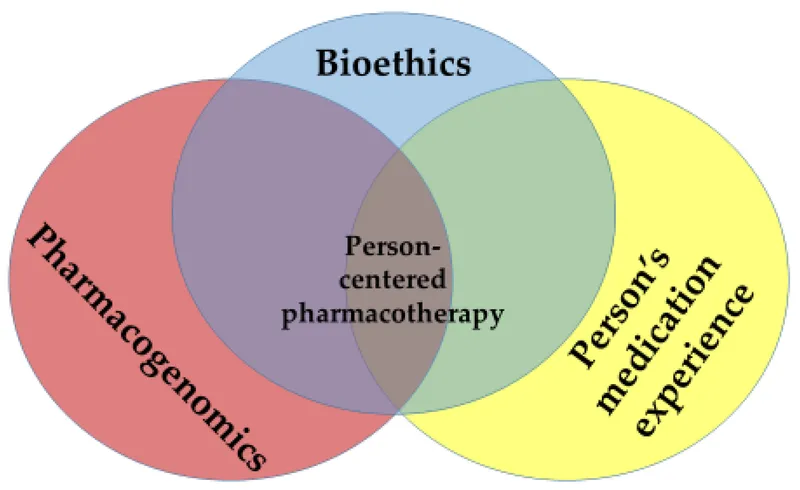
Ethical Foundations - PGx Moral Compass
- Core Ethical Principles in PGx:
- Autonomy: Patient's right to informed consent for PGx testing.
- Beneficence: Maximize benefits; PGx tests should improve treatment outcomes.
- Non-maleficence: "Do no harm"; prevent misuse of genetic data, stigmatization.
- Justice: Ensure equitable access to PGx testing and its advantages.
- Indian Regulatory Context:
- Guided by ICMR National Ethical Guidelines for Biomedical and Health Research Involving Human Participants (2017).
- Key Concepts:
- Genetic Determinism: Misconception that genes solely dictate drug response.
- Genetic Predisposition: Genes indicate likelihood of a response, not certainty.
⭐ ICMR (2017) Guidelines, Section 9.5, mandates pre- and post-test genetic counselling for all genetic testing, including pharmacogenomics.
Consent & Counseling - Genes & Choices
Ensuring patient autonomy and understanding in pharmacogenomic testing.
- Informed Consent Elements: 📌 Patients Really Benefit Learning About Choices Wisely.
- Purpose of test
- Risks & Benefits
- Limitations of test
- Alternatives
- Confidentiality
- Right to Withdraw
- Genetic Counseling Process:
- Key Challenges:
- Low genetic literacy.
- Managing incidental findings (IFs).
- Familial implications (duty to inform vs. confidentiality).
- Vulnerable Populations (India):
- Rural, tribal communities.
- Individuals with low literacy/socioeconomic status.
- Require culturally sensitive counseling.
⭐ A common ethical dilemma: Balancing patient autonomy with the potential duty to warn family members about shared genetic risks revealed through pharmacogenomic testing, especially if the patient refuses disclosure.
Data Dilemmas - Privacy & Protection
- Genetic Privacy & Confidentiality: Essential to protect sensitive genetic information. Informed consent is key.
- Data Security: Implement encryption, access controls, and secure storage to prevent unauthorized access or breaches.
- Key Risks:
- Genetic Discrimination: Unfair treatment in employment or insurance based on genetic predispositions.
- Stigmatization: Social or personal prejudice linked to genetic traits.
- Research Data Management:
- Ownership: Complex; requires clear institutional policies.
- Sharing: Utilize anonymization or de-identification to safeguard patient identity.
- Indian Legal Framework:
- Digital Personal Data Protection Act (DPDPA), 2023: Governs processing of personal data, including sensitive genetic data.
⭐ The DPDPA, 2023, mandates explicit consent for processing sensitive personal data (like genetic data), crucial for preventing its misuse, e.g., in employment or insurance decisions.
Access & Equity - Fair PGx for All
- Goal: Ensure equitable access to pharmacogenomic (PGx) testing and subsequent personalized medicines for all population segments.
- Cost-Effectiveness: Critical evaluation needed for integrating PGx into the resource-constrained Indian public healthcare system.
- Resource Allocation: Significant challenge in distributing limited healthcare funds and infrastructure for advanced PGx services.
- Addressing Disparities:
- Bridging the urban-rural gap in availability and awareness of PGx tests.
- Mitigating impact of socio-economic status on access to personalized medicine.
- Benefit Sharing: Ethical imperative to ensure communities involved in genomic research receive fair benefits from findings.
⭐ The Ayushman Bharat Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) aims to improve healthcare access for vulnerable populations, potentially facilitating PGx test integration in the future.
High‑Yield Points - ⚡ Biggest Takeaways
- Informed consent is mandatory for pharmacogenomic testing, ensuring patient comprehension.
- Patient privacy and data confidentiality of genetic information are paramount.
- Preventing genetic discrimination in employment or insurance is a key concern.
- Ensuring equitable access to pharmacogenomic tests and treatments is vital.
- Genetic counseling is essential for interpreting results and patient understanding.
- Managing incidental findings from genetic tests poses ethical challenges.
- Protecting vulnerable populations in pharmacogenomic research is crucial.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

