Pancreatic Islet Hormones - Islet's Endocrine Symphony
Endocrine pancreas: Islets of Langerhans. 📌 Cells: Beta (Insulin), Alpha (Glucagon), Delta (Somatostatin).
- α-cells (Glucagon): ↑ Blood glucose (glycogenolysis, gluconeogenesis).
- β-cells (Insulin, Amylin): ~70% of islet cells. Insulin: ↓ Blood glucose. Amylin: ↓ Glucagon.
- Proinsulin → Insulin + C-peptide (equimolar).
- δ-cells (Somatostatin): Paracrine inhibition of insulin & glucagon.
- PP-cells (Pancreatic Polypeptide): Regulates pancreatic exocrine/endocrine functions.
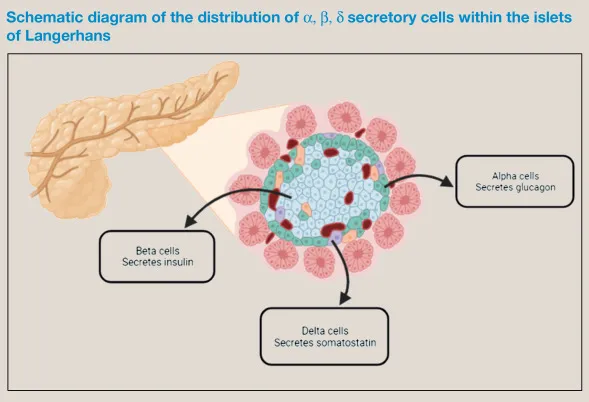
⭐ C-peptide measurement is crucial for assessing endogenous insulin production, especially in diabetic patients on insulin therapy.
Insulin Physiology - Insulin: The Key Master
- Structure: Peptide hormone; A (21 amino acids) & B (30 amino acids) chains, linked by disulfide bonds.
- Synthesis: Pancreatic β-cells: Preproinsulin → Proinsulin (Golgi) → Insulin + C-peptide (secretory granules).
- 📌 C-peptide: Equimolar secretion; reliable marker of endogenous insulin production.
- Secretion Stimuli:
- Primary: ↑ Blood glucose (via GLUT2 on β-cells → ↑ATP → KATP channel closure → Ca2+ influx → exocytosis).
- Others: Amino acids (arginine, leucine), incretins (GLP-1, GIP), vagal stimulation (ACh), β2-agonists.
- Secretion Inhibitors: Somatostatin, sympathetic stimulation (α2-adrenergic), diazoxide, propranolol.
- Receptor: Tyrosine kinase receptor (2α, 2β subunits). Binding → autophosphorylation → IRS activation → PI3K & MAPK pathways.
- Key Actions (Anabolic Hormone):
- ↓ Blood glucose: ↑ GLUT4 translocation (muscle, adipose), ↑ glycogenesis, ↓ gluconeogenesis, ↓ glycogenolysis.
- ↑ Lipogenesis (activates LPL, acetyl-CoA carboxylase), ↓ lipolysis (inhibits HSL).
- ↑ Protein synthesis, ↓ proteolysis.
- ↑ Cellular K+ uptake (stimulates Na+/K+-ATPase).
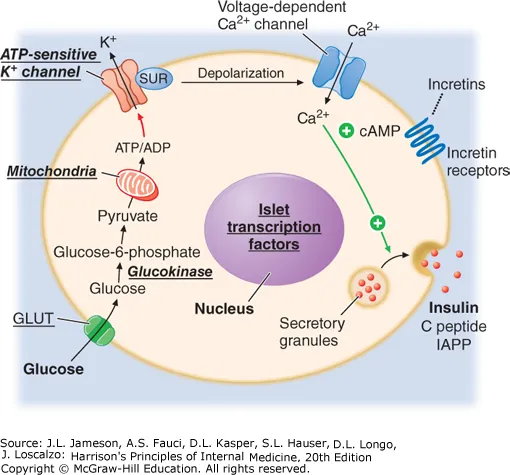
⭐ Insulin promotes K+ entry into cells by stimulating Na+/K+-ATPase activity; this is crucial in managing DKA, as insulin therapy can cause hypokalemia.
Glucagon Physiology - Glucagon: Glucose Lifeguard
- Source: Peptide, pancreatic α-cells.
- Function: ↑ Blood glucose; counter-regulatory to insulin. (📌 Mnemonic: Glucagon when "Glucose-is-GONE")
- Stimuli:
- Hypoglycemia (primary)
- Amino acids (arginine, alanine; post-protein meal)
- Sympathetic (β-adrenergic), Cortisol, CCK
- Inhibitors:
- Hyperglycemia, Insulin
- Somatostatin, GLP-1, fatty acids
- Mechanism (Liver): Gs-coupled receptor → ↑adenylyl cyclase → ↑cAMP → PKA.
- Hepatic Effects:
- ↑ Glycogenolysis (major, rapid glucose output)
- ↑ Gluconeogenesis (sustained glucose production)
- ↑ Ketogenesis (from fatty acid breakdown)
- ↑ Urea cycle activity
- Adipose Tissue: ↑ Lipolysis (HSL activation, generally minor in humans).
⭐ Glucagon's primary, most rapid effect is stimulating hepatic glycogenolysis to elevate blood glucose.
Glucose Regulation & Imbalance - Sweet Balance & Chaos
- Core Balance: Maintained by insulin (↓ blood glucose) & glucagon (↑ blood glucose).
- Normal Fasting Glucose: 70-100 mg/dL.
- Post-Prandial (2hr): <140 mg/dL.
- Insulin (Anabolic): From β-cells.
- ↑ Glucose uptake (GLUT4: muscle, adipose).
- ↑ Glycogenesis, ↑ Lipogenesis.
- ↓ Gluconeogenesis, ↓ Glycogenolysis.
- Glucagon (Catabolic): From α-cells.
- ↑ Glycogenolysis (liver).
- ↑ Gluconeogenesis (liver).
- Flow of Control:
- Imbalance States:
- Diabetes Mellitus (DM): Persistent hyperglycemia.
- Diagnosis: FPG ≥126 mg/dL; PPG (2hr) ≥200 mg/dL; HbA1c ≥6.5%.
- Hypoglycemia: Blood glucose <70 mg/dL.
- Whipple's Triad: Symptoms + Low BG + Relief with glucose.
- Diabetes Mellitus (DM): Persistent hyperglycemia.
⭐ In Diabetic Ketoacidosis (DKA), total body potassium is depleted despite potential normal/high serum K+ due to extracellular shift from acidosis & insulinopenia.
High‑Yield Points - ⚡ Biggest Takeaways
- Insulin (β-cells): anabolic, ↑ glucose uptake via GLUT4 (muscle/adipose). Deficiency → DM Type 1.
- Glucagon (α-cells): catabolic, ↑ glycogenolysis & gluconeogenesis.
- Somatostatin (δ-cells): inhibits insulin & glucagon secretion.
- GLUT2: insulin-independent (β-cells, liver); GLUT4: insulin-dependent (muscle, adipose).
- HbA1c: reflects 2-3 month glucose control; target <7%.
- DKA (T1DM): hyperglycemia, ketosis, acidosis.
- HHS (T2DM): severe hyperglycemia, hyperosmolality, minimal/no ketosis_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

