Parasitology

On this page
🦠 Parasitic Foundations: The Microscopic Invasion Blueprint
Parasites have perfected the art of invasion over millions of years, exploiting human biology with remarkable precision-and your ability to recognize, diagnose, and treat these infections can mean the difference between cure and catastrophic complications. You'll master the lifecycle blueprints that reveal vulnerabilities, build a diagnostic toolkit to catch elusive organisms, decode the clinical patterns that distinguish one parasite from another, and deploy targeted treatments that disrupt these ancient invaders. This lesson transforms parasitology from memorizing names into strategic clinical thinking, equipping you to protect patients across diverse practice settings.
The parasitic world divides into three major kingdoms: protozoa (single-celled organisms), helminths (multicellular worms), and arthropods (vectors and ectoparasites). Each group employs distinct survival strategies, with protozoa utilizing rapid multiplication (dividing every 6-24 hours), helminths focusing on longevity (surviving 10-30 years), and arthropods serving as transmission vectors for over 100 diseases.
📌 Remember: PHAV - Protozoa (single cells), Helminths (worms), Arthropods (vectors), Viral-like (some protozoa) - covers 99% of medically important parasites with distinct diagnostic approaches
-
Protozoa Classification Architecture
- Sarcodina (amoebae): Entamoeba histolytica causing 50 million infections annually
- Mastigophora (flagellates): Giardia lamblia with 200 million cases globally
- Hemoflagellates: Trypanosoma species affecting 300,000 people yearly
- Leishmania: 12 million infected across 98 countries
- Ciliophora (ciliates): Balantidium coli with <1% prevalence
- Sporozoa (spore-formers): Plasmodium causing 241 million malaria cases
-
Helminth Structural Hierarchy
- Nematodes (roundworms): 1.5 billion infections with Ascaris lumbricoides
- Platyhelminthes (flatworms): 240 million schistosomiasis cases
- Trematodes (flukes): 56 million liver fluke infections
- Cestodes (tapeworms): 50 million taeniasis cases globally
⭐ Clinical Pearl: Parasitic infections show geographic clustering with >90% of cases concentrated in tropical/subtropical regions where temperature >20°C and humidity >60% create optimal transmission conditions
| Parasite Group | Size Range | Reproduction Rate | Host Specificity | Diagnostic Method | Treatment Duration |
|---|---|---|---|---|---|
| Protozoa | 1-50 μm | 6-24 hours | High (95%) | Microscopy/PCR | 7-14 days |
| Nematodes | 1mm-35cm | Monthly | Moderate (60%) | Egg detection | 1-3 days |
| Trematodes | 1-7cm | Weekly | Low (30%) | Egg/serology | 1-2 days |
| Cestodes | 3mm-10m | Daily segments | Moderate (70%) | Segment/egg ID | Single dose |
| Arthropods | 1-30mm | Weekly | Variable | Visual/PCR | Topical/systemic |
The parasitic life cycle complexity determines clinical presentation patterns. Simple cycles (direct transmission) like Enterobius vermicularis show family clustering with >80% household infection rates. Complex cycles requiring intermediate hosts like Schistosoma species demonstrate geographic restriction to specific snail habitats within 200 meters of freshwater sources.
📌 Remember: DIVE - Direct (person-to-person), Indirect (environmental), Vector-borne (arthropod), External (skin contact) - covers all four transmission patterns with specific prevention strategies
Understanding parasitic pathogenesis requires recognizing three damage mechanisms: mechanical (tissue destruction), toxic (metabolite release), and immunological (host response). Mechanical damage predominates in helminth infections with >1000 worms causing intestinal obstruction. Toxic effects characterize protozoan infections where metabolic byproducts trigger inflammatory cascades. Immunological damage develops in chronic infections lasting >6 months, leading to autoimmune complications in 15-30% of cases.
Connect these foundational principles through diagnostic approaches to understand how parasitic identification drives targeted therapy selection.
🦠 Parasitic Foundations: The Microscopic Invasion Blueprint
🔬 Diagnostic Mastery: The Parasitic Detection Arsenal
📌 Remember: STOP - Sample timing (circadian rhythms), Technique selection (wet/stained), Optimal processing (concentration methods), Pattern recognition (morphological keys) - ensures >95% diagnostic accuracy
- Sample Collection Optimization
- Stool specimens: Three samples over 7-10 days increases sensitivity to >90%
- Blood samples: Thick/thin smears during fever spikes for malaria
- Timing: Every 6-8 hours during symptomatic periods
- Volume: 10-20 drops for thick smears, 1-2 drops for thin smears
- Urine collection: Terminal stream between 10 AM-2 PM for Schistosoma haematobium
- Tissue samples: Multiple sites with >5mm depth for adequate cellularity
- Microscopic Examination Hierarchy
- Direct wet mount: Immediate motility assessment for trophozoites
- Iodine preparation: Nuclear detail enhancement for cyst identification
- Lugol's iodine: 1:5 dilution for optimal contrast
- Examination time: <30 minutes before artifact formation
- Permanent stains: Trichrome or iron-hematoxylin for species confirmation
- Concentration methods: Formalin-ethyl acetate increases sensitivity 3-5 fold
⭐ Clinical Pearl: Fresh specimens examined within 30 minutes show motile trophozoites in >80% of positive cases, while delayed examination reduces motility detection to <20% after 2 hours at room temperature
| Diagnostic Method | Sensitivity | Specificity | Time Required | Cost Factor | Best Application |
|---|---|---|---|---|---|
| Direct Microscopy | 60-80% | >95% | 10-15 min | Low (1x) | Acute infections |
| Concentration | 85-95% | >95% | 45-60 min | Low (2x) | Light infections |
| Antigen Detection | 90-98% | >98% | 15-30 min | High (10x) | Malaria/Giardia |
| PCR Methods | >98% | >99% | 4-6 hours | Very High (50x) | Species confirmation |
| Serology | 70-90% | 80-95% | 2-4 hours | Moderate (5x) | Chronic infections |
Antigen detection revolutionizes point-of-care diagnosis with rapid diagnostic tests (RDTs) providing results within 15-20 minutes. Malaria RDTs detect histidine-rich protein 2 (HRP2) with >95% sensitivity for P. falciparum at >100 parasites/μL. Giardia antigen tests identify cyst wall proteins with >90% sensitivity even in formed stools where microscopy fails.
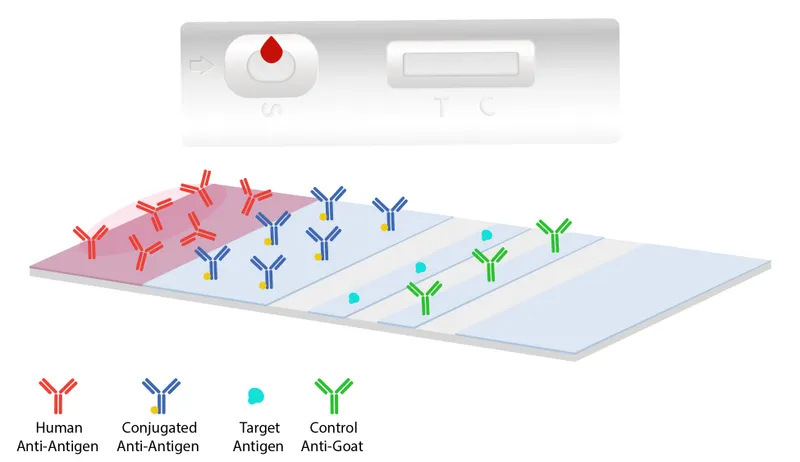
- Molecular Diagnostic Applications
- Real-time PCR: Species identification with >99% accuracy
- Multiplex PCR: Simultaneous detection of 5-10 pathogens
- Gastrointestinal panels: 15-20 targets including parasites/bacteria/viruses
- Results: 4-6 hours with quantitative data
- Loop-mediated amplification (LAMP): Field-deployable with visual readout
- Next-generation sequencing: Metagenomic analysis for unknown pathogens
📌 Remember: MARS - Microscopy (morphology), Antigen (rapid), RNA/DNA (molecular), Serology (antibodies) - represents four diagnostic tiers with increasing complexity and cost
Quantitative assessment guides treatment decisions and prognosis. Malaria parasite density >100,000/μL indicates severe disease requiring intensive monitoring. Schistosoma egg counts >400 eggs/gram stool suggest heavy infection with increased morbidity risk. Microfilariae density >1000/mL blood correlates with >80% transmission potential.
Connect diagnostic mastery through clinical correlation frameworks to understand how laboratory findings translate into therapeutic decisions.
🔬 Diagnostic Mastery: The Parasitic Detection Arsenal
🎯 Clinical Pattern Recognition: The Parasitic Syndrome Decoder
📌 Remember: GIFTS - GI (diarrhea/dysentery), Infectious fever (malaria/leishmaniasis), Filariasis (lymphatic swelling), Tissue invasion (eosinophilia), Skin lesions (cutaneous forms) - covers >90% of parasitic presentations
- Gastrointestinal Syndrome Recognition
- Acute diarrhea (<14 days): Giardia, Cryptosporidium, Cyclospora
- Watery diarrhea: >6 stools/day with minimal blood
- Onset timing: 1-2 weeks post-exposure for most protozoa
- Chronic diarrhea (>30 days): Giardia, Entamoeba histolytica
- Malabsorption signs: Weight loss >5%, steatorrhea
- Eosinophilia: >5% suggests helminth involvement
- Dysentery pattern: Blood/mucus with <10 stools/day
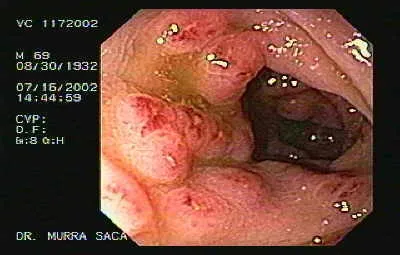
- E. histolytica: Flask-shaped ulcers on colonoscopy
- Balantidium coli: Large trophozoites (50-100 μm)
- Acute diarrhea (<14 days): Giardia, Cryptosporidium, Cyclospora
- Fever Pattern Analysis
- Malaria patterns: Cyclical fever every 48-72 hours
- P. vivax/ovale: 48-hour cycles with rigors
- P. malariae: 72-hour cycles with milder symptoms
- P. falciparum: Irregular patterns with severe complications
- Visceral leishmaniasis: Prolonged fever >2 weeks with hepatosplenomegaly
- Kala-azar: Fever >39°C for >1 month
- Pancytopenia: WBC <4000, platelets <100,000
- African trypanosomiasis: Biphasic fever with lymphadenopathy
- Malaria patterns: Cyclical fever every 48-72 hours
⭐ Clinical Pearl: Eosinophilia >5% occurs in >80% of tissue-invasive helminths but <10% of intestinal protozoa, making it a key discriminator between worm and single-celled infections
| Clinical Syndrome | Key Features | Timeline | Geographic Clues | Diagnostic Priority | Mortality Risk |
|---|---|---|---|---|---|
| Acute Malaria | Fever + rigors | 1-4 weeks | Endemic areas | Immediate | High (15%) |
| Amoebic Dysentery | Blood + mucus | 2-6 weeks | Poor sanitation | Urgent | Moderate (5%) |
| Visceral Leishmaniasis | Prolonged fever | 2-6 months | Mediterranean/India | High | High (20%) |
| Acute Schistosomiasis | Fever + eosinophilia | 4-8 weeks | Freshwater exposure | Moderate | Low (1%) |
| Neurocysticercosis | Seizures + headache | Months-years | Pork consumption | High | Variable |
- Cutaneous leishmaniasis: Painless ulcers with raised borders
- Oriental sore: Single lesion healing in 6-12 months
- New World: Multiple lesions with satellite nodules
- Larva migrans: Serpiginous tracks advancing 2-5 cm/day
- Cutaneous: Hookworm larvae in sandy areas
- Visceral: Toxocara with hepatomegaly
- Onchocerciasis: Subcutaneous nodules with intense pruritus
💡 Master This: Travel history timing correlates with incubation periods - malaria (7-30 days), schistosomiasis (4-8 weeks), filariasis (6-12 months), cysticercosis (months-years) - enabling targeted diagnostic approaches
Eosinophilia magnitude provides diagnostic clues: mild (5-10%) suggests early helminth infection, moderate (10-20%) indicates established tissue invasion, severe (>20%) points to hyperinfection syndromes or allergic responses. Strongyloides hyperinfection in immunocompromised patients shows >30% eosinophilia with >50% mortality if untreated.
- Neurological Syndrome Recognition
- Neurocysticercosis: Seizures in >80% of symptomatic cases
- Parenchymal cysts: Focal seizures with calcifications
- Ventricular cysts: Hydrocephalus with increased ICP
- Cerebral malaria: Altered consciousness with retinal hemorrhages
- African sleeping sickness: Progressive somnolence with behavioral changes
- Neurocysticercosis: Seizures in >80% of symptomatic cases
📌 Remember: HELPS - Helminth eosinophilia, Exposure geography, Latency periods, Pattern recognition, Syndrome clustering - enables rapid differential narrowing from >100 to <5 candidates
Connect pattern recognition through systematic evaluation frameworks to understand how clinical presentations guide targeted diagnostic testing.
🎯 Clinical Pattern Recognition: The Parasitic Syndrome Decoder
⚖️ Systematic Evaluation: The Parasitic Differential Matrix
📌 Remember: SCALE - Size measurements (precise), Characteristic features (unique), Associated findings (clinical), Location specificity (geographic), Epidemiological patterns (transmission) - enables >95% species differentiation
- Protozoan Discrimination Matrix
- Entamoeba species differentiation:
- E. histolytica: 10-25 μm trophozoites with ingested RBCs
- E. dispar: Identical morphology but non-pathogenic
- E. coli: 15-50 μm with >4 nuclei in cysts
- Plasmodium species identification:
- P. falciparum: Ring forms with multiple infections/RBC
- P. vivax: Large rings with Schüffner's dots
- P. malariae: Band forms with 72-hour cycles
- P. ovale: Oval RBCs with fimbriated edges
- Entamoeba species differentiation:
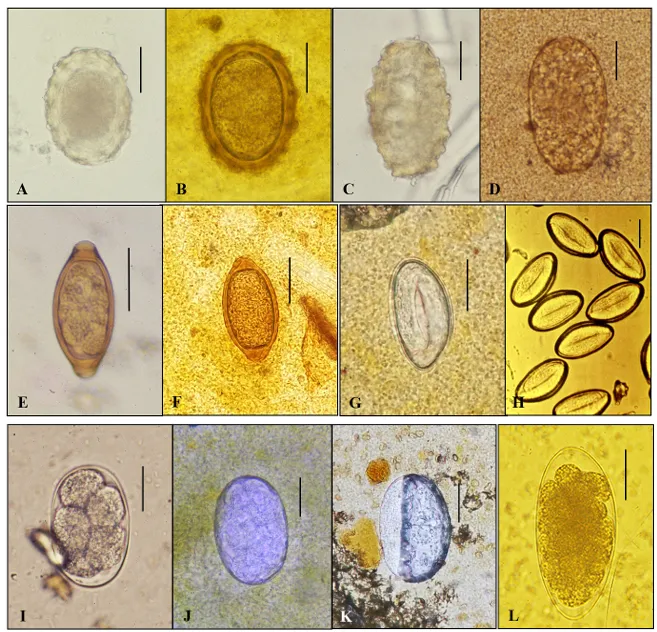
- Helminth Egg Discrimination
- Schistosoma species: Spine location determines species
- S. mansoni: Lateral spine on oval eggs (114-175 μm)
- S. haematobium: Terminal spine on elongated eggs (112-170 μm)
- S. japonicum: No spine on round eggs (70-100 μm)
- Hookworm differentiation: Requires larval culture for species ID
- Necator americanus: Cutting plates in buccal cavity
- Ancylostoma duodenale: Teeth in buccal cavity
- Taenia species: Egg morphology identical, requires proglottid analysis
- Schistosoma species: Spine location determines species
⭐ Clinical Pearl: Molecular methods (PCR) provide definitive species identification for morphologically identical organisms like E. histolytica/dispar (>99% accuracy) and Plasmodium species in mixed infections (>95% sensitivity)
| Organism Pair | Key Discriminator | Clinical Significance | Diagnostic Method | Treatment Difference | Accuracy Rate |
|---|---|---|---|---|---|
| E. histolytica/dispar | Antigen/PCR | Pathogenic vs benign | Molecular | Yes vs no treatment | >99% |
| P. falciparum/vivax | RBC morphology | Severe vs relapsing | Microscopy | Different drugs | >95% |
| S. mansoni/haematobium | Egg spine location | Intestinal vs urinary | Microscopy | Same treatment | >98% |
| Necator/Ancylostoma | Larval culture | Geographic distribution | Culture | Same treatment | >90% |
| T. solium/saginata | Proglottid morphology | Cysticercosis risk | Morphology | Same treatment | >95% |
- Fever patterns: Malaria vs other causes
- Periodic fever: >80% specific for malaria in endemic areas
- Continuous fever: Typhoid, leishmaniasis, other infections
- Diarrheal syndromes: Parasitic vs bacterial
- Chronic diarrhea (>30 days): >70% parasitic in travelers
- Eosinophilia: >90% specific for helminths
- Skin lesions: Parasitic vs other causes
- Serpiginous tracks: >95% specific for larva migrans
- Painless ulcers: Leishmaniasis vs bacterial infections
💡 Master This: Geographic specificity narrows differential diagnosis dramatically - P. falciparum in sub-Saharan Africa (>90% of cases), P. vivax in Asia/Americas (>80%), Leishmania tropica in Middle East (>95%)
Laboratory parameter integration enhances diagnostic accuracy: eosinophilia >10% with travel history suggests helminth infection in >85% of cases. Thrombocytopenia <100,000 with fever indicates malaria in >70% of endemic area travelers. Elevated alkaline phosphatase with eosinophilia points to liver fluke infection in >80% of appropriate geographic exposures.
- Epidemiological Pattern Recognition
- Transmission season: Malaria peaks during rainy seasons
- Water exposure: Schistosomiasis requires freshwater contact
- Food consumption: Taeniasis from undercooked meat
- Vector distribution: Leishmaniasis limited to sandfly habitats
📌 Remember: TEMPO - Timing (seasonal patterns), Exposure type (water/food/vector), Morphological features (size/shape), Pattern recognition (clinical), Outcome correlation (treatment response) - provides systematic approach to species identification
Connect systematic evaluation through treatment selection algorithms to understand how accurate diagnosis drives optimal therapeutic outcomes.
⚖️ Systematic Evaluation: The Parasitic Differential Matrix
🎯 Treatment Algorithms: The Parasitic Warfare Strategy
Antiparasitic therapy operates through five strategic principles: species-specific targeting (drug selection), pharmacokinetic optimization (dosing), resistance prevention (combination therapy), host factor consideration (comorbidities), and outcome monitoring (treatment response). Drug selection accuracy depends on precise species identification, as >40% of antiparasitic agents show species-specific activity with complete inefficacy against morphologically similar organisms.
📌 Remember: SMART - Species identification (precise), Mechanism targeting (specific), Adverse effects (monitoring), Resistance patterns (local), Treatment duration (complete) - ensures >90% cure rates with minimal toxicity
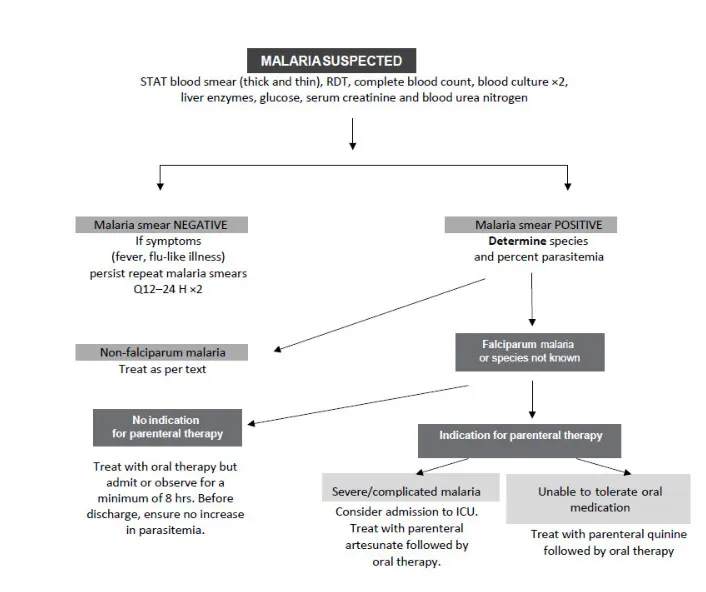
- Antimalarial Treatment Algorithms
- P. falciparum (uncomplicated): Artemisinin combination therapy (ACT)
- Artemether-lumefantrine: 6 doses over 3 days
- Artesunate-amodiaquine: 3 days with food
- Cure rates: >95% in non-resistant areas
- Severe malaria: IV artesunate 2.4 mg/kg at 0, 12, 24 hours
- Loading dose: Not required (rapid distribution)
- Mortality reduction: 15-25% vs quinine
- P. vivax/ovale: Chloroquine + primaquine for hypnozoites
- G6PD testing: Mandatory before primaquine
- Relapse prevention: >95% with 14-day primaquine
- P. falciparum (uncomplicated): Artemisinin combination therapy (ACT)
- Intestinal Protozoa Treatment
- Giardiasis: Metronidazole 250 mg TID × 7 days
- Alternative: Tinidazole 2g single dose (>90% efficacy)
- Pediatric: 15 mg/kg/day divided TID
- Amoebiasis (invasive): Metronidazole + paromomycin
- Tissue phase: Metronidazole 750 mg TID × 10 days
- Luminal phase: Paromomycin 25-35 mg/kg/day × 7 days
- Cryptosporidiosis: Nitazoxanide 500 mg BID × 3 days
- Giardiasis: Metronidazole 250 mg TID × 7 days
⭐ Clinical Pearl: Combination therapy for invasive amoebiasis prevents relapse in >95% of cases by eliminating tissue trophozoites (metronidazole) and luminal cysts (paromomycin), while monotherapy shows 20-30% relapse rates
| Parasite | First-line Drug | Dosage | Duration | Cure Rate | Alternative | Resistance Rate |
|---|---|---|---|---|---|---|
| P. falciparum | Artemether-lumefantrine | 4 tablets BID | 3 days | >95% | Artesunate-amodiaquine | <5% |
| Giardia lamblia | Metronidazole | 250 mg TID | 7 days | >90% | Tinidazole | <10% |
| E. histolytica | Metronidazole | 750 mg TID | 10 days | >95% | Tinidazole | <5% |
| Ascaris lumbricoides | Albendazole | 400 mg single | 1 day | >95% | Mebendazole | <5% |
| Schistosoma | Praziquantel | 40 mg/kg | 1 day | >90% | Oxamniquine | <10% |
- Soil-transmitted helminths: Single-dose therapy
- Albendazole 400 mg for Ascaris, hookworm, Trichuris
- Cure rates: >95% for Ascaris, >80% for hookworm
- Schistosomiasis: Praziquantel 40 mg/kg single dose
- All species: Same dosage with >90% efficacy
- Timing: Avoid during acute Katayama syndrome
- Filariasis: Diethylcarbamazine (DEC) with precautions
- Lymphatic filariasis: DEC 6 mg/kg/day × 12 days
- Onchocerciasis: Ivermectin 150 μg/kg annually
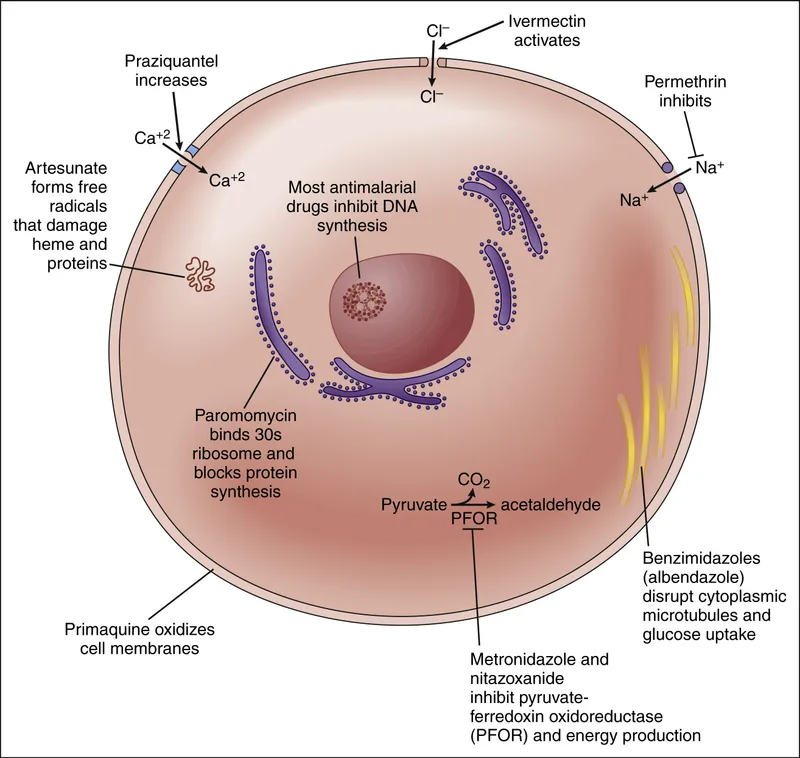
💡 Master This: Drug resistance monitoring requires treatment failure documentation - <90% cure rate at 4 weeks post-treatment suggests emerging resistance and alternative therapy selection
Adverse effect management proves critical for treatment completion: Metronidazole causes metallic taste and GI upset in >50% of patients but rarely requires discontinuation. Praziquantel triggers transient neurological symptoms in 10-15% due to dying parasites but resolves within 24-48 hours. Ivermectin may cause Mazzotti reactions in onchocerciasis with severe pruritus requiring antihistamine support.
- Special Population Considerations
- Pregnancy: Limited options with safety data
- Malaria: Artemether-lumefantrine after first trimester
- Helminths: Defer treatment unless severe disease
- Immunocompromised: Extended therapy often required
- Strongyloides: Ivermectin 200 μg/kg/day × 2 days, repeat in 2 weeks
- Pediatric dosing: Weight-based with age considerations
- Pregnancy: Limited options with safety data
📌 Remember: CURED - Combination therapy (resistance prevention), Understanding mechanisms (drug selection), Resistance monitoring (efficacy tracking), Effect management (adverse events), Duration completion (cure optimization) - maximizes therapeutic success
Connect treatment algorithms through prevention strategies to understand how therapeutic knowledge integrates with public health approaches for comprehensive parasitic disease control.
🎯 Treatment Algorithms: The Parasitic Warfare Strategy
🛡️ Prevention & Control: The Parasitic Defense Network
📌 Remember: SHIELD - Sanitation (water/waste), Host protection (nets/repellents), Intermediate host control (snails/vectors), Education (behavior change), Larvicide application (breeding sites), Drug administration (mass treatment) - provides comprehensive protection
- Vector Control Strategies
- Malaria prevention: Multiple intervention approach
- Long-lasting insecticidal nets (LLINs): >80% household coverage
- Indoor residual spraying: >80% structure coverage
- Larval source management: Breeding site elimination
- Leishmaniasis control: Sandfly management
- Insecticide spraying: Peridomestic areas every 6 months
- Environmental modification: Organic matter removal
- Lymphatic filariasis: Mass drug administration (MDA)
- Annual treatment: >65% population coverage for >5 years
- Drug combination: Albendazole + DEC or ivermectin
- Malaria prevention: Multiple intervention approach
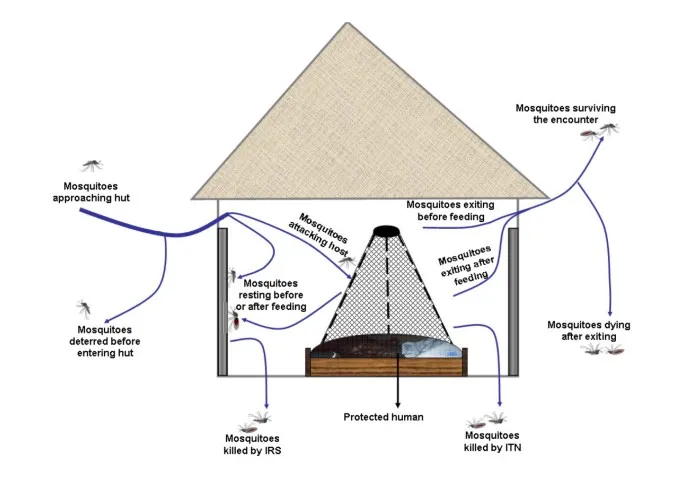
- Environmental Sanitation
- Water quality management: Schistosomiasis prevention
- Safe water sources: >20 meters from contaminated areas
- Water treatment: Boiling or filtration for >1 minute
- Swimming avoidance: Freshwater bodies in endemic areas
- Waste management: Soil-transmitted helminth control
- Improved sanitation: >75% population access reduces transmission by 60%
- Sewage treatment: Proper disposal prevents environmental contamination
- Food safety: Foodborne parasite prevention
- Cooking temperatures: >70°C for >2 minutes kills most parasites
- Freezing: -20°C for >24 hours eliminates tissue parasites
- Water quality management: Schistosomiasis prevention
⭐ Clinical Pearl: Mass drug administration programs achieve elimination when >65% population coverage maintained for >5 consecutive years, reducing microfilariae prevalence to <1% and breaking transmission cycles
| Control Strategy | Target Disease | Coverage Required | Duration | Efficacy Rate | Cost per Person |
|---|---|---|---|---|---|
| LLINs | Malaria | >80% households | 3-5 years | 50-70% | $2-5/year |
| MDA | Lymphatic filariasis | >65% population | >5 years | >90% | $0.50-1/year |
| IRS | Malaria/Leishmaniasis | >80% structures | 6-12 months | 60-80% | $3-8/year |
| Sanitation | STH | >75% access | Permanent | 60-80% | $50-200/person |
| Water treatment | Schistosomiasis | >90% sources | Permanent | >95% | $10-50/person |
- Malaria prevention: Travelers to endemic areas
- Atovaquone-proguanil: Daily dosing starting 1-2 days before travel
- Doxycycline: Daily dosing starting 1-2 days before travel
- Mefloquine: Weekly dosing starting 1-2 weeks before travel
- Strongyloidiasis: Immunocompromised patients
- Ivermectin: 200 μg/kg monthly in high-risk populations
- Pneumocystis: HIV patients with CD4 <200
- Trimethoprim-sulfamethoxazole: Daily or thrice weekly
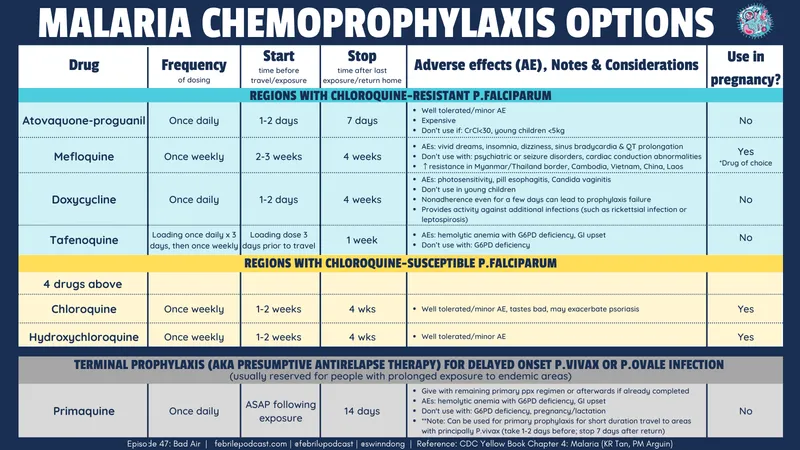
💡 Master This: Integrated control combining vector management, environmental modification, and mass treatment achieves synergistic effects with >90% transmission reduction compared to <60% with single interventions
Surveillance systems enable early outbreak detection and control program monitoring: Active case detection in high-risk areas identifies asymptomatic carriers maintaining transmission. Entomological surveillance tracks vector density and insecticide resistance patterns. Drug efficacy monitoring detects emerging resistance requiring treatment protocol updates.
- Health Education Components
- Behavior change: Personal protection measures
- Bed net usage: Every night usage increases efficacy by 40%
- Repellent application: DEET >20% provides >6 hours protection
- Community engagement: Local participation in control programs
- Community health workers: Training and supervision
- School-based programs: Children as change agents
- Risk communication: Travel medicine counseling
- Pre-travel consultation: 4-6 weeks before departure
- Post-travel screening: Symptomatic and high-risk travelers
- Behavior change: Personal protection measures
📌 Remember: IMPACT - Integrated approach (multiple strategies), Monitoring systems (surveillance), Participation (community), Adaptation (local conditions), Coverage (population-wide), Timing (sustained effort) - ensures long-term success
Connect prevention strategies through rapid reference tools to create comprehensive clinical mastery frameworks for immediate parasitic disease management.
🛡️ Prevention & Control: The Parasitic Defense Network
🎯 Clinical Mastery Arsenal: The Parasitic Command Center
📌 Remember: MASTER - Morphology (identification), Algorithms (systematic), Syndromes (patterns), Treatment (targeted), Evaluation (monitoring), Risk reduction (prevention) - provides comprehensive clinical framework
- Essential Diagnostic Thresholds
- Malaria severity indicators:
- Parasitemia >5%: Severe disease requiring intensive care
- Parasitemia >20%: Exchange transfusion consideration
- Glucose <60 mg/dL: Cerebral malaria complication
- Eosinophilia interpretation:
- 5-10%: Early helminth infection or allergic response
- 10-20%: Established tissue invasion by helminths
- >20%: Hyperinfection or drug hypersensitivity
- Stool examination priorities:
- Fresh specimen <30 minutes: Motile trophozoites visible
- Three specimens: >90% sensitivity for most parasites
- Concentration methods: 3-5 fold sensitivity increase
- Malaria severity indicators:
⭐ Clinical Pearl: Geographic exposure within specific timeframes narrows differential diagnosis dramatically - malaria (7-30 days), schistosomiasis (4-8 weeks), filariasis (6-12 months), cysticercosis (months-years)
| Clinical Scenario | Key Decision Point | Critical Threshold | Immediate Action | Monitoring Parameter | Success Metric |
|---|---|---|---|---|---|
| Fever + Travel | Malaria testing | Within 1 hour | Thick/thin smears | Parasitemia level | <24h diagnosis |
| Chronic Diarrhea | Stool examination | 3 specimens | Fresh + concentrated | Symptom resolution | <7 days |
| Eosinophilia >10% | Helminth workup | Travel history | Stool + serology | Eosinophil count | <4 weeks |
| Skin Lesions | Geographic correlation | Exposure timing | Biopsy/scraping | Lesion healing | <2 weeks |
| Neurological symptoms | Imaging studies | Seizure onset | CT/MRI + CSF | ICP monitoring | <48 hours |
- Life-threatening presentations: Immediate therapy
- Severe malaria: IV artesunate 2.4 mg/kg stat
- Amoebic liver abscess: Metronidazole 750 mg IV TID
- Strongyloides hyperinfection: Ivermectin 200 μg/kg daily
- Empirical therapy indications: High clinical suspicion
- Malaria in endemic area: Treat before confirmation if delay >2 hours
- Giardiasis in travelers: Metronidazole if typical presentation
- Combination therapy requirements: Invasive infections
- Amoebiasis: Tissue + luminal agents required
- Malaria: Artemisinin combinations prevent resistance
- Monitoring and Follow-up Protocols
- Treatment response assessment:
- Malaria: Parasitemia clearance by 72 hours
- Giardiasis: Symptom resolution within 5-7 days
- Helminths: Egg clearance at 4 weeks post-treatment
- Adverse effect monitoring:
- Artemisinin: Delayed hemolysis at 1-3 weeks
- Metronidazole: Peripheral neuropathy with prolonged use
- Ivermectin: Mazzotti reaction in onchocerciasis
- Cure verification:
- Repeat testing: 4 weeks after treatment completion
- Clinical assessment: Symptom resolution and functional improvement
- Treatment response assessment:
💡 Master This: Treatment failure defined as <90% cure rate at 4 weeks suggests drug resistance requiring alternative therapy and resistance surveillance reporting
Prevention counseling completes comprehensive care: Pre-travel consultation 4-6 weeks before departure enables appropriate prophylaxis selection. Post-travel screening for high-risk exposures detects asymptomatic infections. Household screening for index cases identifies additional infections requiring treatment.
- Clinical Pearls for Mastery
- Pattern recognition: Geographic + temporal + clinical clustering
- Diagnostic efficiency: Right test at right time for right patient
- Treatment optimization: Species-specific therapy with resistance awareness
- Outcome monitoring: Systematic follow-up ensures cure verification
- Prevention integration: Individual and public health approaches
📌 Remember: EXPERT - Exposure history (detailed), Xamination (systematic), Pattern recognition (clinical), Evaluation (diagnostic), Rx (treatment), Tracking (follow-up) - defines clinical excellence in parasitic disease management
Quality metrics for parasitic disease management: Diagnostic accuracy >95%, treatment completion >90%, cure rates >85%, adverse events <10%, prevention counseling 100%. Expert practitioners achieve these benchmarks consistently through systematic approaches and continuous learning.
🎯 Clinical Mastery Arsenal: The Parasitic Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























