Gas Laws & Pressure - Dive Deep Basics
- Pressure Basics: 1 ATA at sea level. ↑ 1 ATA per 10m (33ft) seawater depth.
⭐ At sea level, atmospheric pressure is 1 ATA; this pressure doubles to 2 ATA at just 10 meters (33 feet) of seawater depth.
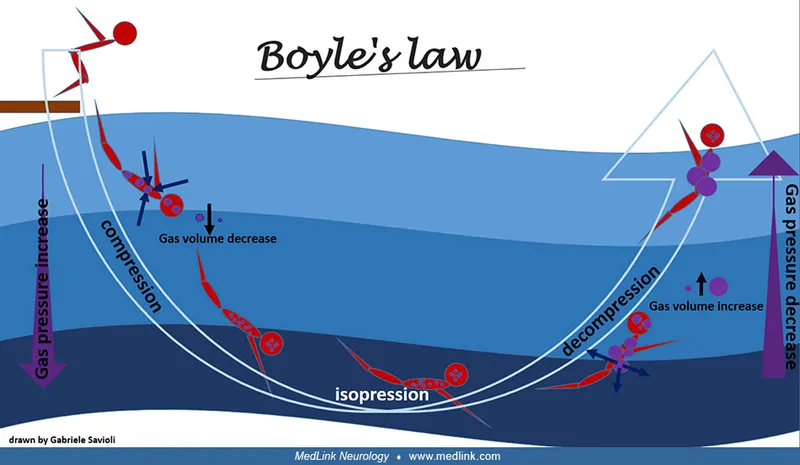
- Boyle's Law: $P_1V_1 = P_2V_2$. Gas volume $\propto$ 1/Pressure (temp constant).
- Key for barotrauma (lungs, ears, sinuses). 📌 Boyle's = Barotrauma.
- Dalton's Law: $P_{total} = \sum P_{partial}$.
- Basis of gas toxicities (N₂ narcosis, O₂ toxicity). 📌 Dalton's Diverse Pressures.
- Henry's Law: Dissolved gas amount $\propto$ its partial pressure ($C = kP_{gas}$).
- Basis of Decompression Sickness (DCS). 📌 Henry's = Helps dissolve.
- Basis of Decompression Sickness (DCS). 📌 Henry's = Helps dissolve.
Dive Reflex & Responses - Aquatic Adaptations
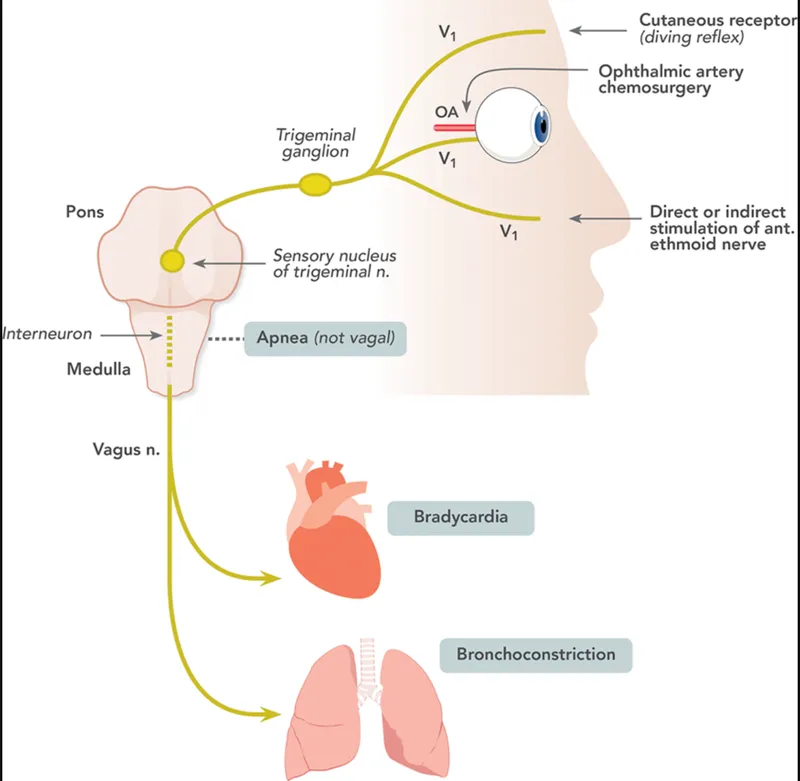
⭐ The mammalian diving reflex, crucial for oxygen conservation, involves the triad of apnea, bradycardia, and peripheral vasoconstriction.
- Mammalian Diving Reflex (MDR):
- Trigger: Cold water on face/nostrils (activates trigeminal nerve receptors).
- Purpose: Conserve $O_2$; protect vital organs (brain, heart).
- Key Responses (The Triad):
- Apnea: Cessation of breathing.
- Bradycardia: Profound slowing of heart rate (↓HR).
- Peripheral Vasoconstriction: Blood shunted from periphery to core circulation.
- Physiological Pathway:
- Other Adaptations:
- Splenic contraction: Autotransfusion of RBCs, ↑ $O_2$ carrying capacity.
- Blood shift: Central pooling of blood to protect thoracic organs from collapse at depth.
Barotrauma & Gas Toxicity - Pressure's Perils
-
Barotrauma: Tissue injury from pressure imbalance between gas-filled body spaces & environment (Boyle's Law: $P_1V_1 = P_2V_2$).
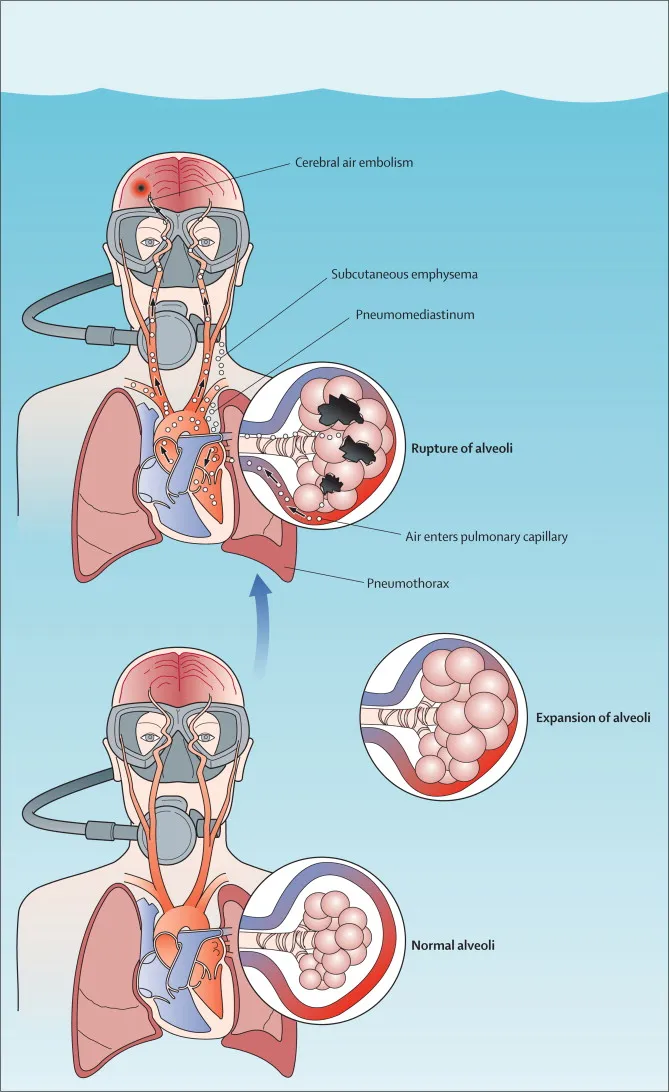
- Ascent (Gas Expansion):
- Pulmonary Barotrauma (PBT): Most dangerous. Breath-holding → alveolar rupture → Arterial Gas Embolism (AGE), pneumothorax.
- GI tract, dental.
- Descent (Gas Compression - "Squeeze"): Middle ear (most common), sinus, mask, tooth.
⭐ Pulmonary barotrauma of ascent (e.g., arterial gas embolism) is the most dangerous form of barotrauma and can occur from holding breath during ascent from even shallow depths.
- Ascent (Gas Expansion):
-
Gas Toxicity: Due to ↑ partial pressures with depth.
- Nitrogen Narcosis ("Rapture of the deep"):
- Impaired neuro-function (📌 Martini's Law: like 1 martini per 10m depth beyond 30m).
- Usually significant at depths >30-40m.
- Oxygen Toxicity:
- CNS: Seizures, visual disturbances. Risk ↑ if $P_{O2}$ > 1.4-1.6 ATA.
- Pulmonary (Lorrain Smith effect): Cough, dyspnea with prolonged exposure $P_{O2}$ > 0.5 ATA.
- CO2 Toxicity (Hypercapnia):
- Causes: Skip breathing, equipment malfunction.
- Symptoms: Headache, dyspnea, confusion.
- Nitrogen Narcosis ("Rapture of the deep"):
Decompression Sickness - Bubble Trouble
- Aka: Caisson disease, Bends.
- Pathophysiology: Rapid ascent → ↓ ambient pressure. Dissolved N₂ (from depth, Henry's Law) forms bubbles in blood/tissues.
- Risk factors: Deep/long dives, rapid ascent, cold, obesity, Patent Foramen Ovale (PFO).

- Types:
- Type I (Mild): "Bends" (joint pain), skin (mottling, itching), lymphatic obstruction.
- Type II (Serious): Neurological (spinal cord, brain - "staggers", paralysis), cardiopulmonary ("chokes" - dyspnea, chest pain), shock.
- Dysbaric Osteonecrosis (chronic bone necrosis).
- Prevention: Slow ascent, adhere to dive tables/computers, safety stops.
⭐ The definitive treatment for Decompression Sickness (DCS) is recompression therapy in a hyperbaric chamber, along with 100% oxygen.
High‑Yield Points - ⚡ Biggest Takeaways
- Boyle's Law: Inverse P-V relationship; causes barotrauma (ear/lung squeeze).
- Henry's Law: ↑Pressure → ↑dissolved gas; underlies Nitrogen Narcosis & DCS.
- Nitrogen Narcosis: Euphoria/impaired judgment at depth (>30m).
- Decompression Sickness (DCS): N₂ bubbles from rapid ascent; treat: recompression.
- Oxygen Toxicity: CNS (seizures)/pulmonary damage from high PO₂ (>1.4 ATA).
- Shallow Water Blackout: Pre-dive hyperventilation → ↓PaCO₂ → hypoxic LOC.
- Diving Reflex: Bradycardia, peripheral vasoconstriction, blood shift to conserve O₂.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

