AMR Basics - Resistance Rising
- Antimicrobial Resistance (AMR): Microbe's ability to survive drugs, rendering treatments ineffective.
- Types:
- Intrinsic: Natural insensitivity (e.g., Gram-negatives to Vancomycin via outer membrane).
- Acquired: New resistance via genetic change (mutation, Horizontal Gene Transfer - HGT).
- Impact: ↑ Treatment failure, ↑ morbidity/mortality, ↑ costs. Major global health threat.
⭐ Enzymatic inactivation (e.g., β-lactamases vs. penicillins) is a very common acquired resistance mechanism.
Resistance Mechanisms - Bugs' Smart Defenses
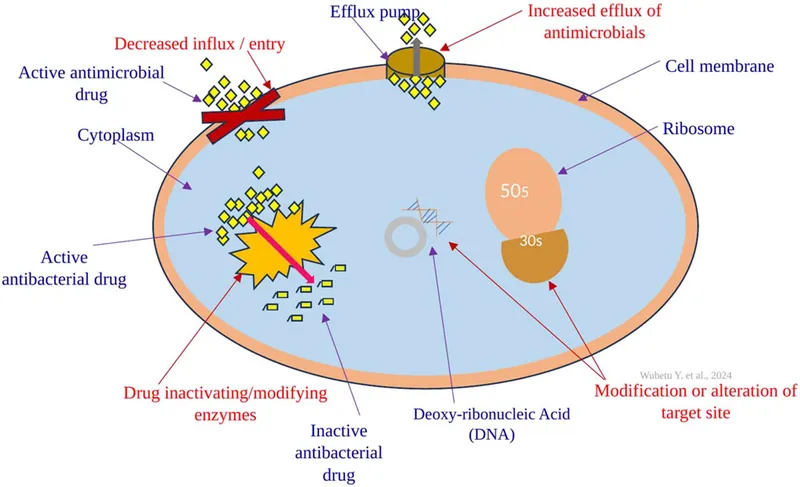
Bacteria employ several key strategies to resist antimicrobial drugs:
| Mechanism | Action | Key Examples (Affected Drugs) |
|---|---|---|
| Enzymatic Inactivation | Degrade/modify drug | β-lactamases (β-lactams); AMEs (Aminoglycosides) |
| Target Modification | Alter drug's binding site | PBP2a (mecA) in MRSA (β-lactams); VanA/B in VRE (Vancomycin) |
| Efflux Pumps | Pump drug out of cell | Tetracyclines, Macrolides, Fluoroquinolones |
| Decreased Permeability | ↓ Drug entry (e.g., porin loss) | β-lactams, Fluoroquinolones (Gram-negatives) |
| Target Protection | Shield drug target from antibiotic | Qnr proteins (Fluoroquinolones) |
Genetic Players - Resistance Gene Carriers
- Location of Resistance Genes:
- Chromosomal: Intrinsic resistance or acquired via mutation. Generally less mobile.
- Extrachromosomal (Mobile Genetic Elements - MGEs): Primary drivers of rapid resistance spread.
- Plasmids: Self-replicating, circular DNA. Often carry multiple resistance genes (e.g., R-factors).
⭐ Plasmids are pivotal in the horizontal transfer of resistance to multiple antibiotic classes (e.g., genes encoding ESBLs, NDM-1).
- Transposons ("Jumping Genes"): DNA segments that can move within or between genomes (chromosome or plasmid). Carry resistance genes (e.g., $Tn_3$ for ampicillin resistance).
- Integrons: Genetic elements that capture and express gene cassettes, including those encoding antimicrobial resistance. Frequently found within transposons or plasmids.
- Class 1 integrons are the most prevalent in clinical isolates.
- Plasmids: Self-replicating, circular DNA. Often carry multiple resistance genes (e.g., R-factors).
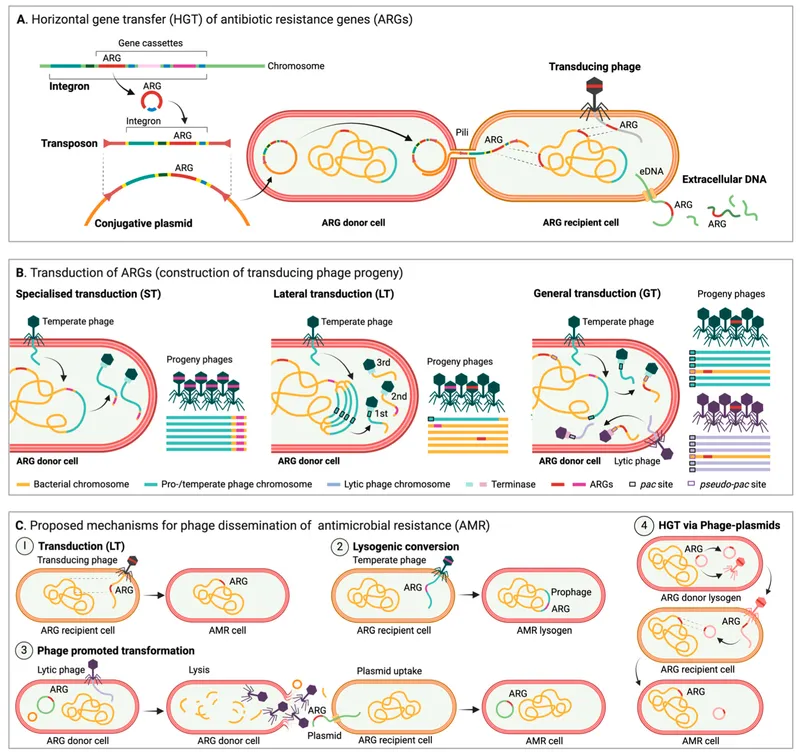
Gene Transfer - Spreading the Bad News
Horizontal Gene Transfer (HGT) is the primary way bacteria acquire and spread antimicrobial resistance genes, distinct from vertical inheritance. Key mechanisms:
- Transformation: Competent bacteria directly uptake naked DNA fragments (e.g., from lysed cells) from their environment.
- Transduction: Bacteriophages (bacterial viruses) accidentally transfer bacterial DNA from one bacterium to another.
- Generalized: Any bacterial gene can be transferred.
- Specialized: Only genes adjacent to the prophage integration site are transferred.
- Conjugation: "Bacterial mating." Direct transfer of genetic material (often plasmids like R-factors) between bacteria via a sex pilus. 📌 F-factor dependent.
⭐ Plasmids carrying multiple resistance genes (MDR plasmids) are a major clinical concern, often spread by conjugation.
High‑Yield Points - ⚡ Biggest Takeaways
- Horizontal Gene Transfer (HGT) via conjugation, transduction, transformation is key for rapid spread.
- Plasmids (R-factors), transposons, and integrons are crucial mobile genetic elements.
- Mechanisms: Enzyme inactivation (β-lactamases like ESBLs, carbapenemases), target modification (PBP2a in MRSA).
- Other mechanisms: Active efflux pumps, decreased permeability, and target bypass.
- Chromosomal mutations (e.g., gyrA for fluoroquinolones) also confer resistance.
- Vancomycin resistance (e.g., vanA gene) in staphylococci is a major concern.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

