Sterilization and Disinfection

On this page
🧬 The Microbial Battlefield: Sterilization and Disinfection Mastery
Preventing infection hinges on knowing exactly how to eliminate or reduce microbial threats on instruments, surfaces, and materials. You'll master the science behind heat, pressure, and chemical agents that destroy pathogens, then learn to match the right method to each clinical scenario-from surgical tools requiring absolute sterility to environmental surfaces needing practical disinfection. By integrating physical principles, chemical mechanisms, and evidence-based protocols, you'll build the clinical judgment to protect patients and prevent healthcare-associated infections with precision and confidence.
Sterilization and disinfection represent the critical defense mechanisms against healthcare-associated infections, which affect 1 in 25 hospitalized patients annually. Master these concepts, and you control the invisible battlefield where patient outcomes are determined before the first incision is made.
📌 Remember: STERILE - Steam under pressure, Temperature 121°C, Ethylene oxide gas, Radiation gamma rays, Instruments must be clean, Load configuration matters, Exposure time 15 minutes minimum
The hierarchy of microbial resistance forms the foundation for all sterilization protocols:
- Prions (Highest resistance)
- Creutzfeldt-Jakob disease agents
- Require 134°C for 18 minutes or 1M NaOH
- Resistance factor: 1000x greater than vegetative bacteria
- Bacterial Spores
- Bacillus stearothermophilus, Clostridium difficile
- Standard biological indicators for steam sterilization
- D-value: 1.5-2.0 minutes at 121°C
- Bacillus stearothermophilus: 2.0 minutes
- Bacillus subtilis: 1.8 minutes
- Mycobacteria
- M. tuberculosis, M. bovis
- Lipid-rich cell wall provides protection
- Require 80°C for 10 minutes (moist heat)
- Non-enveloped Viruses
- Hepatitis A, poliovirus, norovirus
- Protein capsid protection
- Alcohol resistance: 60-70% ethanol ineffective
- Fungi and Vegetative Bacteria (Lowest resistance)
- Most susceptible to standard disinfection
- Killed by 70% alcohol in 30 seconds
| Microorganism | D-Value (121°C) | Required Log Reduction | Clinical Significance | Resistance Mechanism |
|---|---|---|---|---|
| Prions | >60 minutes | 6-log | CJD transmission | Protein misfolding |
| B. stearothermophilus | 2.0 minutes | 6-log | Steam sterilization BI | Spore coat protection |
| M. tuberculosis | 0.5 minutes | 6-log | TB transmission | Waxy cell wall |
| Hepatitis B | 0.1 minutes | 4-log | Bloodborne pathogen | Envelope stability |
| E. coli | 0.02 minutes | 6-log | Surgical site infection | Minimal resistance |
💡 Master This: Spaulding's classification system determines the level of processing required - critical items (enter sterile tissue) require sterilization, semi-critical items (contact mucous membranes) need high-level disinfection, non-critical items (intact skin contact) require low-level disinfection
Understanding microbial resistance patterns unlocks the logic behind every sterilization protocol and disinfection choice in clinical practice.
🧬 The Microbial Battlefield: Sterilization and Disinfection Mastery
⚡ The Heat Engine: Physical Sterilization Powerhouse
📌 Remember: STEAM parameters - Saturated steam required, Temperature 121°C standard, Exposure time 15 minutes, Air removal essential, Moisture content 3-17% optimal
Steam Sterilization Parameters and Mechanisms
- Standard Gravity Displacement
- Temperature: 121°C (250°F)
- Pressure: 15 psi above atmospheric
- Time: 15-30 minutes depending on load
- Wrapped instruments: 30 minutes
- Unwrapped metal instruments: 15 minutes
- Liquids: 15-20 minutes
- Pre-vacuum (Prevac) Cycles
- Temperature: 132-134°C (270-273°F)
- Pressure: 27-32 psi above atmospheric
- Time: 4-10 minutes exposure
- Wrapped instruments: 4 minutes
- Porous loads: 10 minutes
- Air removal: 99% vacuum achieved
| Sterilization Method | Temperature | Time | Pressure | Penetration | Applications |
|---|---|---|---|---|---|
| Steam (Gravity) | 121°C | 15-30 min | 15 psi | Excellent | Wrapped instruments |
| Steam (Prevac) | 134°C | 4-10 min | 30 psi | Superior | Porous materials |
| Dry Heat | 160°C | 120 min | Atmospheric | Poor | Powders, oils |
| ETO Gas | 55°C | 2-24 hrs | Subatmospheric | Excellent | Heat-sensitive |
| Hydrogen Peroxide | 50°C | 45-75 min | Vacuum | Good | Electronics |
Dry Heat Sterilization Mechanisms
Dry heat achieves sterilization through oxidative destruction of cellular components, requiring higher temperatures and longer exposure times due to poor heat penetration:
- Hot Air Ovens
- Temperature: 160°C for 120 minutes
- Temperature: 170°C for 60 minutes
- Temperature: 180°C for 30 minutes
- Heat transfer: Convection only (no moisture)
- Incineration
- Temperature: >1000°C
- Complete oxidation to CO₂ and H₂O
- Used for: Pathological waste, contaminated materials
💡 Master This: Steam sterilization is 1000x more efficient than dry heat due to latent heat of vaporization - steam releases 540 calories/gram when condensing, compared to 0.5 calories/gram/°C for dry air heating
Physical sterilization methods provide the foundation for understanding how controlled energy application eliminates microbial life through predictable thermal destruction mechanisms.
⚡ The Heat Engine: Physical Sterilization Powerhouse
🧪 Chemical Warfare: The Molecular Arsenal
📌 Remember: CHEMICAL classes - Chlorine compounds (oxidizing), Hydrogen peroxide (oxidizing), Ethylene oxide (alkylating), Mercury compounds (protein binding), Iodine solutions (halogenating), Caldehydes (cross-linking), Alcohols (membrane disruption), Lysol phenolics (membrane damage)
Oxidizing Agents: Electron Theft Mechanisms
-
Hydrogen Peroxide (H₂O₂)
- Mechanism: Hydroxyl radical formation (•OH)
- Concentration: 6-35% for sterilization
- Contact time: 6-8 hours for sterilization
- High-level disinfection: 6-8 minutes at 7.5%
- Sterilization: 6 hours at 7.5%
- Sporicidal activity: 4-log reduction in 30 minutes
- Material compatibility: Non-corrosive to most metals
-
Peracetic Acid (PAA)
- Mechanism: Peroxide + acetic acid synergy
- Concentration: 0.2-0.35% for sterilization
- Contact time: 12-50 minutes at 50-56°C
- Advantages: No toxic residues, rapid action
- Sporicidal: 6-log reduction in 5 minutes
| Chemical Agent | Mechanism | Concentration | Contact Time | Spectrum | Residue |
|---|---|---|---|---|---|
| H₂O₂ Plasma | Hydroxyl radicals | 58% → Gas | 45-75 min | Broad | None |
| Peracetic Acid | Oxidation | 0.2-0.35% | 12-50 min | Broad | Acetic acid |
| Ethylene Oxide | Alkylation | 450-1200 mg/L | 2-24 hrs | Broad | Toxic |
| Glutaraldehyde | Cross-linking | 2-3.4% | 6-10 hrs | Broad | Toxic |
| OPA | Protein binding | 0.55% | 12 min | Limited | Minimal |
- Ethylene Oxide (ETO)
- Mechanism: Alkylation of amino, carboxyl, sulfhydryl groups
- Concentration: 450-1200 mg/L (8-12% in CO₂)
- Temperature: 37-63°C optimal
- Humidity: 30-80% relative humidity required
- Penetration: Excellent through packaging materials
- Aeration time: 8-12 hours to remove toxic residues
- ETO residue limit: <25 ppm for most devices
- Ethylene chlorohydrin: <25 ppm
- Ethylene glycol: <250 ppm
⭐ Clinical Pearl: ETO sterilization requires 4 critical parameters - gas concentration, temperature, humidity, and time. Failure of any parameter results in sterilization failure, with humidity <30% being the most common cause of cycle failure
Aldehydes: Protein Cross-Linking Mechanisms
-
Glutaraldehyde
- Mechanism: Cross-linking of amino groups in proteins/nucleic acids
- Concentration: 2.0-3.4% alkaline solution
- pH: 7.5-8.5 for optimal activity
- Contact time: 10 hours for sterilization
- High-level disinfection: 20-45 minutes
- Mycobactericidal: 20 minutes
- Sporicidal: 3-10 hours
- Temperature: 20-25°C standard, 35-37°C accelerated
-
Ortho-phthalaldehyde (OPA)
- Mechanism: Protein binding without cross-linking
- Concentration: 0.55% aqueous solution
- Contact time: 12 minutes at 20°C
- Advantages: No activation required, faster action
- Limitations: Not sporicidal, stains proteins
💡 Master This: Glutaraldehyde achieves sterilization through bifunctional alkylation - each molecule can cross-link two amino groups simultaneously, creating irreversible protein denaturation that's 1000x more lethal than monofunctional aldehydes
Chemical sterilants provide precise molecular targeting mechanisms that enable sterilization of heat-sensitive medical devices while maintaining material integrity.
🧪 Chemical Warfare: The Molecular Arsenal
🎯 Pattern Recognition: Clinical Decision Frameworks
📌 Remember: SPAULDING classification - Surgical instruments (critical), Patient care items (semi-critical), Ambulatory surfaces (non-critical), Universal precautions apply, Level of processing determined by risk, Disinfection vs sterilization choice, Infection control priority, Non-negotiable for critical items, Guidelines evidence-based
Device Classification and Processing Requirements
-
Critical Items (Enter sterile tissue/vascular system)
- Risk level: High - direct bloodstream/tissue contact
- Processing requirement: Sterilization (6-log reduction)
- Examples: Surgical instruments, implants, catheters
- Failure consequence: Surgical site infection risk 2-5%
- Methods: Steam, ETO, hydrogen peroxide, peracetic acid
- Steam sterilization: 99.9999% efficacy
- ETO sterilization: 99.9999% efficacy
- Chemical indicators required for every load
-
Semi-Critical Items (Contact mucous membranes)
- Risk level: Moderate - mucosal barrier contact
- Processing requirement: High-level disinfection (6-log vegetative, some spores)
- Examples: Endoscopes, respiratory equipment, anesthesia circuits
- Failure consequence: Healthcare-associated infection risk 0.1-1%
- Methods: Glutaraldehyde, OPA, hydrogen peroxide, peracetic acid
- Contact time: 12-45 minutes depending on agent
- Mycobactericidal activity required

| Device Category | Risk Level | Processing | Log Reduction | Contact Time | Monitoring |
|---|---|---|---|---|---|
| Critical | High | Sterilization | 6-log all | Variable | BI + CI |
| Semi-Critical | Moderate | HLD | 6-log vegetative | 12-45 min | MEC + CI |
| Non-Critical | Low | LLD | 3-log vegetative | 1-10 min | Visual |
| Environmental | Minimal | Cleaning | 2-log reduction | <1 min | None |
| Spill Response | Variable | Disinfection | 4-log target | 10 min | Protocol |
-
Bloodborne Pathogen Exposure
- Target organisms: HIV, HBV, HCV
- Required activity: Virucidal against enveloped viruses
- Minimum concentration: 70% alcohol for 30 seconds
- Alternative agents: 1:10 bleach solution (5000 ppm chlorine)
- Contact time: 1-10 minutes depending on organic load
-
Spore-Forming Bacteria (C. difficile, B. anthracis)
- Required activity: Sporicidal agents only
- Effective agents: Bleach (1000-5000 ppm), hydrogen peroxide (>3%)
- Ineffective agents: Alcohol, quaternary ammonium, phenolics
- Contact time: 5-10 minutes for vegetative forms, >30 minutes for spores
⭐ Clinical Pearl: "When in doubt, go up" - if device classification is uncertain, use the higher level of processing. Semi-critical items can be sterilized, but critical items cannot be only disinfected without compromising patient safety
Material Compatibility Assessment
-
Heat-Sensitive Materials
- Plastics: PVC, polyethylene (max 60°C)
- Electronics: Circuit boards, sensors (max 50°C)
- Optics: Fiber optics, lenses (thermal expansion risk)
- Processing options: ETO, hydrogen peroxide plasma, peracetic acid
- Contraindications: Steam sterilization causes melting/warping
-
Chemical-Sensitive Materials
- Metals: Aluminum (alkali-sensitive), carbon steel (corrosion-prone)
- Rubber: Natural latex (ozone-sensitive), silicone (alcohol-swelling)
- Processing considerations: pH neutral agents preferred
- Rinse requirements: Sterile water final rinse for critical items
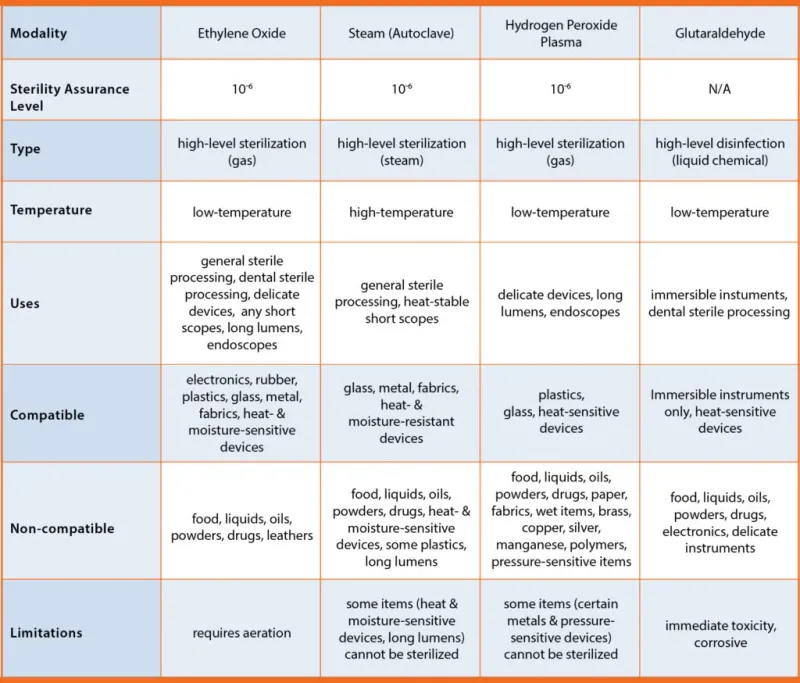
💡 Master This: Material compatibility determines method selection - steam sterilization is preferred when compatible due to superior penetration, no toxic residues, and immediate use capability, but 40% of medical devices require low-temperature alternatives
Pattern recognition in sterilization selection integrates device risk, pathogen targets, and material constraints to ensure optimal processing outcomes while maintaining device integrity.
🎯 Pattern Recognition: Clinical Decision Frameworks
⚖️ Evidence-Based Treatment Algorithms
📌 Remember: MONITOR validation - Mechanical indicators (time/temp), Outcome assessment (sterility), Numerical parameters (pressure), Indicators biological (spores), Testing chemical (color change), Ongoing surveillance (routine), Records documentation (traceability)
Biological Indicator Validation Protocols
-
Steam Sterilization Monitoring
- Primary BI: Geobacillus stearothermophilus spores
- Spore population: 10⁵-10⁶ CFU per indicator
- D-value: 1.5-2.0 minutes at 121°C
- Z-value: 7-10°C (temperature coefficient)
- Frequency: Every load containing implants, weekly for routine loads
- Incubation: 48-56°C for 48 hours (rapid: 3 hours)
- Positive control: Unprocessed BI must show growth
- Negative control: Processed BI must show no growth
- Invalid test: >10% positive controls fail to grow
-
ETO Sterilization Monitoring
- Primary BI: Bacillus atrophaeus spores
- Spore population: 10⁶ CFU per indicator
- D-value: 2.5-3.0 minutes at standard ETO conditions
- Frequency: Every load (no exceptions)
- Incubation: 35-37°C for 7 days
| Sterilization Method | Biological Indicator | D-Value | Incubation | Frequency | Interpretation |
|---|---|---|---|---|---|
| Steam 121°C | G. stearothermophilus | 1.5-2.0 min | 56°C/48hr | Weekly/Implants | Growth = Failure |
| Steam 134°C | G. stearothermophilus | 0.5-1.0 min | 56°C/48hr | Weekly/Implants | Growth = Failure |
| ETO | B. atrophaeus | 2.5-3.0 min | 37°C/7days | Every load | Growth = Failure |
| H₂O₂ Plasma | G. stearothermophilus | 1.0-1.5 min | 56°C/48hr | Every load | Growth = Failure |
| Dry Heat | B. atrophaeus | 4.5-5.0 min | 37°C/7days | Weekly | Growth = Failure |
-
Class 1 Indicators (Process Indicators)
- Purpose: External indication of exposure
- Color change: Immediate visual confirmation
- Information: Processed vs unprocessed only
- Limitation: No sterilization confirmation
- Usage: Every package external placement
-
Class 5 Indicators (Integrating Indicators)
- Purpose: Multi-parameter response (time, temperature, steam)
- Performance: Equivalent to biological indicators
- Response time: Real-time during cycle
- Accuracy: ±2°C temperature, ±10% time
- Advantage: Immediate release of non-implant loads
⭐ Clinical Pearl: Immediate-use steam sterilization (IUSS) requires Class 5 integrating indicators plus mechanical monitoring - biological indicators are not practical due to 3-hour minimum incubation time for rapid results
Quality Assurance Protocols
-
Daily Monitoring Requirements
- Bowie-Dick test: Pre-vacuum sterilizer air removal verification
- Mechanical indicators: All cycles documented (time, temperature, pressure)
- Chemical indicators: Class 1 on every package, Class 5 in test packs
- Biological indicators: Weekly routine, every implant load
- Water quality: <10 CFU/mL for steam generation
-
Failure Response Protocols
- Immediate actions: Stop processing, quarantine load, investigate cause
- Recall criteria: Any positive BI requires recall of all loads since last negative BI
- Documentation: Complete traceability from patient to sterilizer cycle
- Corrective action: Root cause analysis and preventive measures
- Notification: Infection control, risk management, affected departments
💡 Master This: Triple monitoring (mechanical + chemical + biological) provides 99.99% confidence in sterilization efficacy - mechanical indicators confirm cycle parameters, chemical indicators verify package exposure, biological indicators prove microbial kill achievement
Evidence-based monitoring algorithms ensure sterilization process validation through systematic verification of critical parameters and biological efficacy confirmation.
⚖️ Evidence-Based Treatment Algorithms
🔗 Multi-System Integration: The Infection Control Ecosystem
📌 Remember: INTEGRATE systems - Infection surveillance networks, Nosocomial prevention protocols, Tracking and traceability, Environmental monitoring, Guideline compliance, Risk assessment tools, Antimicrobial stewardship, Technology integration, Education and training
Environmental Bioburden and Sterilization Load
-
Operating Room Bioburden Dynamics
- Baseline air quality: <10 CFU/m³ in Class 100 environments
- Personnel shedding: 10⁴-10⁶ particles/person/minute
- Surface contamination: <5 CFU/cm² on critical surfaces
- Impact on sterilization: Higher bioburden requires longer exposure times
- Clean instruments: Standard cycle parameters sufficient
- Contaminated instruments: Extended cycle may be required
- Biofilm presence: 10-1000x increased resistance
-
Water System Integration
- Steam generation quality: <10 CFU/mL, <0.1 EU/mL endotoxin
- Final rinse water: Sterile for critical items, potable for semi-critical
- Biofilm prevention: Chlorine dioxide (0.1-0.8 ppm) continuous treatment
- System validation: Monthly microbiological testing, quarterly endotoxin testing
| System Component | Monitoring Parameter | Target Value | Impact on Sterilization | Integration Point |
|---|---|---|---|---|
| Air Quality | CFU/m³ | <10 Class 100 | Bioburden reduction | Pre-processing |
| Water Systems | CFU/mL | <10 steam | Steam quality | Process medium |
| Surface Bioburden | CFU/cm² | <5 critical | Recontamination risk | Post-processing |
| Personnel Hygiene | Hand CFU | <10 post-wash | Cross-contamination | Handling |
| Waste Management | Pathogen load | 6-log reduction | Environmental safety | Disposal |
-
Multidrug-Resistant Organism (MDRO) Considerations
- MRSA: Standard sterilization effective (vegetative bacteria)
- VRE: Standard sterilization effective (vegetative bacteria)
- CRE: Standard sterilization effective (vegetative bacteria)
- C. difficile: Requires sporicidal agents (spore-forming)
- Alcohol-based hand rub: Ineffective against spores
- Soap and water: Mechanical removal required
- Environmental disinfection: Bleach (1000-5000 ppm chlorine)
-
Biofilm-Associated Resistance
- Resistance increase: 10-1000x compared to planktonic cells
- Penetration barrier: Extracellular polymeric substances (EPS)
- Enhanced cleaning: Enzymatic detergents + ultrasonic energy
- Extended contact: 2-10x normal disinfectant exposure time
- Prevention strategy: Single-use devices for high-risk procedures
⭐ Clinical Pearl: Biofilm-embedded organisms require 10-1000x higher disinfectant concentrations - this explains why single-use devices are preferred for high-risk procedures and why mechanical cleaning is essential before chemical disinfection
Technology Integration and Smart Monitoring
-
Real-Time Sterilization Monitoring
- RFID tracking: Individual instrument traceability from patient to sterilizer
- Wireless sensors: Continuous temperature/pressure monitoring
- Automated documentation: Zero transcription error systems
- Predictive maintenance: Algorithm-based equipment failure prediction
- Integration benefits: 99.9% documentation accuracy, 50% reduced errors
-
Artificial Intelligence Applications
- Pattern recognition: Anomaly detection in sterilization cycles
- Predictive analytics: Failure prediction 24-48 hours in advance
- Optimization algorithms: Cycle parameter adjustment for efficiency
- Quality trending: Statistical process control with real-time alerts
- Outcome correlation: Infection rates linked to processing parameters
💡 Master This: Digital integration transforms sterilization from reactive to predictive - AI-powered systems can predict sterilizer failures 24-48 hours before occurrence, preventing processing delays and ensuring continuous patient care capability
Multi-system integration creates comprehensive infection prevention ecosystems where molecular sterilization science scales to institutional patient safety outcomes through interconnected monitoring and control systems.
🔗 Multi-System Integration: The Infection Control Ecosystem
🎯 Clinical Mastery Arsenal: Rapid Decision Tools
📌 Remember: MASTER decision framework - Method selection criteria, Agent compatibility matrix, Sterilization parameter requirements, Timing and contact specifications, Efficacy validation requirements, Risk assessment protocols
Essential Clinical Decision Matrix
| Clinical Scenario | Primary Pathogen | Device Type | Method Choice | Critical Parameters | Validation |
|---|---|---|---|---|---|
| Surgical Instruments | Mixed flora | Critical/Metal | Steam 134°C | 4 min exposure | BI weekly |
| Flexible Endoscopes | P. aeruginosa | Semi-critical | HLD/OPA | 12 min contact | MEC daily |
| C. diff Outbreak | Spore-forming | Environmental | Bleach 5000ppm | 5 min contact | ATP testing |
| Heat-Sensitive | Vegetative | Critical/Plastic | H₂O₂ Plasma | 45-75 min cycle | BI every load |
| Emergency/IUSS | Mixed flora | Critical/Metal | Steam 134°C | 4 min unwrapped | Class 5 CI |
- 30-Second Device Assessment
- Step 1: Spaulding classification (5 seconds)
- Critical = Sterilization required
- Semi-critical = HLD minimum
- Non-critical = LLD acceptable
- Step 2: Heat stability check (5 seconds)
- Metal/glass = Steam preferred
- Plastic/electronics = Low-temperature methods
- Step 3: Urgency evaluation (5 seconds)
- Routine = Standard protocols
- Emergency = IUSS considerations
- Step 4: Pathogen risk (10 seconds)
- Standard = Routine processing
- Spores/biofilm = Enhanced protocols
- Step 5: Method selection (5 seconds)
- Apply decision matrix result
- Step 1: Spaulding classification (5 seconds)
⭐ Clinical Pearl: "Critical + Heat-Stable = Steam" - this simple rule covers >80% of sterilization decisions in acute care settings, with steam sterilization being the gold standard for surgical instruments due to superior penetration, no residues, and immediate availability
Emergency Response Protocols
-
Sterilization Failure Response (<30 minutes)
- Immediate: Stop processing, quarantine affected loads
- 5 minutes: Notify infection control and risk management
- 15 minutes: Identify all potentially affected patients
- 30 minutes: Initiate recall protocol if indicated
- Documentation: Complete traceability within 2 hours
-
Outbreak Response Sterilization
- C. difficile: Switch to sporicidal agents (bleach 1000-5000 ppm)
- CRE/MDRO: Enhanced cleaning + standard sterilization
- Prion disease: Special protocols (134°C for 18 minutes + 1M NaOH)
- Biofilm: Enzymatic cleaning + extended contact times
💡 Master This: Time-critical decisions require pre-established protocols - emergency sterilization protocols must be immediately accessible and regularly drilled, as delays >30 minutes can compromise surgical schedules and patient safety outcomes
Clinical mastery integrates rapid assessment skills with evidence-based protocols to ensure optimal sterilization decisions under time pressure while maintaining patient safety standards.
🎯 Clinical Mastery Arsenal: Rapid Decision Tools
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























