Special Populations Overview - Not One Size Fits All
- Special Populations: Groups (e.g., elderly, pediatrics, pregnant, organ impairment) with distinct physiological or pathological traits altering drug response.
- Pharmacokinetic (PK) Variations: Altered Absorption, Distribution, Metabolism (e.g., ↓ CYP450 activity), and Excretion (e.g., ↓ GFR).
- Pharmacodynamic (PD) Variations: Changes in receptor sensitivity, number, or post-receptor events.
- Individualized Therapy: Crucial to tailor dosage, ensuring efficacy and minimizing adverse drug reactions (ADRs) due to ↑ variability.
⭐ Key factors influencing inter-individual variability in drug response include genetics (pharmacogenomics), age, organ function (renal/hepatic), disease state, and concomitant drug use (drug-drug interactions).
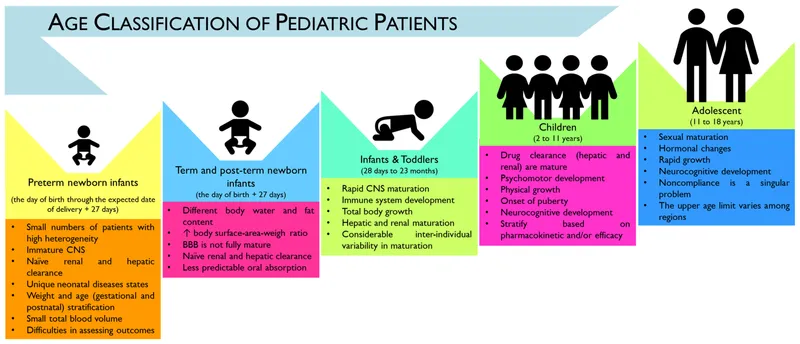
Pediatric Pharmacology - Tiny Patients, Big Risks
- Pharmacokinetic (PK) Changes:
- Absorption: ↑ Gastric pH (neonate), variable emptying.
- Distribution: ↑ Total Body Water, ↓ Plasma protein binding (esp. neonates).
- Metabolism: Immature enzymes (e.g., glucuronidation ↓ in neonates).
- Excretion: ↓ Glomerular Filtration Rate (GFR) (doubles by 1 week, adult levels by 6-12 months).
- Pharmacodynamic (PD) Differences: Altered receptor sensitivity, paradoxical reactions (e.g., benzodiazepines).
- Key Problematic Drugs & Risks: 📌 C.A.S.T.F.
- Chloramphenicol: Gray Baby Syndrome.
- Aspirin: Reye's Syndrome.
- Sulfonamides: Kernicterus (neonates).
- Tetracyclines: Teeth/bone issues.
- Fluoroquinolones: Cartilage damage.
- Dosing Considerations: Typically $mg/kg$ or Body Surface Area ($BSA$).
⭐ Gray Baby Syndrome: Caused by chloramphenicol accumulation due to deficient glucuronidation in neonates. Presents with vomiting, flaccidity, hypothermia, gray color, shock, and cardiovascular collapse.
Geriatric Pharmacology - Elderly & Polypharmacy Perils
- PK Changes: ↓Renal clearance (Est. by Cockcroft-Gault: $CrCl = \frac{(140 - age) \times Wt (kg)}{72 \times SCr (mg/dL)} (\times 0.85 \text{ if female})$), ↓hepatic metabolism (Phase I), ↑Vd (lipid-soluble drugs due to ↑body fat), ↓Vd (water-soluble drugs due to ↓body water), ↓serum albumin.
- PD Changes: ↑Sensitivity to drugs (e.g., benzodiazepines, opioids, antipsychotics), impaired homeostatic mechanisms (e.g., ↑risk of orthostatic hypotension, altered thermoregulation).
- Polypharmacy: Concurrent use of ≥5 drugs. Risks: ↑Adverse Drug Reactions (ADRs), drug interactions, non-adherence, prescribing cascade.
- Beers Criteria / STOPP-START: Tools to identify Potentially Inappropriate Medications (PIMs) & guide safer prescribing.
- Key PIMs: Strong anticholinergics, long-acting benzodiazepines, certain NSAIDs, sliding scale insulin.
- 📌 Anticholinergic side effects: "Can't see, can't pee, can't spit, can't shit."
- Common Problematic Interactions:
- Warfarin + NSAIDs (↑bleeding risk).
- ACE Inhibitors + K-sparing diuretics (↑hyperkalemia risk).
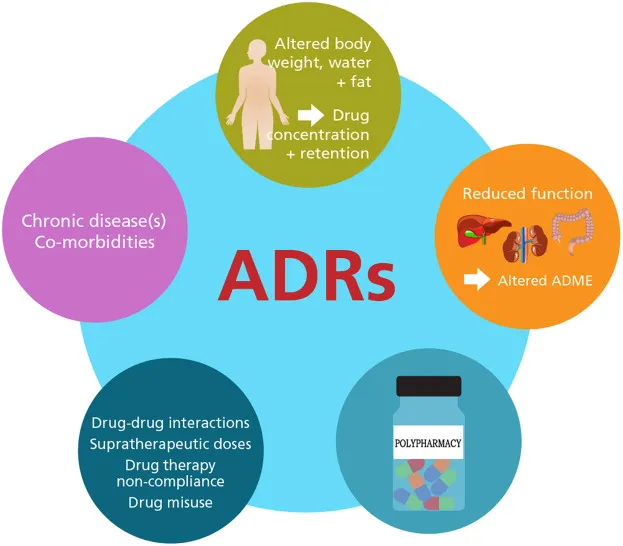
⭐ Benzodiazepines and anticholinergic drugs are frequently implicated in preventable adverse drug events and hospitalizations in the elderly population due to increased sensitivity and accumulation.
Pregnancy & Lactation - Maternal Meds, Safe Steps
- PK Changes (Pregnancy): ↑Vd, ↑GFR, ↑metabolism (some drugs), ↓plasma proteins. N/V → altered absorption.
- Placental Transfer: Factors: lipid solubility, MW, protein binding, transporters.
- Teratogenicity: Fetal harm. Critical period: organogenesis (1st trimester).
- Major Teratogens (📌 Mnemonic: TWA VIM):
- Thalidomide: Phocomelia
- Valproate: NTDs
- Warfarin: Nasal hypoplasia
- ACEi: Renal dysgenesis
- Isotretinoin: CNS/craniofacial defects
- Methotrexate: CNS/limb defects
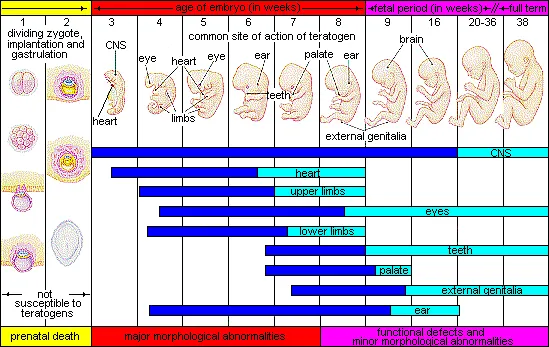
- Major Teratogens (📌 Mnemonic: TWA VIM):
- Labeling: Old FDA (A,B,C,D,X). New: PLLR (descriptive risk summary).
- Lactation: Excretion factors: M/P ratio, pKa, lipid solubility. Resource: LactMed.
- Avoid/caution: Lithium, amiodarone, cytotoxic drugs.
⭐ Warfarin exposure during 6th-9th week gestation can cause nasal hypoplasia & chondrodysplasia punctata.
Organ Impairment - System SOS, Dose Adjust
- Renal Impairment: Impacts drug excretion. Adjust dose (↓dose, ↑interval) based on CrCl/eGFR.
- Dose adjust: Aminoglycosides, Digoxin, Lithium, Gabapentin.
- Nephrotoxic (avoid/monitor): NSAIDs, Contrast media, Amphotericin B.
- Hepatic Impairment: Impacts drug metabolism. Assess with Child-Pugh score (A, B, C).
- Caution: Paracetamol (high doses), Statins (active/decompensated disease).
⭐ Metformin is contraindicated if eGFR < 30 mL/min/1.73m².
High‑Yield Points - ⚡ Biggest Takeaways
- Pediatrics: Dose by weight/BSA; immature organ function (hepatic/renal) alters drug PK.
- Geriatrics: High risk from polypharmacy & ↓ renal clearance; increased drug sensitivity.
- Pregnancy: Avoid teratogens; drugs cross placenta; physiological PK changes occur.
- Lactation: Assess drug excretion in breast milk & potential infant risk.
- Renal Impairment: Dose adjustment crucial for drugs with significant renal excretion.
- Hepatic Impairment: Reduced drug metabolism & altered protein binding; monitor closely.
- Genetic Polymorphisms: Impact drug metabolizing enzymes (e.g., CYPs), causing varied responses.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

