Radiation's Cellular Hit - Target Practice
- Direct Action: Radiation directly ionizes key cellular targets (DNA, RNA, proteins). Predominant with high-LET radiation (α, neutrons). Causes base damage, SSBs, DSBs.
- Indirect Action: Radiation interacts with $H_2O$, creating free radicals.
- $H_2O \xrightarrow{radiation} H_2O^{\cdot+} + e^- \rightarrow H_3O^+ + OH^\cdot$
- $OH^\cdot$ (hydroxyl radical) is most damaging.
⭐ Indirect action, mediated by free radicals like the hydroxyl radical, accounts for about 2/3 of DNA damage from low-LET radiation (X-rays, γ-rays).
- Target Theory: Cell death if critical targets (DNA) hit. $D_0$: dose for 37% survival.
DNA's Radiation Wounds - Code Red & Fix

- DNA: Principal target for radiation cell death & heritable effects.
- Damage Mechanisms:
- Direct Action: Radiation directly ionizes DNA.
- Indirect Action: Via H₂O radiolysis (OH• radicals); dominant for X/γ-rays.
- Lesion Types:
- Base Damage (BD): e.g., 8-oxoG.
- Single-Strand Breaks (SSBs): Usually readily repaired.
- Double-Strand Breaks (DSBs): Most lethal; cause mutations/aberrations.
- DNA-Protein Crosslinks (DPCs)
- Clustered Lesions: Multiple damages nearby.
- Repair Pathways:
- Base Excision Repair (BER): For non-bulky base damage.
- Nucleotide Excision Repair (NER): For bulky adducts.
- Single-Strand Break Repair (SSBR): Involves PARP.
- DSB Repair:
- NHEJ (Non-Homologous End Joining): Fast, error-prone; G1, S, G2 phases.
- HRR (Homologous Recombination): Accurate, uses sister chromatid; late S/G2.
⭐ Double-Strand Breaks (DSBs) are the most critical lesions for radiation-induced cell killing, chromosomal aberrations, and mutations.
Cell Cycle Sensitivity - Phase Fright
- Cell vulnerability to radiation fluctuates through its cycle.
- Most Radiosensitive Phases:
- M phase (Mitosis): Chromosomes condensed, limited repair time.
- G2 phase: Pre-mitotic checkpoint, damage assessed before division.
- Most Radioresistant Phase:
- Late S phase: DNA homologous recombination repair most active.
- Intermediate Sensitivity:
- G1 phase, Early S phase.
- 📌 Mnemonic: "Mighty G2 Guards, Late S Survives."

⭐ Cells are generally most radiosensitive in M and G2 phases, and most radioresistant during the late S phase due to efficient DNA repair mechanisms.
Cell Death & Survival - Counting Casualties
-
Linear-Quadratic (LQ) Model: Describes cell kill: $S = e^{-(\alpha D + \beta D^2)}$
- $\alpha$: linear component (non-repairable damage).
- $\beta$: quadratic component (repairable damage interaction).
- $\alpha/\beta$ ratio: Dose where linear kill ($\alpha D$) equals quadratic kill ($\beta D^2$).
-
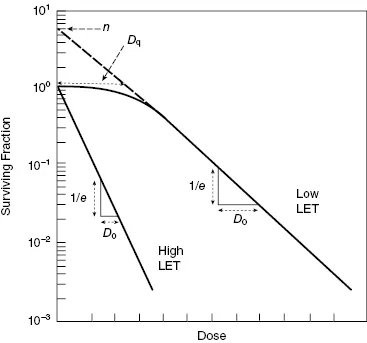
Cell Survival Curve: Plots surviving fraction vs. dose.
- $D_0$: Mean lethal dose (dose to reduce survival to 37% on exponential part). $\uparrow D_0 = \uparrow$Radioresistance.
- $D_q$: Quasi-threshold dose (measures shoulder, SLDR capacity).
- $n$: Extrapolation number (also measures shoulder).
-
Modes of Radiation-Induced Cell Death:
Death Type Feature RT Relevance Mitotic Catastrophe Mitotic failure post-irradiation Most common Apoptosis Programmed; p53 dependent/independent Cell-type dependent Necrosis Uncontrolled; inflammation High dose effect Senescence Irreversible G1 arrest Contributes to effect
⭐ The alpha/beta ratio is key in radiotherapy. Low (2-4 Gy) for late-responding tissues (more fractionation sensitive); high (8-10 Gy) for early-responding tissues & most tumors (less fractionation sensitive).
Response Modifiers - Dialing Damage
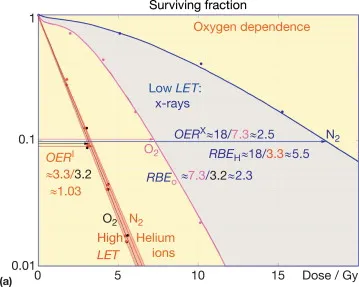
- Oxygen Effect: ↑ radiosensitivity.
- OER ($OER = \frac{Dose\ in\ hypoxia}{Dose\ in\ aerated\ conditions}$):
Radiation Type OER X-rays/γ-rays 2.5-3.5 Neutrons ~1.6 α-particles 1 - Mechanism: Fixes free-radical DNA damage.
- OER ($OER = \frac{Dose\ in\ hypoxia}{Dose\ in\ aerated\ conditions}$):
⭐ Oxygen is the most potent radiosensitizer; its effect is maximal at $pO_2$ levels around 20-30 mmHg and negligible for high-LET radiations.
- Radiosensitizers:
- Halogenated pyrimidines (5-BUdR, 5-IUdR)
- Hypoxic sensitizers (Misonidazole)
- Radioprotectors:
- Sulfhydryl compounds (Amifostine/WR-2721) - free radical scavengers. DRF ~2.7.
- Cell Cycle:
- Most sensitive: M, G2 phases.
- Most resistant: Late S phase. (📌 Mnemonic: Mighty God 2 kill, Strong to survive)
 vs pO2 OR Graph of RBE vs LET)
vs pO2 OR Graph of RBE vs LET)
High‑Yield Points - ⚡ Biggest Takeaways
- DNA is the principal target for radiation-induced cell death.
- Double-Strand Breaks (DSBs) are the most lethal DNA damage.
- Cells are most radiosensitive in M and G2 phases; most resistant in late S phase.
- Oxygen significantly enhances radiosensitivity (OER).
- Law of Bergonie & Tribondeau: Actively dividing, undifferentiated cells are more sensitive.
- The 4 R's of Radiobiology: Repair, Repopulation, Redistribution, and Reoxygenation.
- Apoptosis & mitotic catastrophe: key mechanisms of radiation-induced cell death.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

