Fertility and Infertility

On this page
🌱 Fertility and Infertility: The Reproductive Symphony
Fertility hinges on a precisely timed hormonal cascade, coordinated follicular maturation, robust spermatogenesis, and the intricate biology of fertilization-when any link falters, infertility emerges. You'll master the neuroendocrine axes governing reproduction, trace oocyte and sperm development from primordial stages to competent gametes, and learn the systematic clinical approach to diagnosing and treating infertility. This lesson equips you to recognize disruptions in the reproductive symphony and deploy evidence-based interventions that restore the possibility of conception.
📌 Remember: FERTILE - Follicles mature, Estrogen rises, Receptive endometrium, Timing ovulation, Implantation window, Luteal support, Embryonic development. Each component must synchronize perfectly for conception success.
The reproductive timeline operates with remarkable precision: ovulation occurs within 24-36 hours of the LH surge, sperm remain viable for 72-120 hours in optimal cervical mucus, and the implantation window spans just 2-4 days during the mid-luteal phase. These narrow timeframes explain why even fertile couples have only a 20-25% chance of conception per cycle.
Modern fertility assessment has revolutionized with advanced diagnostics. Anti-Müllerian hormone (AMH) levels predict ovarian reserve with 85% accuracy, while 3D ultrasound detects uterine anomalies in 95% of cases. Semen analysis parameters have evolved beyond basic counts-DNA fragmentation indices above 30% significantly impact fertilization rates, and strict morphology below 4% normal forms correlates with reduced pregnancy rates.
⭐ Clinical Pearl: Couples under 35 years should seek evaluation after 12 months of unprotected intercourse, while those over 35 warrant assessment after 6 months. This age-based approach reflects the exponential decline in female fertility-from 25% monthly conception rates at age 25 to 5% at age 40.
| Age Group | Monthly Conception Rate | Time to Conception (50%) | Time to Conception (90%) | Miscarriage Risk | Live Birth Rate (IVF) |
|---|---|---|---|---|---|
| 20-24 years | 25% | 3 months | 12 months | 10% | 55% |
| 25-29 years | 23% | 4 months | 15 months | 12% | 50% |
| 30-34 years | 18% | 6 months | 20 months | 15% | 42% |
| 35-39 years | 12% | 9 months | 30 months | 25% | 32% |
| 40-44 years | 5% | 24 months | 60+ months | 40% | 15% |
💡 Master This: The "fertile window" extends 6 days before ovulation through 24 hours after, but peak fertility occurs in the 3 days preceding ovulation. This knowledge transforms timing strategies and explains why ovulation prediction kits detecting LH surge optimize conception chances.
Environmental factors increasingly influence fertility patterns. Air pollution exposure reduces sperm concentration by 15-20%, while endocrine disruptors in plastics decrease testosterone levels by 10-15%. Lifestyle modifications can reverse many factors-weight loss of 5-10% in obese women restores ovulation in 70% of cases, while smoking cessation improves sperm parameters within 3 months.
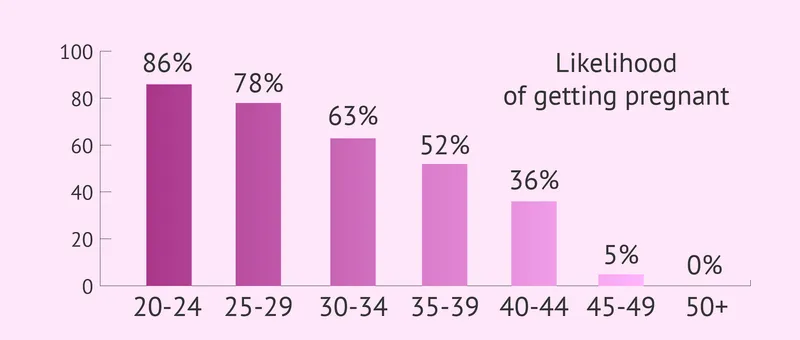
Understanding fertility and infertility requires integrating reproductive physiology with clinical assessment skills, where pattern recognition and systematic evaluation guide successful outcomes. Connect these foundational concepts through hormonal orchestration to understand the precise mechanisms driving reproductive success.
🌱 Fertility and Infertility: The Reproductive Symphony
🎼 Hormonal Orchestration: The Reproductive Command Center
📌 Remember: PULSE - Pulsatile GnRH release, Upstream hypothalamic control, LH and FSH secretion, Steroid hormone feedback, Estrogen and progesterone effects. Disruption at any level cascades through the entire reproductive axis.
FSH initiates follicular recruitment during the first 5 days of the cycle, with levels peaking at 8-12 IU/L in reproductive-age women. The dominant follicle emerges by day 7-8, producing increasing estradiol levels that create negative feedback, suppressing FSH to 3-5 IU/L by mid-cycle. This FSH decline prevents multiple ovulations-a protective mechanism that occasionally fails, resulting in 1-2% natural twin rates.
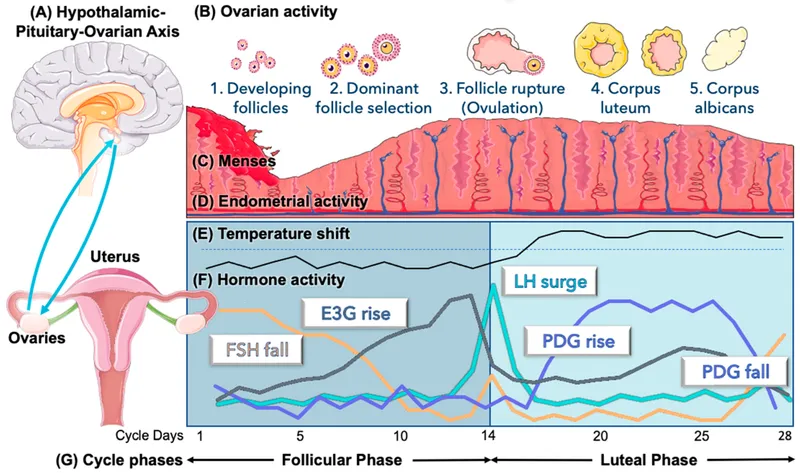
The LH surge represents reproductive physiology's most dramatic event. Estradiol levels above 200 pg/mL for 48+ hours trigger positive feedback, causing LH to spike from baseline 5-10 IU/L to peak levels of 40-100 IU/L within 24-36 hours. This surge initiates the ovulation cascade: follicular wall weakening, cumulus expansion, and oocyte maturation completion.
⭐ Clinical Pearl: The LH surge occurs 34-36 hours before ovulation, making LH detection kits valuable for timing intercourse or intrauterine insemination. Peak fertility occurs 12-24 hours before ovulation, when cervical mucus reaches optimal spinnbarkeit of 10+ cm and ferning patterns appear.
| Cycle Phase | Duration | FSH (IU/L) | LH (IU/L) | Estradiol (pg/mL) | Progesterone (ng/mL) | Key Events |
|---|---|---|---|---|---|---|
| Early Follicular | Days 1-5 | 8-12 | 5-10 | 20-50 | <1 | Follicle recruitment |
| Late Follicular | Days 6-13 | 3-8 | 5-15 | 50-300 | <1 | Dominant follicle |
| Ovulatory | Days 14-16 | 5-15 | 40-100 | 200-400 | 1-3 | LH surge, ovulation |
| Early Luteal | Days 17-21 | 2-8 | 2-10 | 100-200 | 5-15 | Corpus luteum |
| Late Luteal | Days 22-28 | 3-10 | 2-8 | 50-150 | 10-25 | Luteal maintenance |
💡 Master This: Progesterone's thermogenic effect raises basal body temperature by 0.4-0.6°F post-ovulation, creating the biphasic pattern essential for confirming ovulation. This temperature shift, sustained for 12-14 days, indicates adequate corpus luteum function and successful ovulation.
The menstrual cycle's precision extends beyond hormone levels to cellular interactions. Granulosa cells express FSH receptors and produce estradiol through aromatase activity, while theca cells respond to LH and produce androgens as estrogen precursors. This two-cell, two-gonadotropin system explains why both FSH and LH are essential for normal ovarian function.
Disruptions in hormonal orchestration manifest in predictable patterns. Hypothalamic dysfunction from stress, weight loss, or excessive exercise creates hypogonadotropic hypogonadism with low FSH and LH levels. PCOS demonstrates hyperandrogenism with elevated LH:FSH ratios typically >2:1, while premature ovarian insufficiency shows hypergonadotropic hypogonadism with FSH levels >40 IU/L.
Understanding hormonal orchestration reveals how reproductive timing mechanisms create the foundation for successful conception, where precise feedback loops and cellular interactions determine fertility outcomes. Connect these hormonal patterns through ovarian dynamics to understand how follicular development and ovulation create the reproductive opportunity window.
🎼 Hormonal Orchestration: The Reproductive Command Center
🥚 Ovarian Dynamics: The Follicular Development Theater
📌 Remember: STAGES - Small primordial start, Transition to primary, Antral cavity formation, Graafian follicle maturity, Estrogen peak production, Surge triggers ovulation. Each stage requires specific growth factors and hormonal support for progression.
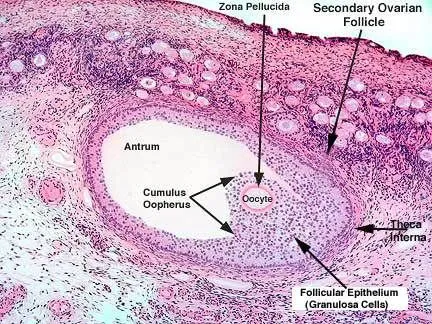
The follicular development timeline spans three distinct phases. The initial 65 days involve gonadotropin-independent growth from primordial to early antral stages, driven by local growth factors including kit ligand, bone morphogenetic proteins, and anti-Müllerian hormone. The final 20 days require FSH stimulation for continued growth, with the dominant follicle emerging during the first week of the menstrual cycle.
Antral follicle count (AFC) serves as the most reliable ovarian reserve marker. Transvaginal ultrasound performed during days 2-5 of the cycle counts follicles measuring 2-10mm in diameter. Normal AFC ranges from 15-25 in women under 35, declining to 5-10 by age 40. AFC below 5 indicates diminished ovarian reserve, while counts above 25 suggest PCOS.
⭐ Clinical Pearl: Anti-Müllerian hormone (AMH) levels correlate strongly with AFC and predict ovarian response to stimulation. Normal AMH ranges from 1.5-4.0 ng/mL in reproductive-age women, with levels below 1.0 ng/mL indicating poor ovarian reserve and values above 5.0 ng/mL suggesting PCOS.
| Age Group | Normal AFC | AMH Range (ng/mL) | Expected IVF Response | Pregnancy Rate/Cycle | Ovarian Reserve Status |
|---|---|---|---|---|---|
| 25-30 years | 20-30 | 2.5-6.0 | 15-20 oocytes | 25-30% | Excellent |
| 31-35 years | 15-25 | 1.5-4.0 | 10-15 oocytes | 20-25% | Good |
| 36-40 years | 10-20 | 1.0-2.5 | 8-12 oocytes | 15-20% | Fair |
| 41-43 years | 5-15 | 0.5-1.5 | 5-8 oocytes | 8-12% | Poor |
| >44 years | <10 | <0.5 | <5 oocytes | <5% | Very Poor |
Follicular fluid composition changes dramatically during development. Early antral follicles contain high concentrations of anti-Müllerian hormone and inhibin B, while preovulatory follicles show elevated estradiol levels reaching 200-400 pg/mL per follicle. The LH surge triggers dramatic changes: hyaluronic acid increases 10-fold, creating cumulus expansion, while proteolytic enzymes weaken the follicular wall.
💡 Master This: The "two-cell, two-gonadotropin" system explains estrogen production. Theca cells respond to LH by producing androgens, which diffuse to granulosa cells where FSH-stimulated aromatase converts them to estradiol. This cooperation explains why both gonadotropins are essential for normal follicular function.
Ovulation mechanics involve precise molecular events occurring over 36-44 hours post-LH surge. Cumulus cells surrounding the oocyte expand through hyaluronic acid synthesis, while the follicular wall weakens via collagenase and plasmin activation. The oocyte completes meiosis I, extruding the first polar body and arresting in metaphase II until fertilization.
Corpus luteum formation transforms the ruptured follicle into an endocrine powerhouse. Granulosa and theca cells luteinize under LH influence, producing progesterone levels of 15-25 ng/mL by 7 days post-ovulation. Without pregnancy, the corpus luteum regresses after 12-14 days, causing progesterone withdrawal and menstruation initiation.
Understanding ovarian dynamics reveals how follicular development creates the cellular foundation for reproduction, where precise timing and hormonal coordination determine ovulation success. Connect these ovarian processes through male reproductive physiology to understand how sperm production and function complement female fertility mechanisms.
🥚 Ovarian Dynamics: The Follicular Development Theater
🏃♂️ Male Reproductive Mastery: The Sperm Production Pipeline
📌 Remember: SPERM - Sertoli cell support, Proliferation phase (16 days), Epididymal maturation, Reproductive competence, Mature gamete production. The entire process from stem cell to mature sperm requires 74-84 days, explaining why lifestyle changes take 3 months to impact semen quality.
The blood-testis barrier creates a unique immunological environment essential for spermatogenesis. Sertoli cells form tight junctions that prevent immune recognition of developing sperm, which express antigens not present during early development. This barrier also regulates the microenvironment, maintaining optimal concentrations of hormones, growth factors, and nutrients.
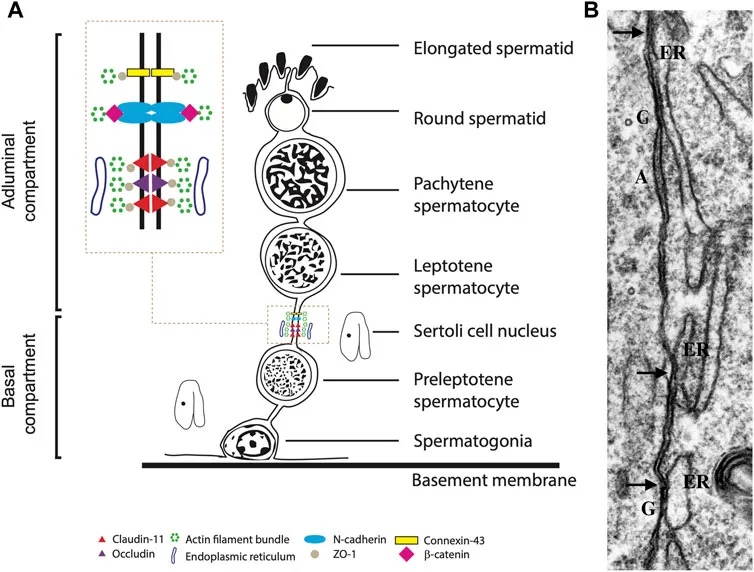
Normal semen parameters reflect the efficiency of this production system. The WHO 2010 criteria establish reference values: concentration ≥15 million/mL, total count ≥39 million, motility ≥40%, progressive motility ≥32%, and normal morphology ≥4%. These thresholds represent the 5th percentile of fertile men, meaning 95% of men with proven fertility exceed these values.
⭐ Clinical Pearl: Sperm concentration varies dramatically within individuals-2-3 fold differences between samples are normal. This variability necessitates two separate analyses at least 7 days apart for accurate assessment. Abstinence periods of 2-7 days optimize sample quality.
| Parameter | WHO 2010 Reference | Optimal Range | Clinical Significance | Impact on Fertility |
|---|---|---|---|---|
| Volume | ≥1.5 mL | 2.0-5.0 mL | Accessory gland function | Low volume suggests obstruction |
| Concentration | ≥15 million/mL | 20-200 million/mL | Testicular function | <5 million severely impaired |
| Total Count | ≥39 million | 50-300 million | Overall production | <10 million poor prognosis |
| Motility | ≥40% | 50-80% | Sperm function | <20% requires ART |
| Morphology | ≥4% | 5-15% | Genetic integrity | <2% DNA fragmentation risk |
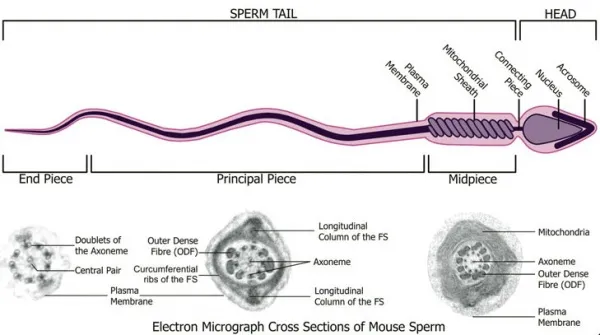
Epididymal maturation transforms testicular sperm into fertilization-competent gametes. During the 10-14 day epididymal transit, sperm acquire progressive motility, develop capacitation potential, and undergo membrane modifications essential for zona pellucida binding. Epididymal dysfunction, often subclinical, contributes to unexplained male infertility.
💡 Master This: Capacitation represents sperm's final maturation step, occurring in the female reproductive tract over 4-6 hours. This process involves cholesterol efflux, protein tyrosine phosphorylation, and hyperactivated motility development, enabling zona pellucida penetration and fertilization.
Male fertility assessment extends beyond semen analysis to include hormonal evaluation. Testosterone levels below 300 ng/dL indicate hypogonadism, while FSH elevation above 7.6 IU/L suggests primary testicular dysfunction. LH:FSH ratios help distinguish hypothalamic-pituitary disorders from primary gonadal failure.
Environmental factors significantly impact spermatogenesis. Heat exposure above 35°C for 30+ minutes daily reduces sperm concentration by 15-20%. Varicoceles, present in 15% of general population and 40% of infertile men, create scrotal hyperthermia and oxidative stress. Surgical repair improves semen parameters in 60-70% of cases.
Oxidative stress represents a major threat to sperm function, affecting 30-40% of infertile men. Reactive oxygen species damage sperm membranes, DNA, and mitochondria, reducing motility and fertilization capacity. Antioxidant supplementation with vitamin C, vitamin E, and coenzyme Q10 improves sperm parameters in 60% of men with elevated oxidative stress markers.
Understanding male reproductive mastery reveals how sperm production and function create the paternal contribution to conception, where cellular precision and environmental optimization determine fertilization success. Connect these male factors through fertilization dynamics to understand how sperm-egg interaction creates the foundation for embryonic development.
🏃♂️ Male Reproductive Mastery: The Sperm Production Pipeline
🎯 Fertilization Dynamics: The Conception Convergence
📌 Remember: JOURNEY - Journey through cervix, Oviduct navigation, Uterine transit, Reservoir formation, Navigation to ampulla, Egg encounter, Yielding fertilization. Each step eliminates defective sperm, creating a natural selection process for fertilization competence.
The cervical mucus barrier changes dramatically throughout the menstrual cycle. During the fertile window, estrogen stimulation creates 98% water content with parallel protein fibers, allowing sperm penetration. Post-ovulation, progesterone transforms mucus into a viscous barrier with <95% water content and cross-linked proteins, effectively blocking sperm passage.
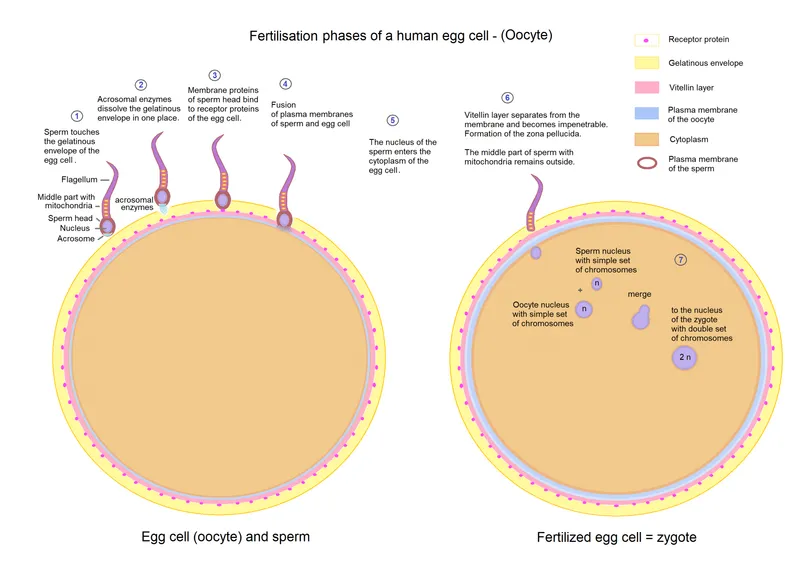
Capacitation transforms sperm from transport-competent to fertilization-competent cells. This 4-6 hour process involves cholesterol efflux from sperm membranes, protein tyrosine phosphorylation, and calcium influx. Capacitated sperm develop hyperactivated motility with 150+ μm flagellar amplitude, essential for zona pellucida penetration.
⭐ Clinical Pearl: The cumulus-oocyte complex creates a 200-300 μm barrier surrounding the egg. Hyaluronidase released from capacitated sperm disperses cumulus cells, while hyperactivated motility enables penetration through this expanded matrix. Cumulus expansion occurs 12-18 hours before ovulation.
| Fertilization Stage | Duration | Key Events | Success Rate | Critical Factors |
|---|---|---|---|---|
| Sperm Transport | 30min-6hrs | Cervical penetration, uterine transit | 0.1% reach tubes | Mucus quality, motility |
| Capacitation | 4-6 hours | Membrane changes, hyperactivation | 60-80% of sperm | Calcium, bicarbonate |
| Zona Binding | 15-30 min | ZP3 recognition, acrosome reaction | 10-20% bind | Sperm morphology |
| Zona Penetration | 15-30 min | Enzymatic digestion, motility | 1-5% penetrate | Acrosomal enzymes |
| Membrane Fusion | 1-5 min | Sperm-egg fusion, cortical reaction | >95% if reached | Membrane integrity |
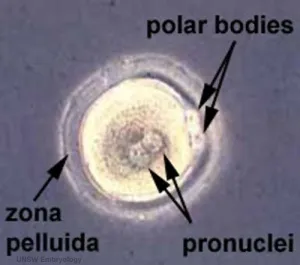
Sperm-egg membrane fusion initiates a cascade of activation events. Sperm entry triggers calcium oscillations in the oocyte, causing cortical granule exocytosis that modifies the zona pellucida to prevent polyspermy. The oocyte completes meiosis II, extruding the second polar body, while sperm and egg pronuclei form and migrate toward each other.
💡 Master This: Pronuclear formation and fusion represent fertilization's culmination. The sperm nucleus decondenses over 6-8 hours, forming the male pronucleus, while the egg forms the female pronucleus. These structures migrate together, and their membranes dissolve, allowing chromosome alignment for the first mitotic division.
Fertilization timing critically impacts success rates. Insemination 6 hours before ovulation yields optimal pregnancy rates, while insemination 24+ hours post-ovulation shows dramatically reduced success. This narrow window reflects oocyte aging, where delayed fertilization increases chromosomal abnormalities and embryonic arrest rates.
Molecular mechanisms of fertilization reveal remarkable specificity. Sperm surface protein IZUMO1 and egg surface protein JUNO mediate membrane fusion, while sperm-specific phospholipase C zeta triggers calcium oscillations. These discoveries explain some cases of fertilization failure and guide development of assisted reproductive technologies.
Fertilization assessment in clinical practice relies on pronuclear observation 16-18 hours post-insemination. Normal fertilization shows two distinct pronuclei with aligned nucleoli, indicating successful sperm-egg fusion and proper genetic activation. Abnormal fertilization patterns-including 0, 1, or 3+ pronuclei-occur in 15-25% of inseminated oocytes.
Understanding fertilization dynamics reveals how gamete interaction creates the foundation for embryonic development, where molecular precision and temporal coordination determine conception success. Connect these fertilization mechanisms through clinical assessment strategies to understand how systematic evaluation identifies and addresses fertility challenges.
🎯 Fertilization Dynamics: The Conception Convergence
🔬 Clinical Assessment Mastery: The Fertility Detective Framework
📌 Remember: DETECTIVE - Detailed history taking, Examination findings, Timing of tests, Evaluate ovulation, Check sperm parameters, Tubal patency assessment, Imaging studies, Verify uterine factors, Endocrine evaluation. Systematic investigation prevents missed diagnoses.
Female evaluation begins with comprehensive history and physical examination. Menstrual cycle regularity indicates ovulatory function-cycles 21-35 days with <4 day variation suggest normal ovulation in 95% of cases. Hirsutism, acne, and weight gain point toward PCOS, while galactorrhea suggests hyperprolactinemia. Previous pregnancies, contraceptive history, and sexually transmitted infections provide crucial diagnostic clues.
Ovulation assessment utilizes multiple complementary methods. Basal body temperature charting shows biphasic patterns with 0.4-0.6°F elevation post-ovulation, while ovulation predictor kits detect LH surge 24-36 hours before ovulation. Mid-luteal progesterone levels >3 ng/mL confirm ovulation, with levels >10 ng/mL indicating adequate luteal function.
⭐ Clinical Pearl: Day 21 progesterone testing assumes a 28-day cycle with day 14 ovulation. For irregular cycles, test 7 days after suspected ovulation based on LH surge or temperature rise. Progesterone levels >15 ng/mL suggest excellent luteal function and optimal endometrial preparation.
| Assessment Method | Timing | Normal Values | Clinical Significance | Limitations |
|---|---|---|---|---|
| BBT Charting | Daily throughout cycle | Biphasic pattern, 0.4-0.6°F rise | Confirms ovulation occurred | Retrospective, affected by illness |
| LH Predictor Kits | Mid-cycle daily | Positive surge detection | Predicts ovulation 24-36hrs | Doesn't confirm ovulation |
| Mid-luteal Progesterone | 7 days post-ovulation | >10 ng/mL | Confirms ovulation, luteal adequacy | Single measurement limitation |
| Transvaginal Ultrasound | Serial monitoring | Follicle >18mm, corpus luteum | Real-time follicle tracking | Requires multiple visits |
| Endometrial Biopsy | Late luteal phase | Secretory changes | Luteal phase assessment | Invasive, rarely performed |
Tubal patency assessment utilizes hysterosalpingography (HSG) as the gold standard screening test. Performed during days 7-10 of the cycle, HSG demonstrates tubal anatomy and patency with 85% sensitivity for tubal occlusion. Laparoscopy with chromopertubation provides definitive assessment but requires surgical intervention.
💡 Master This: HSG timing is crucial-perform after menstruation ends but before ovulation to avoid disrupting a potential pregnancy. Oil-based contrast media may have therapeutic effects, with pregnancy rates 10-15% higher in the 6 months following HSG compared to water-based contrast.
Uterine factor assessment includes transvaginal ultrasound to evaluate anatomy and endometrial thickness. Mid-luteal endometrial thickness >7mm indicates adequate estrogen stimulation, while thickness >10mm suggests optimal implantation potential. Saline infusion sonohysterography (SIS) detects intrauterine pathology with 95% sensitivity for polyps and fibroids.
Hormonal evaluation targets specific clinical scenarios. Day 3 FSH and estradiol levels assess ovarian reserve, with FSH >10 IU/L or estradiol >80 pg/mL suggesting diminished reserve. TSH screening detects thyroid dysfunction in 5-10% of infertile women, while prolactin elevation occurs in 15% of women with ovulatory dysfunction.
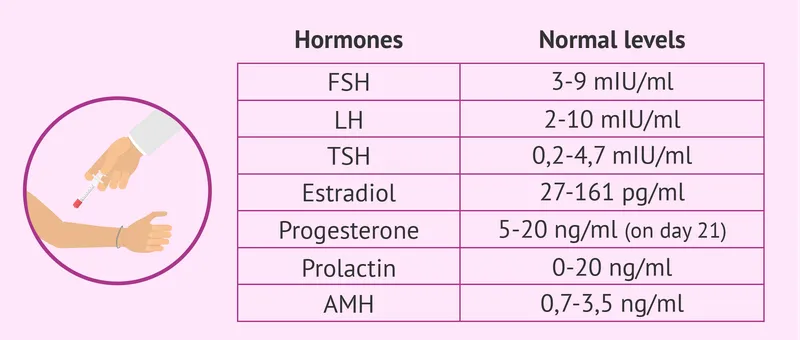
Advanced testing addresses specific clinical scenarios. Anti-Müllerian hormone (AMH) provides the most accurate ovarian reserve assessment, remaining stable throughout the cycle. Antral follicle count (AFC) complements AMH, with both tests predicting ovarian response to stimulation and guiding treatment protocols.
Understanding clinical assessment mastery reveals how systematic evaluation identifies fertility challenges, where comprehensive testing and logical sequencing create the foundation for targeted treatment strategies. Connect these diagnostic approaches through treatment optimization to understand how evidence-based interventions restore reproductive potential and maximize conception success.
🔬 Clinical Assessment Mastery: The Fertility Detective Framework
🚀 Treatment Optimization: The Fertility Restoration Arsenal
📌 Remember: OPTIMIZE - Ovulation induction protocols, Patient-specific dosing, Timing coordination, Individualized monitoring, Multiple treatment cycles, Implantation enhancement, Zygote development support, Embryo transfer optimization. Each component requires precise calibration for maximum success.
Ovulation induction represents the most successful fertility intervention. Clomiphene citrate, starting at 50mg daily for 5 days, induces ovulation in 70-80% of anovulatory women with 40-50% achieving pregnancy within 6 cycles. Letrozole demonstrates superior efficacy in PCOS patients, with 22% live birth rates per cycle compared to 18% with clomiphene.
Gonadotropin therapy provides more potent ovulation induction for clomiphene-resistant patients. Starting doses of 75-150 IU FSH daily require careful monitoring to prevent ovarian hyperstimulation syndrome (OHSS). The "step-up" protocol increases doses by 37.5-75 IU every 5-7 days until follicular development occurs, achieving ovulation in 90-95% of patients.
⭐ Clinical Pearl: Intrauterine insemination (IUI) timing is critical for success. Perform IUI 24-36 hours after hCG trigger or 12-24 hours after natural LH surge. Sperm preparation concentrates motile sperm to 10-20 million total motile count, with success rates 15-20% per cycle in unexplained infertility.
| Treatment Modality | Success Rate/Cycle | Cumulative Success (6 cycles) | Cost Range | Ideal Candidates |
|---|---|---|---|---|
| Clomiphene + TI | 15-20% | 60-70% | $100-200 | Anovulation, unexplained |
| Letrozole + TI | 18-25% | 65-75% | $150-250 | PCOS, anovulation |
| Clomiphene + IUI | 18-22% | 70-80% | $500-800 | Mild male factor, unexplained |
| Gonadotropins + IUI | 20-25% | 75-85% | $2000-3000 | Clomiphene resistance |
| IVF | 35-45% | 85-95% | $12000-15000 | Tubal factor, severe male factor |
Embryo transfer optimization significantly impacts IVF success. Single embryo transfer (SET) reduces multiple pregnancy rates from 25-30% to <5% while maintaining acceptable pregnancy rates in good-prognosis patients. Blastocyst transfer on day 5 shows 10-15% higher implantation rates compared to day 3 cleavage-stage transfer.
💡 Master This: Preimplantation genetic testing (PGT) revolutionizes embryo selection. PGT-A (aneuploidy screening) improves implantation rates from 35-40% to 60-70% per transfer in women over 35. This technology reduces miscarriage rates and time to pregnancy while enabling confident single embryo transfer.
Surgical interventions address anatomical factors affecting fertility. Laparoscopic treatment of endometriosis improves pregnancy rates by 30-50% in mild to moderate disease, while hysteroscopic removal of intrauterine pathology restores normal conception rates in 80-90% of cases. Tubal surgery success depends on disease severity-pregnancy rates range from 70% for mild adhesions to 20% for severe hydrosalpinx.
Male factor treatment has evolved significantly with advanced reproductive techniques. Intracytoplasmic sperm injection (ICSI) overcomes severe male factor infertility, achieving fertilization rates >80% even with very low sperm counts. Testicular sperm extraction (TESE) enables biological fatherhood in men with azoospermia, with sperm retrieval rates 40-60% in non-obstructive azoospermia.
Lifestyle optimization enhances treatment success across all modalities. Weight loss of 5-10% in obese women improves ovulation rates and pregnancy outcomes, while smoking cessation increases IVF success rates by 30-50%. Antioxidant supplementation improves sperm parameters in 60% of men with elevated oxidative stress markers.
Treatment monitoring requires precise coordination of multiple parameters. Ultrasound monitoring tracks follicular development, with lead follicles ≥18mm indicating maturity. Estradiol levels guide stimulation adjustments, with levels >200 pg/mL per mature follicle suggesting adequate response. LH monitoring prevents premature ovulation during controlled stimulation cycles.
Understanding treatment optimization reveals how evidence-based interventions restore reproductive potential, where individualized protocols and precise monitoring create the foundation for conception success and the realization of parenthood dreams.
🚀 Treatment Optimization: The Fertility Restoration Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app

















