Overview & Ideals - Blueprinting Immunity
- Vaccine: Agent providing active acquired immunity against a specific disease.
- Immunity:
- Active: Own antibodies produced (post-vaccine/infection).
- Passive: Ready-made antibodies transferred (maternal/antitoxin).
- Herd Immunity: Indirect protection for unvaccinated when a high proportion of population is immune.
- Goals: Disease prevention, control, eradication; individual & community protection.
- Ideal Vaccine (📌 SHE IS CHeap):
- Safe (no/minimal harm)
- Humoral & Cellular immunity (strong response)
- Effective (high protection)
- Inexpensive (affordable)
- Stable (long shelf-life)
- Convenient (single dose, easy admin)
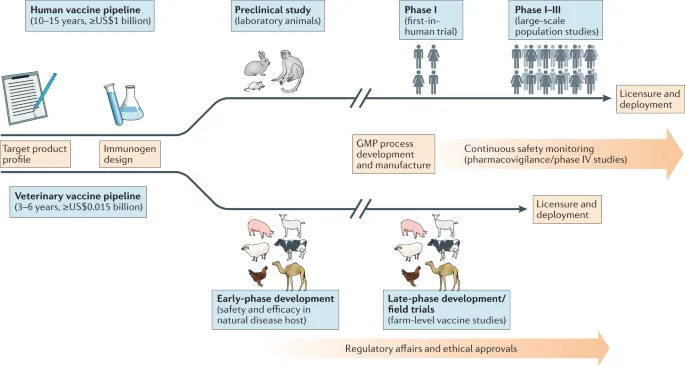
- Development Pipeline: Exploratory → Pre-clinical → Clinical Trials (I-III) → Regulatory Review → Manufacturing → Post-marketing Surveillance (IV).
⭐ Adjuvants (e.g., Alum) are added to vaccines to boost immunogenicity by enhancing the host immune response.
Clinical Trial Phases - Testing the Shield
Vaccines undergo rigorous testing, starting with:
- Pre-clinical Studies: In vitro assays and in vivo animal models establish initial safety profiles, immunogenicity, and potential efficacy through dose-ranging and challenge studies.
Clinical trials then proceed in phases:
| Phase | Primary Goal | Participants | Subject Type |
|---|---|---|---|
| Phase I | Assess safety, tolerability, pharmacokinetics (PK), find safe dose range. | 20-100 | Healthy volunteers |
| Phase II | Evaluate initial efficacy, further safety, confirm optimal dose. | 100-500 | Target population |
| Phase III | Confirm definitive efficacy in large diverse group, monitor safety. Pivotal trials. | 1000s | Target population |
| Phase IV | Post-marketing: long-term efficacy, rare adverse events (AEs), AEFI monitoring. | Population-wide | Approved users |
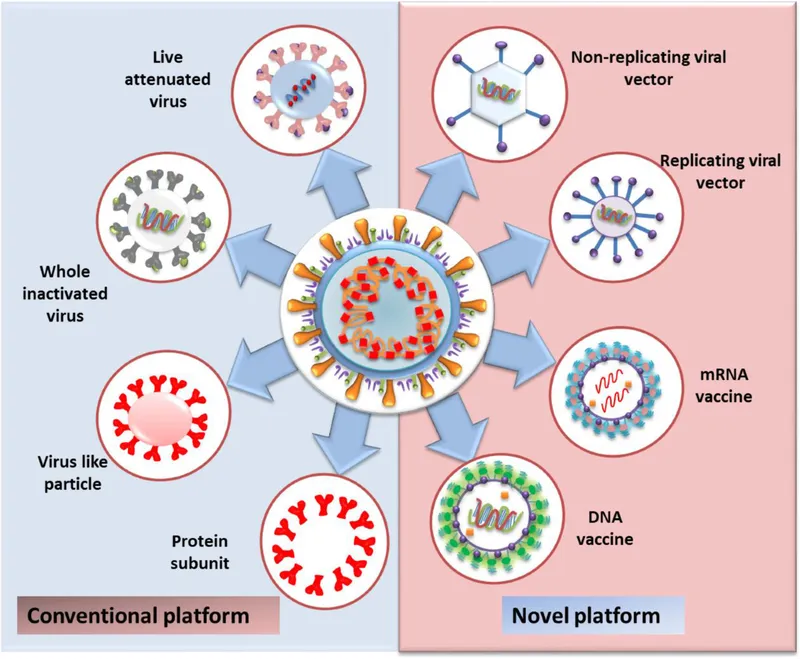
Vaccine Types & Platforms - Crafting the Armor
Vaccines use varied antigen forms. Production: attenuation (live), inactivation (killed), recombinant tech (subunit/mRNA). 📌 Romance In Brazil Makes My Tummy Yellow (Rotavirus, Influenza-nasal, BCG, MMR, Typhoid-oral, Yellow Fever).
- Live-attenuated: Weakened. BCG, MMR, OPV, Rotavirus, Varicella. Strong immunity (IgA, CMI). Risk: Reversion, CI in immunocompromised.
- Inactivated: Killed. IPV, Rabies, Hep A, Covaxin, wP; Influenza (fractional). Safe. Weaker immunity, boosters.
- Subunit: Specific antigens.
- Protein: Hep B, aP. Polysaccharide: PPSV23, Typhoid Vi. Conjugate: Hib, PCV13, Meningococcal.
- Fewer side effects. Adjuvants often needed.
- Toxoid: Inactivated toxins. Tetanus, Diphtheria. Targets toxin.
- Newer:
- Viral Vector (harmless virus): Covishield, Sputnik V.
- mRNA (genetic code in LNP): Pfizer, Moderna. DNA (conceptual).
- Rapid development. Storage issues (mRNA).
⭐ Conjugate vaccines convert T-independent polysaccharide antigens into T-dependent antigens, enabling immune response in infants <2 years and memory.
Quality & Regulation - Guardian Checkpoints
- Key Quality Control (QC) Tests:
- Identity: Confirms correct immunogen.
- Purity: Free from contaminants.
- Potency: Measures immunogenic strength/activity.
- Sterility: Absence of microbial contamination.
- Safety: General safety, pyrogenicity/endotoxin, abnormal toxicity tests.
- Stability Testing: Determines shelf-life under varied conditions.
- Good Manufacturing Practices (GMP): Ensure consistent production, quality, safety.
- Regulatory Oversight:
- National Regulatory Authorities (NRAs) e.g., CDSCO (India).
- WHO Prequalification (PQ) for UN procurement.
- Lot Consistency & Release: NRA reviews/tests each batch pre-market.
- AEFI Surveillance: Monitoring & reporting Adverse Events Following Immunization.
⭐ The critical importance of maintaining the 'cold chain' (temperature-controlled supply chain) for vaccine efficacy, especially for temperature-sensitive vaccines like live attenuated and mRNA vaccines.
High‑Yield Points - ⚡ Biggest Takeaways
- Clinical trials progress through Phase I (safety), II (efficacy/dosage), III (large-scale efficacy), and IV (post-marketing).
- Maintaining the cold chain (+2°C to +8°C) is essential for vaccine potency.
- Adjuvants (e.g., Alum) boost immunogenicity; preservatives (e.g., Thimerosal) ensure sterility.
- Killed vaccines use inactivation (formalin); live attenuated vaccines use attenuation (serial passage).
- Recombinant DNA technology produces vaccines like Hepatitis B (HBsAg).
- Good Manufacturing Practices (GMP) ensure vaccine quality and safety during production.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

