Microbial Biodegradation - Tiny Titans Tackle Toxins
- Process: Microbes enzymatically transform/degrade organic pollutants. Central to bioremediation & nutrient cycling.
- Key Microbes:
- Bacteria: Pseudomonas (versatile, e.g., P. putida), Bacillus, Rhodococcus.
- Fungi: White-rot fungi (e.g., Phanerochaete chrysosporium - ligninolytic enzymes), Aspergillus.
- Pathways:
- Aerobic: $O_2$ as terminal electron acceptor. Efficient. $Organic Pollutant + O_2 \rightarrow CO_2 + H_2O + Biomass$.
- Anaerobic: Alternative electron acceptors ($NO_3^-$, $SO_4^{2-}$, $Fe^{3+}$). Slower, for recalcitrant compounds.
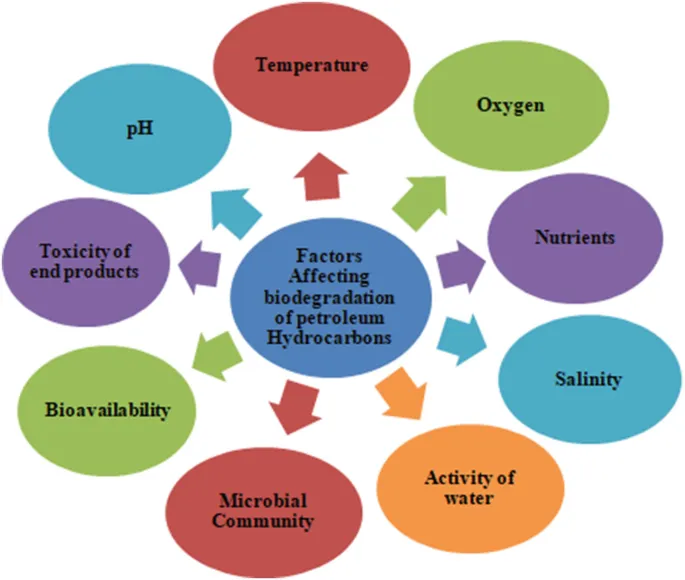
- Influencing Factors: Pollutant bioavailability & structure, microbial consortia, temperature, pH, oxygen, nutrients (C:N:P ratio).
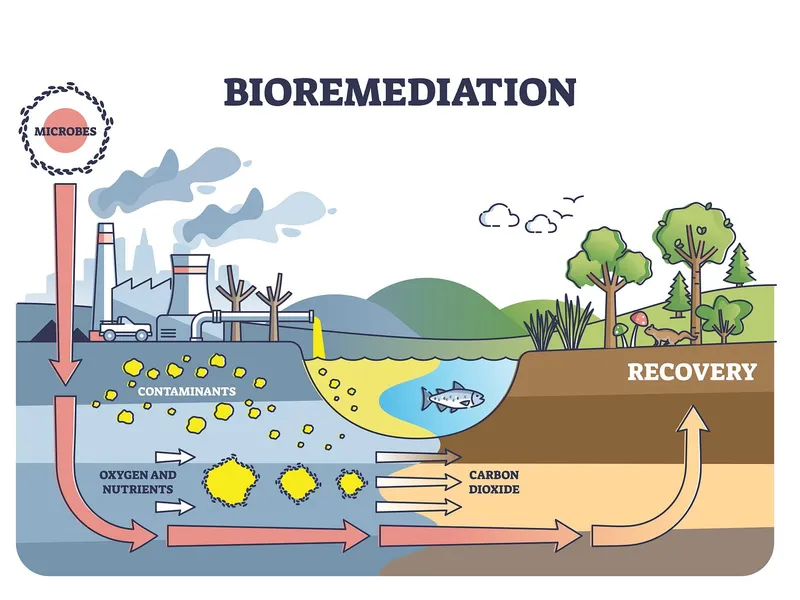
⭐ Cometabolism: Microbes degrade a pollutant they cannot use as a primary carbon or energy source, requiring the presence of another growth-supporting substrate (co-substrate).
Microbial Biodegradation - What Makes Microbes Munch?
| Factor Category | Key Factors | Impact on Rate |
|---|---|---|
| Environmental | Temp, pH, O₂, Moisture, Nutrients (C:N ratio, e.g., 10:1-30:1) | Optimal ranges ↑ |
| Substrate | Structure (complexity↓), Conc., Bioavailability (↑) | Simpler, available ↑ |
| Microbial | Degraders, Enzymes, Acclimatization | Specificity, adaptation ↑ |
- Cometabolism: Degradation of non-growth substrate with a growth substrate.
- Recalcitrance: Resistance to breakdown (e.g., xenobiotics like plastics).
- Oxygen:
- Aerobic: Faster (e.g., hydrocarbons).
- Anaerobic: Slower (e.g., reductive dehalogenation of chlorinated solvents).
⭐ Bioaugmentation (add microbes) & Biostimulation (add nutrients) boost in-situ biodegradation.
Microbial Biodegradation - How Microbes Demolish Dregs
Microbes enzymatically transform complex organic pollutants into simpler, less toxic substances. This process is central to natural attenuation and engineered bioremediation.
- Aerobic Degradation:
- Requires $O_2$ as the terminal electron acceptor.
- Key Enzymes: Oxygenases (monooxygenases, dioxygenases) incorporate oxygen atoms into the pollutant molecule, initiating breakdown; peroxidases.
- Outcome: Often leads to complete mineralization (e.g., to $CO_2$, $H_2O$, and biomass). Example: Hydrocarbon degradation by Pseudomonas.
- Anaerobic Degradation:
- Occurs in the absence of $O_2$.
- Alternative electron acceptors: $NO_3^-$ (denitrification), $SO_4^{2-}$ (sulfate reduction), $Fe^{3+}$, $CO_2$ (methanogenesis).
- Key Enzymes: Reductases, dehalogenases, hydrolases.
- Outcome: Generally slower; may result in partial degradation or biotransformation. Example: Reductive dehalogenation of trichloroethylene (TCE).
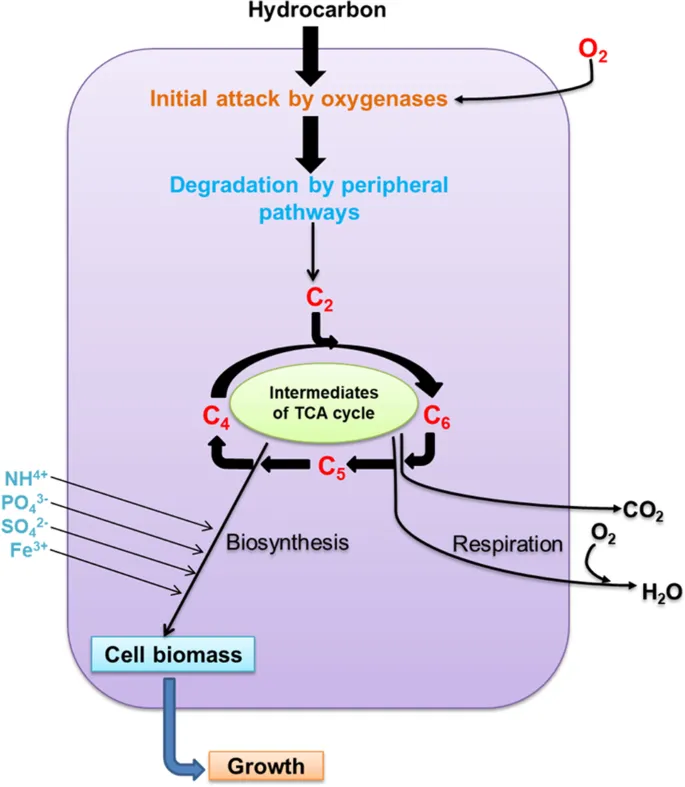
- Key Concepts:
- Mineralization: Complete breakdown of organic pollutants to inorganic substances like $CO_2$, $H_2O$, and mineral salts.
- Cometabolism: Fortuitous degradation of a substance by microbial enzymes produced for other substrates, without providing energy or nutrients to the microbe.
⭐ Pseudomonas putida is a classic example of a bacterium used in bioremediation due to its diverse metabolic pathways for degrading aromatic hydrocarbons (e.g., toluene).
Microbial Biodegradation - Pollutant Purge Pros
Microbial transformation of pollutants into less toxic or inert substances, cleaning the environment.
- Key Players:
- Bacteria: Pseudomonas, Bacillus, Alcaligenes, Rhodococcus.
- Fungi: Phanerochaete chrysosporium (white-rot), Aspergillus, Penicillium.
- Mechanisms:
- Aerobic: $O_2$ as terminal electron acceptor. Leads to complete degradation (mineralization) e.g., $Pollutant + O_2 \rightarrow CO_2 + H_2O + Biomass$. Faster.
- Anaerobic: $NO_3^-$, $SO_4^{2-}$, $Fe^{3+}$, $CO_2$ as electron acceptors. Slower, crucial for chlorinated compounds (e.g., PCE, TCE).
- Factors Influencing: Pollutant structure & concentration, microbial population (presence of degraders), oxygen, pH, temperature, nutrients. 📌 Contaminant, Environment, Microbes (CEMent pollutants' fate).
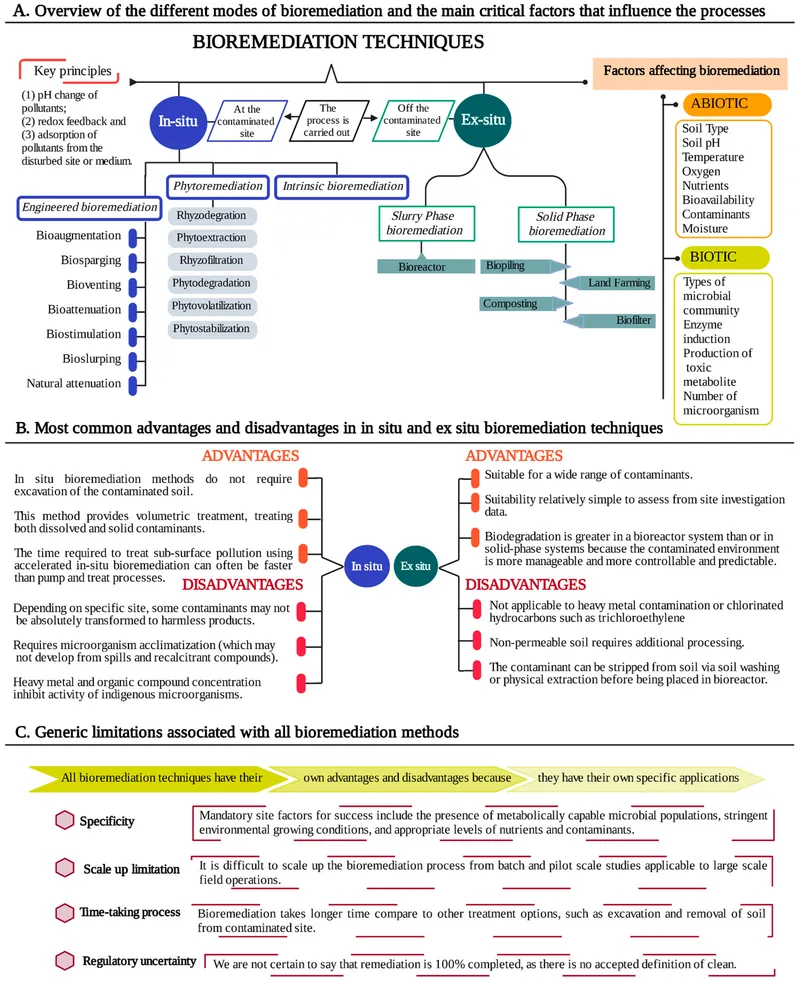
- Bioremediation Strategies:
- In-situ: Treatment at the contaminated site (e.g., bioventing, biosparging, bioaugmentation).
- Ex-situ: Removal of contaminated material for treatment (e.g., landfarming, biopiles, bioreactors).
- Xenobiotics: Synthetic compounds resistant to degradation (e.g., plastics, pesticides, PCBs). Often require:
- Co-metabolism: Gratuitous degradation by enzymes for other substrates.
⭐ Pseudomonas putida strains are renowned for degrading diverse aromatic hydrocarbons (e.g., toluene, naphthalene) and were subjects of early genetic engineering for enhanced bioremediation capabilities (e.g., "superbug" for oil spills).
High‑Yield Points - ⚡ Biggest Takeaways
- Microbial metabolism is central to pollutant degradation.
- Xenobiotics are synthetic compounds often recalcitrant to biodegradation.
- Cometabolism involves degrading a non-growth substrate with a growth substrate.
- Bioaugmentation introduces specific microbes; biostimulation enhances native microbial activity.
- Aerobic degradation, often involving oxygenases, is typically faster than anaerobic degradation, which may use reductases.
- Key factors: pollutant structure, microbial consortia, bioavailability, environmental conditions (pH, temperature, O₂).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

