 35
- Endometrial polyps: 10-15% prevalence
- Pregnancy-related: always exclude first
- Ectopic pregnancy: 2% of all pregnancies
- Miscarriage: 15-20% of recognized pregnancies
- Structural causes: increase with age
| Age Group | Primary Causes | Key Features | Investigation Priority | Success Rate |
|---|---|---|---|---|
| 13-19 years | Anovulation (90%) | Irregular, heavy | CBC, coags if severe | 95% normalize |
| 20-29 years | Pregnancy, PCOS | Regular/irregular | βhCG, TSH, prolactin | 85% treatable |
| 30-39 years | Fibroids, polyps | Heavy, regular | Imaging + βhCG | 80% structural |
| 40-45 years | Adenomyosis | Painful, heavy | MRI, endometrial biopsy | 75% identifiable |
| >45 years | Perimenopause, cancer | Irregular timing | Endometrial sampling | 70% organic |
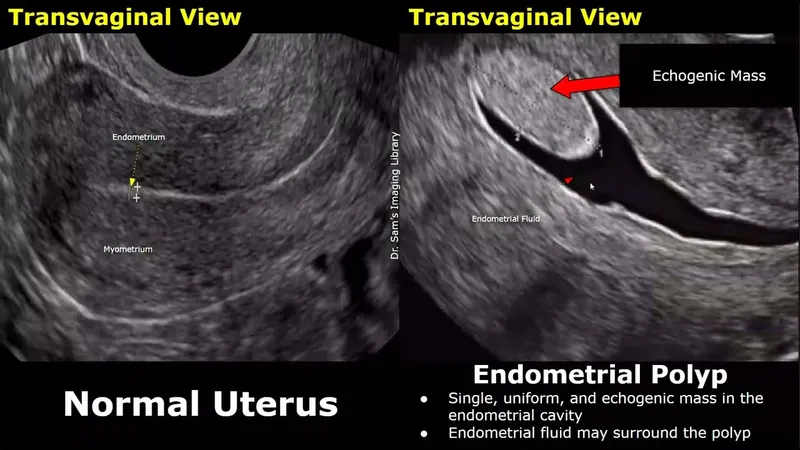
Bleeding Pattern Discrimination
Bleeding characteristics provide crucial diagnostic clues, with specific patterns correlating strongly with underlying pathophysiology.
- Ovulatory vs Anovulatory Bleeding
- Ovulatory bleeding: predictable timing, consistent flow
- Cycle length: 21-35 days consistently
- Premenstrual symptoms: breast tenderness, mood changes
- Mittelschmerz: mid-cycle pain in 40%
- Anovulatory bleeding: unpredictable, variable flow
- Cycle length: >35 days or <21 days
- No premenstrual symptoms: absence of progesterone effects
- Breakthrough bleeding: estrogen withdrawal patterns
- Ovulatory bleeding: predictable timing, consistent flow
💡 Master This: Intermenstrual bleeding in reproductive-age women requires cervical cytology and STI screening first. If normal, structural evaluation with transvaginal ultrasound identifies polyps in 60% and fibroids in 30% of cases.
Associated Symptom Patterns
Symptom clusters provide powerful diagnostic discrimination, with specific combinations pointing toward targeted evaluations.
-
Pain-Associated Bleeding
- Dysmenorrhea + heavy bleeding: adenomyosis (30% of women >35)
- Pelvic pain + irregular bleeding: endometriosis (10% reproductive age)
- Acute pain + bleeding: ovarian cyst rupture or ectopic pregnancy
- Ectopic pregnancy: βhCG typically 1500-3000 mIU/ml
- Corpus luteum cyst: progesterone >3ng/ml confirms ovulation
-
Systemic Symptom Clusters
- Weight gain + hirsutism + oligomenorrhea: PCOS (6-12% prevalence)
- Galactorrhea + amenorrhea: hyperprolactinemia (5-10% of disorders)
- Heat intolerance + weight loss + oligomenorrhea: hyperthyroidism
- Free T4 >1.7ng/dl with TSH <0.1 mIU/L
- Cold intolerance + weight gain + menorrhagia: hypothyroidism
- TSH >4.5 mIU/L affects 15-20% with menstrual disorders
⭐ Clinical Pearl: Galactorrhea occurs in 25% of hyperprolactinemia cases but can be physiologic in 5-10% of reproductive-age women. Prolactin >100ng/ml suggests pituitary adenoma requiring MRI evaluation.
These pattern recognition frameworks transform complex menstrual presentations into systematic diagnostic pathways, enabling rapid identification of underlying pathophysiology and targeted therapeutic interventions.
🔍 Pattern Recognition: The Clinical Detective's Toolkit
🎯 Diagnostic Precision: The Investigation Arsenal
First-Line Investigation Strategy
Systematic evaluation begins with high-yield screening tests that identify common pathology while ruling out emergencies. Pregnancy testing remains mandatory in all reproductive-age women with bleeding irregularities.
📌 Remember: FIRST investigation protocol for abnormal bleeding:
- Full blood count (hemoglobin, platelets)
- Iron studies (ferritin, TIBC)
- Reproductive hormones (βhCG, TSH, prolactin)
- Structural imaging (transvaginal ultrasound)
- Tissue sampling (if indicated by age/risk factors)
-
Essential Laboratory Panel
- Complete Blood Count: assess anemia and thrombocytopenia
- Hemoglobin <12g/dl: indicates significant blood loss
- Platelet count <100,000: suggests bleeding disorder
- MCV >100fl: may indicate B12/folate deficiency
- Iron Studies: quantify iron deficiency
- Ferritin <15ng/ml: confirms iron deficiency
- TIBC >450μg/dl: suggests iron depletion
- Transferrin saturation <16%: indicates functional deficiency
- Complete Blood Count: assess anemia and thrombocytopenia
-
Hormonal Assessment
- βhCG: mandatory in reproductive age
- Sensitivity >99% for pregnancy detection
- Quantitative levels guide ectopic pregnancy evaluation
- TSH: screens thyroid dysfunction
- Normal range: 0.4-4.0 mIU/L
- >4.5 mIU/L: investigate hypothyroidism
- Prolactin: if galactorrhea or amenorrhea
- Normal: <25ng/ml in reproductive age
- >100ng/ml: suggests pituitary adenoma
- βhCG: mandatory in reproductive age
| Investigation | Indication | Normal Values | Abnormal Threshold | Clinical Action |
|---|---|---|---|---|
| Hemoglobin | All patients | 12-15.5 g/dl | <10 g/dl | Iron supplementation |
| Ferritin | Heavy bleeding | 15-150 ng/ml | <15 ng/ml | Oral iron therapy |
| βhCG | Reproductive age | <5 mIU/ml | Any elevation | Pregnancy evaluation |
| TSH | Irregular cycles | 0.4-4.0 mIU/L | >4.5 mIU/L | Thyroid function tests |
| Prolactin | Galactorrhea/amenorrhea | <25 ng/ml | >100 ng/ml | MRI pituitary |
Advanced Imaging Protocols
Transvaginal ultrasound serves as the primary imaging modality for structural evaluation, providing real-time assessment of uterine architecture and ovarian morphology with 85-90% sensitivity for detecting abnormalities.
-
Ultrasound Assessment Parameters
- Endometrial thickness: varies with cycle phase
- Proliferative phase: 4-8mm normal
- Secretory phase: 8-16mm normal
- Postmenopausal: <4mm normal, >4mm requires biopsy
- Uterine measurements: assess size and contour
- Normal length: 6-10cm
- Normal width: 3-5cm
- Fibroid detection: sensitivity 95% for >2cm lesions
- Endometrial thickness: varies with cycle phase
-
Specialized Imaging Indications
- Saline infusion sonohysterography (SIS)
- Polyp detection: sensitivity 95%, specificity 88%
- Submucosal fibroid mapping: accuracy >90%
- Endometrial cavity assessment: superior to standard ultrasound
- MRI pelvis: gold standard for adenomyosis
- Junctional zone >12mm: diagnostic for adenomyosis
- T2 hyperintensity: indicates endometrial glands in myometrium
- Sensitivity 89%, specificity 96% for adenomyosis diagnosis
- Saline infusion sonohysterography (SIS)
💡 Master This: 3D ultrasound improves fibroid mapping accuracy to >95% and enables precise volume calculations for treatment planning. Doppler studies help differentiate vascular malformations from structural lesions.
Tissue Sampling Strategies
Endometrial sampling provides definitive histologic diagnosis when imaging suggests endometrial pathology or clinical risk factors warrant tissue evaluation.
-
Endometrial Biopsy Indications
- Age >45 years with new-onset bleeding
- Endometrial thickness >4mm postmenopausal
- Failed medical management of heavy bleeding
- Risk factors: obesity, diabetes, PCOS, tamoxifen use
- BMI >30: 3-fold increased endometrial cancer risk
- Diabetes: 2.8-fold increased risk
- PCOS: 2.7-fold increased risk due to unopposed estrogen
-
Sampling Techniques
- Office endometrial biopsy: Pipelle or Novak curette
- Sensitivity 81-99% for endometrial cancer
- Specificity 98% for malignancy detection
- Inadequate sample rate: 8-54% depending on technique
- Hysteroscopy with directed biopsy
- Gold standard for focal lesions
- Sensitivity >95% for polyps and submucosal fibroids
- Complete visualization of endometrial cavity
- Office endometrial biopsy: Pipelle or Novak curette
⭐ Clinical Pearl: Insufficient tissue on office biopsy in postmenopausal bleeding requires hysteroscopy or D&C due to 5-10% malignancy risk. Cervical stenosis increases inadequate sampling to >50% with office procedures.
This comprehensive diagnostic approach ensures systematic evaluation while minimizing patient discomfort and healthcare costs. Evidence-based protocols guide appropriate test selection and interpretation, leading to accurate diagnoses and optimal treatment outcomes.
🎯 Diagnostic Precision: The Investigation Arsenal
🛠️ Treatment Mastery: The Therapeutic Command Center
Medical Management Algorithms
First-line medical therapy targets underlying pathophysiology while providing symptom relief. Treatment selection depends on patient age, fertility desires, contraceptive needs, and bleeding patterns.
📌 Remember: MEDS approach to medical management:
- Menstrual suppression (hormonal contraceptives)
- Endometrial stabilization (progestins, antifibrinolytics)
- Disorder-specific therapy (thyroid, prolactin, PCOS)
- Supportive care (iron, NSAIDs, lifestyle)
-
Hormonal Therapies
- Combined Oral Contraceptives (COCs)
- Mechanism: endometrial atrophy, ovulation suppression
- Efficacy: 40-50% reduction in menstrual blood loss
- Success rate: 80-85% patient satisfaction
- Contraindications: age >35 + smoking, thromboembolism history
- Progestin-Only Options
- Levonorgestrel IUD: 90% reduction in bleeding by 12 months
- Depot medroxyprogesterone: amenorrhea in 50% by 1 year
- Oral progestins: norethindrone 5-10mg daily for cycle regulation
- Combined Oral Contraceptives (COCs)
-
Non-Hormonal Therapies
- Tranexamic acid: antifibrinolytic therapy
- Mechanism: plasmin inhibition, clot stabilization
- Efficacy: 40-50% reduction in blood loss
- Dosing: 1.3g TID during menses only
- Contraindications: thromboembolism risk, renal impairment
- NSAIDs: prostaglandin synthesis inhibition
- Mefenamic acid: 500mg TID during menses
- Efficacy: 20-30% reduction in bleeding
- Additional benefit: dysmenorrhea relief
- Tranexamic acid: antifibrinolytic therapy
| Treatment | Mechanism | Efficacy | Duration | Side Effects | Contraindications |
|---|---|---|---|---|---|
| COCs | Endometrial atrophy | 40-50% reduction | Continuous | Nausea, headache | Thromboembolism risk |
| LNG-IUD | Local progestin | 90% reduction | 5 years | Irregular bleeding | Uterine anomalies |
| Tranexamic acid | Antifibrinolytic | 40-50% reduction | Cycle days 1-5 | GI upset | Thrombosis history |
| NSAIDs | Prostaglandin inhibition | 20-30% reduction | Cycle days 1-3 | Gastric irritation | Peptic ulcer disease |
| DMPA | Ovulation suppression | 50% amenorrhea | 3 months | Weight gain, bone loss | Osteoporosis risk |
Surgical Intervention Strategies
Minimally invasive procedures offer definitive treatment for structural abnormalities and medication-refractory bleeding. Hysteroscopic interventions achieve success rates >90% for appropriate candidates.
-
Hysteroscopic Procedures
- Polypectomy: first-line for endometrial polyps
- Success rate: >95% for symptom resolution
- Recurrence: <5% at 2 years
- Malignancy detection: 2-5% in reproductive age
- Submucosal myomectomy: fertility-preserving option
- Type 0 fibroids: completely intracavitary, easiest resection
- Type 1 fibroids: <50% intramural, good candidates
- Type 2 fibroids: >50% intramural, challenging resection
- Polypectomy: first-line for endometrial polyps
-
Endometrial Ablation
- Second-generation techniques: global ablation methods
- Thermal balloon: 87% success at 5 years
- Microwave ablation: 90% satisfaction at 2 years
- Radiofrequency: 85% amenorrhea/hypomenorrhea at 1 year
- Patient selection criteria
- Completed childbearing: pregnancy contraindicated post-ablation
- Normal endometrial cavity: no submucous fibroids >3cm
- Endometrial thickness <12mm: optimal ablation conditions
- Second-generation techniques: global ablation methods
💡 Master This: Endometrial ablation achieves amenorrhea in 45-60% and significant improvement in 85-90% of patients. Pregnancy rates post-ablation are <1% but carry high complication risk requiring reliable contraception.
Disorder-Specific Protocols
Targeted therapy addresses underlying pathophysiology for specific conditions, achieving superior outcomes compared to symptomatic treatment alone.
-
PCOS Management
- First-line: combined oral contraceptives
- Cycle regulation: >90% achieve regular cycles
- Androgen suppression: 50-70% reduction in hirsutism
- Endometrial protection: reduces cancer risk by 50%
- Insulin sensitizers: metformin 1500-2000mg daily
- Ovulation restoration: 50-60% in insulin-resistant patients
- Weight loss: 5-10% reduction in responsive patients
- Cardiovascular benefits: improved lipid profiles
- First-line: combined oral contraceptives
-
Thyroid Disorder Management
- Hypothyroidism: levothyroxine replacement
- Target TSH: 0.4-2.5 mIU/L for reproductive age
- Cycle normalization: 80% within 3 months of euthyroid state
- Fertility improvement: pregnancy rates increase 2-3 fold
- Hyperthyroidism: antithyroid medications
- Methimazole: preferred except first trimester pregnancy
- Cycle restoration: 65% achieve regular menses within 6 months
- Hypothyroidism: levothyroxine replacement
⭐ Clinical Pearl: Weight loss of 5-10% in overweight PCOS patients restores ovulation in 55-100% of cases and improves insulin sensitivity by 25-30%. Lifestyle modification should be first-line therapy before pharmacologic intervention.
This comprehensive treatment approach ensures individualized care that addresses specific pathophysiology while optimizing patient outcomes and quality of life. Evidence-based protocols guide therapy selection and monitoring strategies for optimal long-term results.
🛠️ Treatment Mastery: The Therapeutic Command Center
🎯 Clinical Mastery Arsenal: The Expert's Rapid Reference
Essential Clinical Thresholds
Master these numbers for instant clinical application and evidence-based decision-making:
📌 Critical Numbers Arsenal:
- Normal cycle: 21-35 days, 3-7 days duration, 5-40ml loss
- Heavy bleeding: >80ml or >7 days or hemoglobin drop >2g/dl
- Endometrial thickness: <4mm postmenopausal, <16mm premenopausal
- PCOS criteria: 2 of 3 - oligoovulation, hyperandrogenism, PCO morphology
- Prolactin: >25ng/ml abnormal, >100ng/ml suggests adenoma
- TSH: >4.5 mIU/L affects cycles, >10 mIU/L causes infertility
Rapid Diagnostic Framework
| Clinical Presentation | Age Group | First-Line Tests | Key Threshold | Immediate Action |
|---|---|---|---|---|
| Heavy regular bleeding | <20 years | CBC, coagulation | Hgb <10 g/dl | Iron + hormonal therapy |
| Irregular bleeding | 20-39 years | βhCG, TSH, prolactin | βhCG >5 mIU/ml | Pregnancy evaluation |
| New heavy bleeding | >40 years | Imaging + biopsy | Endometrium >16mm | Tissue sampling |
| Amenorrhea + galactorrhea | Any age | Prolactin, MRI | Prolactin >100 ng/ml | Pituitary imaging |
| Hirsutism + oligomenorrhea | Reproductive | Testosterone, DHEAS | Free T >70 ng/dl | PCOS evaluation |
Treatment Decision Matrix
Evidence-based treatment selection based on patient characteristics and clinical priorities:
- First-Line Therapies by Clinical Scenario
- Adolescent heavy bleeding: combined oral contraceptives (80% success)
- Adult heavy bleeding + contraception needed: levonorgestrel IUD (90% reduction)
- Heavy bleeding + contraception contraindicated: tranexamic acid (40% reduction)
- PCOS + insulin resistance: metformin + COCs (70% cycle regulation)
- Structural lesions: hysteroscopic resection (>95% success)
💡 Clinical Pearl: Levonorgestrel IUD provides superior long-term outcomes with 90% bleeding reduction, 95% satisfaction, and 50% amenorrhea by 2 years. Cost-effective compared to medical therapy beyond 1 year.
Red Flag Recognition
Immediate evaluation required for these high-risk presentations:
⚠️ Emergency Indicators:
- Hemoglobin <7g/dl: transfusion consideration
- Postmenopausal bleeding: 10% malignancy risk
- Bleeding + positive pregnancy test: ectopic pregnancy until proven otherwise
- Acute severe pain + bleeding: ovarian torsion or rupture
- Prepubertal bleeding: trauma or malignancy evaluation
This clinical mastery framework enables rapid, accurate assessment and evidence-based management of menstrual disorders, ensuring optimal patient outcomes through systematic, expert-level clinical decision-making.
🎯 Clinical Mastery Arsenal: The Expert's Rapid Reference
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















