Basics & Lingo - Germ Warfare 101
- Disinfectant: Kills/inactivates microbes on inanimate objects.
- Antiseptic: Prevents/arrests microbial growth on living tissues.
- Sterilant: Kills all microbial life, including spores.
- Bacteriostatic: Inhibits bacterial growth (reversible).
- Bactericidal: Kills bacteria (irreversible).
⭐ Key difference: Antiseptics are used on living tissues, while disinfectants are for non-living surfaces/objects.
- Levels of Disinfection:
- High: Kills all microbes (not high numbers of bacterial spores).
- Intermediate: Kills M. tuberculosis, most viruses, fungi (not spores).
- Low: Kills most vegetative bacteria, some viruses/fungi (not TB, not spores).
- Ideal Properties: Broad spectrum, rapid action, non-toxic, non-corrosive, stable, affordable, good penetration, not inactivated by organic matter.
Chemical Classes - The Agent Arsenal
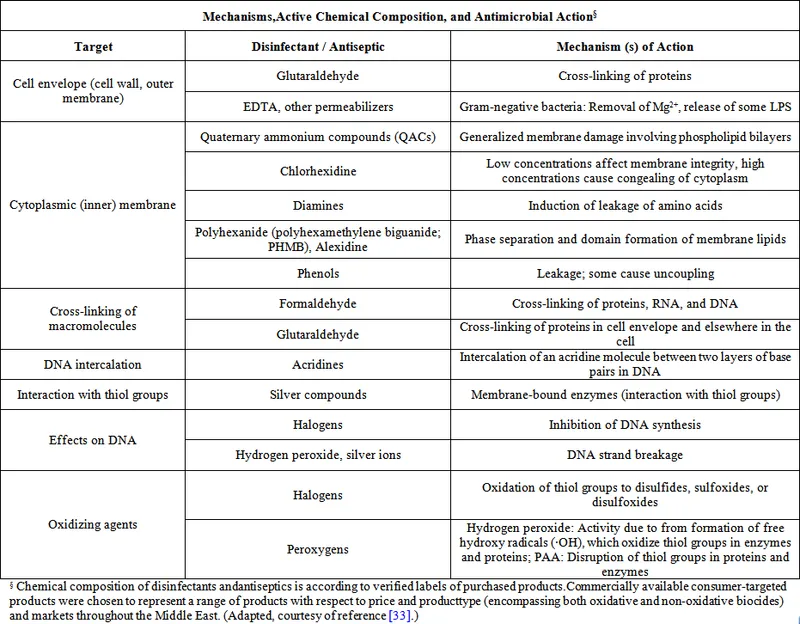
⭐ Aldehydes (e.g., Glutaraldehyde) sterilize.
| Class | Spectrum | MoA | E.g. | Uses | Adv / Disadv |
|---|---|---|---|---|---|
| Alcohols | B,F,V(env) | Denatures protein | Ethanol (70%), IPA | Skin, surface | Rapid / Volatile, no spores |
| Aldehydes | B,F,V,S (sterilant) | Alkylates prot/NA | Glutaraldehyde, Formalin | Sterilize, Hi-level D. | Sporicidal / Toxic, irritant |
| Halogens | B,F,V,S(some) | Oxidize | Iodine, Bleach | $H_2O$, surface, skin | Broad / Corrosive, inactivated |
| Phenolics | B,F,V(some) | Memb. damage, prot den | Phenol, Chloroxylenol | Surface, equip. | Stable / Irritant, toxic |
| QACs | B(G+),F,V(env) | Memb. disrupt | Benzalkonium Cl | Surface, skin | Non-corrosive / Soap-inact, no spores |
| Biguanides | B,F,V(some) | Memb. disrupt | Chlorhexidine | Skin, oral | Persistent / Ototoxic |
| Peroxygens | B,F,V,S(hi conc) | Oxidize (radicals) | $H_2O_2$, Peracetic acid | Wound, sterilize (PAA) | Broad, eco / Unstable, corrosive |
| Heavy Metals | B(some) | Inactivates SH- enzymes | $AgNO_3$, Hg cmpds | Eye, topical | Oligodynamic / Toxic, resistance |
| Dyes | B(G+),F(some) | DNA intercalate | Crystal violet | Skin, sel. media | Selective / Stains, narrow |
Action & Factors - Efficacy Equation
- Efficacy hinges on interacting factors. 📌 Mnemonic: CONTPHORM
- Concentration: ↑ conc. → ↑ efficacy (to optimum).
- Organic Matter (blood, pus): Significantly ↓ efficacy.
⭐ Organic matter greatly reduces efficacy of many disinfectants (e.g., halogens, QACs).
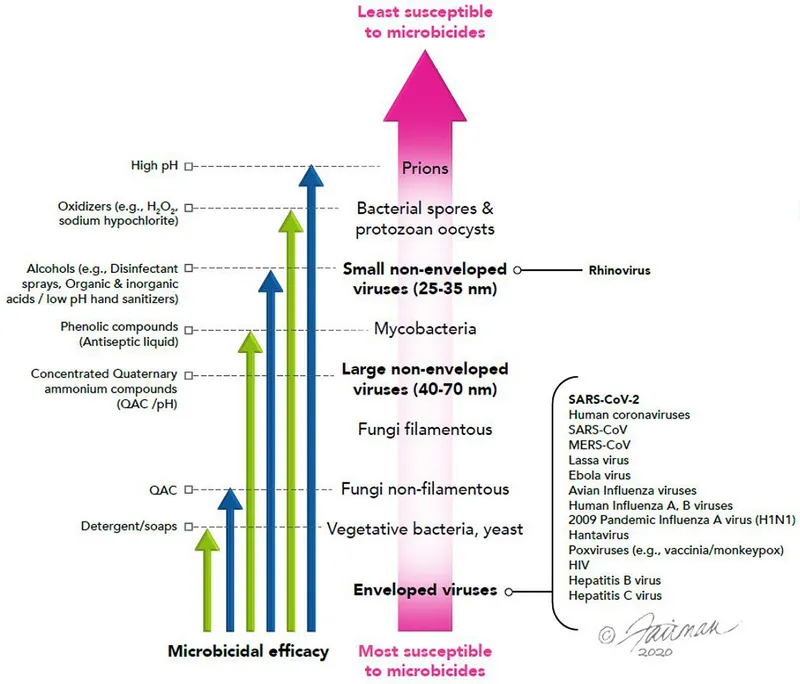
- Nature of Microorganism: Resistance hierarchy (Spores > Mycobacteria > Non-enveloped viruses > Fungi > Bacteria > Enveloped viruses).
- Time (Contact): Sufficient duration is critical.
- PH: Agent-specific optimal pH.
- H (Other Factors):
- Temperature: ↑ temp → ↑ rate (e.g., 10°C rise doubles rate).
- Number of Microbes (Bioburden): ↑ load → ↑ challenge.
- Biofilms: Protective, increase resistance.
Key Agents In-Depth - Clinical Hit List
| Agent | Uses & Conc. | Spectrum | Pros | Cons |
|---|---|---|---|---|
| Alcohol | Skin (70%), surfaces | Bacteria, Fungi, Enveloped Viruses | Rapid action, non-staining | Flammable, drying, not sporicidal |
| Chlorhexidine | Skin (0.5-4%), oral, catheter | Bacteria (G+/G-), some Viruses, Fungi | Good residual action, low irritation | Ototoxic (middle ear), rare allergy |
| Povidone-Iodine | Skin/wound (7.5-10%) | Broad (Bacteria, Viruses, Fungi, Spores) | Sustained iodine release | Stains, Iodism, thyroid issues |
| Glutaraldehyde | Instruments (HLD) (2%) | All microbes incl. Spores | Non-corrosive to most | Toxic fumes, irritant, needs activation |
| $H_2O_2$ | Wound (3%), HLD (>7.5%) | Broad; Sporicidal (high conc.) | $H_2O, O_2$ byproducts | Tissue damage (high conc.), unstable |
| $NaOCl$ | Surfaces (0.5-1%), water | Broad incl. Spores (high conc.) | Fast-acting, inexpensive | Corrosive, inactivated by organic matter |
High‑Yield Points - ⚡ Biggest Takeaways
- Alcohols (Ethanol, Isopropanol) denature proteins; not sporicidal.
- Aldehydes (Formaldehyde, Glutaraldehyde) are sporicidal; Glutaraldehyde for heat-sensitive instrument sterilization.
- Halogens: Iodine (antiseptic), Chlorine compounds (disinfectants, hypochlorite for blood spills).
- Phenols (Lysol, Dettol) disrupt cell membranes; effective against Mycobacteria.
- Quaternary Ammonium Compounds (QACs, Cetrimide) are cationic surfactants; inactivated by soaps.
- Biguanides (Chlorhexidine) disrupt cell membranes; good residual activity, skin antiseptic.
- Ethylene Oxide: sterilant gas for heat-labile items; carcinogenic, explosive.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

