Bacteriology

On this page
🦠 Bacteriology Fundamentals: The Bacterial Mastery Blueprint
Master bacteriology, and you unlock the logic behind every infectious disease pattern in clinical medicine. This lesson builds your understanding through 12 major bacterial families, each with distinct virulence mechanisms, clinical presentations, and treatment strategies. You'll learn to recognize patterns that predict diagnosis, understand why certain bacteria cause specific diseases, and develop frameworks for rapid clinical decision-making. The journey moves from foundational microbiology through pathogenesis, clinical correlation, and evidence-based management-transforming isolated facts into integrated clinical expertise.
🧬 The Bacterial Architecture: Structural Command Center
Bacterial classification rests on fundamental structural features that dictate virulence, antibiotic susceptibility, and clinical behavior. Understanding cell wall architecture, metabolic requirements, and morphological patterns enables rapid identification and predicts therapeutic responses.

Gram Staining: The Primary Discriminator
The Gram stain remains the most powerful initial diagnostic tool, separating bacteria into two fundamental groups based on peptidoglycan thickness and outer membrane presence.
-
Gram-Positive Architecture
- Thick peptidoglycan layer (20-80 nm, 60-90% of cell wall mass)
- Teichoic acids embedded in peptidoglycan
- No outer membrane or lipopolysaccharide (LPS)
- Retains crystal violet-iodine complex: purple/blue appearance
- Examples: Staphylococcus, Streptococcus, Bacillus, Clostridium, Corynebacterium, Listeria
- Susceptible to lysozyme and β-lactam antibiotics (target peptidoglycan synthesis)
- Resistance mechanisms: altered penicillin-binding proteins (PBPs), β-lactamase production
-
Gram-Negative Architecture
- Thin peptidoglycan layer (2-7 nm, 10-20% of cell wall mass)
- Outer membrane containing LPS (endotoxin)
- Periplasmic space between inner and outer membranes
- Decolorizes with alcohol, counterstains with safranin: pink/red appearance
- Examples: Enterobacteriaceae, Pseudomonas, Neisseria, Haemophilus, Vibrio, Bordetella, Brucella
- LPS triggers massive cytokine release (TNF-α, IL-1, IL-6) → septic shock
- Outer membrane acts as permeability barrier: intrinsic resistance to many antibiotics
📌 Remember: PEPTIDOGLYCAN THICKNESS PREDICTS STAINING - Gram-positive organisms have 40-60 times more peptidoglycan than Gram-negative bacteria, creating the differential retention of crystal violet that defines the Gram stain.
Morphological Classification Framework
Bacterial shape and arrangement provide immediate diagnostic clues that narrow differential diagnoses before culture results.
| Morphology | Gram-Positive Examples | Gram-Negative Examples | Key Clinical Associations | Distinguishing Features |
|---|---|---|---|---|
| Cocci (spherical) | Staphylococcus (clusters), Streptococcus (chains), Enterococcus (pairs/chains) | Neisseria (diplococci), Moraxella | Pyogenic infections, meningitis, endocarditis | Catalase test separates staph (+) from strep (-) |
| Bacilli (rods) | Bacillus, Clostridium, Corynebacterium, Listeria | Enterobacteriaceae, Pseudomonas, Haemophilus, Bordetella, Brucella | Diarrheal diseases, wound infections, pneumonia | Spore formation (anaerobes), oxidase test (Pseudomonas +) |
| Coccobacilli (short rods) | - | Haemophilus, Brucella, Bordetella | Respiratory infections, zoonoses | Require special growth factors (X, V factors) |
| Spirochetes (spiral) | - | Treponema, Borrelia, Leptospira | Syphilis, Lyme disease, leptospirosis | Dark-field microscopy or special stains required |
| Curved/Comma-shaped | - | Vibrio, Campylobacter | Cholera, gastroenteritis | Oxidase-positive, motile |
⭐ Clinical Pearl: Gram stain from CSF showing Gram-negative diplococci has >95% positive predictive value for N. meningitidis meningitis-immediate empiric ceftriaxone can be life-saving before culture confirmation. Gram-positive diplococci suggest S. pneumoniae (70-80% of bacterial meningitis in adults).
Oxygen Requirements: Metabolic Territories
-
Obligate Aerobes (require O₂)
- Pseudomonas aeruginosa, Mycobacterium tuberculosis, Bordetella pertussis
- Generate ATP via oxidative phosphorylation
- Possess catalase and superoxide dismutase to neutralize reactive oxygen species
- Clinical: Typically cause infections in well-oxygenated tissues (lungs, wounds)
-
Facultative Anaerobes (grow with or without O₂)
- Most clinically significant bacteria: Staphylococcus, Streptococcus, Enterobacteriaceae
- Metabolic flexibility enables colonization of diverse body sites
- Can switch between aerobic respiration and fermentation
- Clinical: Cause infections throughout the body
-
Obligate Anaerobes (killed by O₂)
- Clostridium species, Bacteroides fragilis, Fusobacterium
- Lack catalase and superoxide dismutase
- Clinical: Infections in oxygen-poor environments (abscesses, necrotic tissue, colon)
- Clues: Foul-smelling discharge, gas in tissues, infections below diaphragm
💡 Master This: Anaerobic bacteria cause <10% of bacteremias but account for >60% of intra-abdominal abscesses. Recognition requires understanding tissue oxygen tension-any infection involving bowel perforation, aspiration pneumonia, or necrotizing fasciitis demands anaerobic coverage (metronidazole, carbapenems, β-lactam/β-lactamase inhibitors).
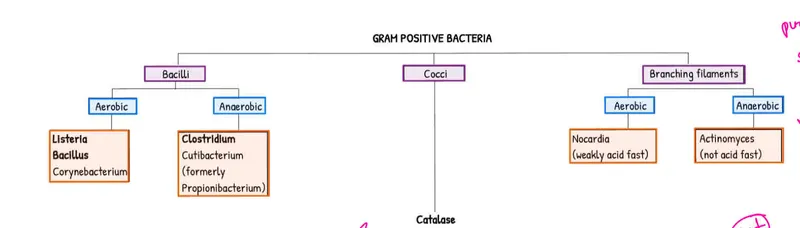
Special Staining and Acid-Fast Organisms
- Acid-Fast Bacteria (Mycobacterium, Nocardia partially)
- Cell wall contains 60-70% mycolic acids (long-chain fatty acids)
- Resist decolorization by acid-alcohol after carbolfuchsin staining
- Ziehl-Neelsen or Kinyoun stains: bright red on blue background
- Clinical significance: M. tuberculosis (TB), M. leprae (leprosy), non-tuberculous mycobacteria (NTM)
- Slow growth (doubling time 15-20 hours for M. tuberculosis vs 20-30 minutes for E. coli)
- Intrinsic resistance to most antibiotics due to impermeable cell wall
- Require prolonged multi-drug therapy (6-24 months)
📌 Remember: ACID-FAST = MYCOLIC ACIDS - The waxy mycolic acid layer makes mycobacteria resistant to Gram staining, most antibiotics, and host immune defenses. This same impermeability requires specialized drugs (rifampin, isoniazid, ethambutol) that can penetrate the lipid-rich barrier.
Understanding these foundational structural principles enables prediction of antibiotic susceptibility patterns, guides empiric therapy selection, and explains why certain bacteria cause specific clinical syndromes. Connect these architectural features through virulence mechanisms to understand pathogenesis patterns.
🧬 The Bacterial Architecture: Structural Command Center
🎯 Virulence Arsenal: Pathogenic Mechanisms Decoded
Bacterial pathogenicity depends on coordinated expression of virulence factors-molecular weapons that enable colonization, immune evasion, tissue invasion, and toxin-mediated damage. Mastering these mechanisms reveals why specific bacteria cause characteristic diseases and predicts therapeutic targets.
Adhesion and Colonization Strategies
Successful infection begins with bacterial attachment to host tissues, mediated by specific adhesins that recognize host cell receptors.
-
Pili (Fimbriae): The Grappling Hooks
- Protein appendages extending from bacterial surface
- Type 1 pili: Mannose-sensitive, bind bladder epithelium (E. coli UTIs)
- P pili: Bind kidney epithelium (E. coli pyelonephritis, 70% of cases)
- Bundle-forming pili: Enteropathogenic E. coli (EPEC) intestinal attachment
- Type IV pili: N. gonorrhoeae, P. aeruginosa, V. cholerae
- Enable twitching motility and microcolony formation
- Antigenic variation in N. gonorrhoeae prevents immune clearance
-
Non-Pili Adhesins
- Protein A: S. aureus binds Fc region of IgG (blocks opsonization)
- M protein: Group A Streptococcus (GAS) binds fibrinogen, prevents complement deposition
- Filamentous hemagglutinin (FHA): B. pertussis binds respiratory epithelium
- Internalins: L. monocytogenes binds E-cadherin (enables placental invasion)
⭐ Clinical Pearl: E. coli strains causing pyelonephritis possess P pili in >90% of cases, while cystitis strains may lack them. This explains why some UTI strains remain confined to the bladder while others ascend to kidneys-P pili binding to Gal-Gal disaccharide on renal epithelium is the critical discriminator.
Toxin-Mediated Pathogenesis
Bacterial toxins cause disease through two fundamental mechanisms: exotoxins (secreted proteins) and endotoxin (structural LPS).
- A-B Toxins: Enzymatic Weapons
- Structure: B (binding) subunit attaches to host receptor; A (active) subunit enters cell
- Diphtheria toxin: Inhibits EF-2 (protein synthesis) → pseudomembrane, myocarditis, neuropathy
- Cholera toxin: Activates adenylate cyclase → ↑ cAMP → massive Cl⁻/H₂O secretion (10-20 L/day diarrhea)
- Shiga toxin (Shigella, EHEC): Cleaves 28S rRNA → protein synthesis inhibition → bloody diarrhea, HUS
- Pertussis toxin: Inhibits Gi protein → ↑ cAMP → lymphocytosis (>20,000-100,000/μL), impaired immunity
- Tetanus toxin: Blocks glycine/GABA release (Renshaw cells) → spastic paralysis, lockjaw
- Botulinum toxin: Blocks ACh release (neuromuscular junction) → flaccid paralysis, descending pattern
📌 Remember: TOXIN MECHANISM PREDICTS CLINICAL SYNDROME - A-B toxins causing ↑ cAMP (cholera, pertussis, anthrax EF) produce secretory diarrhea or edema; those blocking protein synthesis (diphtheria, Shiga) cause tissue necrosis; neurotoxins blocking neurotransmitters (tetanus, botulinum) cause paralysis-mechanism determines disease.
-
Membrane-Damaging Toxins
- α-toxin (S. aureus): Pore-forming, causes hemolysis, tissue necrosis
- Streptolysin O (SLO): S. pyogenes, oxygen-labile, immunogenic (ASO titers)
- Streptolysin S (SLS): Oxygen-stable, non-immunogenic, causes β-hemolysis
- Panton-Valentine leukocidin (PVL): S. aureus, kills WBCs, necrotizing pneumonia
- Phospholipase C (C. perfringens): Lecithinase, destroys cell membranes → gas gangrene
-
Superantigens: Immune System Hijackers
- Bypass normal antigen processing, directly cross-link MHC-II and TCR
- Activate 5-20% of T cells (vs <0.01% for normal antigens)
- Massive cytokine release (IL-2, IFN-γ, TNF-α) → toxic shock syndrome
- TSST-1: S. aureus toxic shock syndrome (tampons, surgical wounds)
- Streptococcal pyrogenic exotoxins (SPE): Scarlet fever, streptococcal toxic shock
- Clinical triad: Fever >38.9°C, hypotension <90 mmHg, diffuse erythroderma with desquamation
💡 Master This: Endotoxin (LPS) from Gram-negative bacteria triggers TLR4 on macrophages, releasing TNF-α and IL-1 within 30-90 minutes. This explains why Gram-negative sepsis causes rapid-onset shock, while Gram-positive sepsis (lacking endotoxin) typically has slower progression. Mortality in Gram-negative septic shock remains 30-50% despite antibiotics because cytokine cascade continues after bacterial killing.
Immune Evasion Mechanisms
-
Capsule: The Invisibility Cloak
- Polysaccharide or polypeptide coating prevents phagocytosis
- S. pneumoniae: >95 serotypes based on capsule; vaccine targets most common
- H. influenzae type b (Hib): Polyribosyl-ribitol phosphate (PRP) capsule
- N. meningitidis: Serogroups A, B, C, W-135, Y based on capsular polysaccharide
- K. pneumoniae: Hypermucoviscous strains cause invasive infections (liver abscess)
- Clinical: Encapsulated bacteria cause >80% of bacterial meningitis in children
-
Antigenic Variation
- N. gonorrhoeae: Pilin gene recombination → evades antibody response
- Borrelia recurrentis: Variable membrane proteins → relapsing fever cycles
- S. pneumoniae: Capsular switching → vaccine escape variants
-
Intracellular Survival
- M. tuberculosis: Prevents phagosome-lysosome fusion, survives in macrophages
- Salmonella typhi: Survives in macrophages → systemic dissemination, carrier state
- Listeria monocytogenes: Escapes phagosome via listeriolysin O, replicates in cytoplasm
- Brucella: Inhibits lysosomal fusion, chronic intracellular infection (months-years)

Invasion and Dissemination
-
Type III Secretion Systems (T3SS): Molecular Syringes
- Inject effector proteins directly into host cells
- Salmonella: SPI-1 invades intestinal epithelium, SPI-2 enables macrophage survival
- Shigella: Invades M cells, spreads laterally through epithelium via actin polymerization
- Yersinia: Yops (Yersinia outer proteins) inhibit phagocytosis, induce apoptosis
- Pseudomonas: ExoS, ExoT, ExoU cause tissue damage and immune suppression
-
Biofilm Formation
- Organized bacterial communities in extracellular matrix
- 100-1000× more resistant to antibiotics than planktonic bacteria
- S. epidermidis: Catheter-associated infections, prosthetic valve endocarditis
- P. aeruginosa: Chronic lung infections in cystic fibrosis (>90% by age 18)
- Enterococcus: Urinary catheter biofilms, difficult to eradicate
⭐ Clinical Pearl: Biofilm infections require 10-100× higher antibiotic concentrations for bacterial killing compared to planktonic infections. This explains why catheter-associated S. epidermidis bacteremia often requires device removal-antibiotics alone achieve cure in only 20-30% of cases when foreign body remains.
| Virulence Factor | Mechanism | Example Bacteria | Clinical Manifestation | Therapeutic Target |
|---|---|---|---|---|
| Capsule | Anti-phagocytic | S. pneumoniae, H. influenzae, N. meningitidis | Meningitis, pneumonia, sepsis | Conjugate vaccines (90-95% efficacy) |
| Protein A | Binds IgG Fc | S. aureus | Prevents opsonization, persistent infection | - |
| M protein | Anti-complement | S. pyogenes | Pharyngitis, rheumatic fever | - |
| IgA protease | Cleaves IgA | N. gonorrhoeae, H. influenzae, S. pneumoniae | Mucosal colonization | - |
| Coagulase | Fibrin clot formation | S. aureus | Abscess formation, protection from phagocytes | - |
| Hyaluronidase | Breaks down connective tissue | S. pyogenes, C. perfringens | Rapid tissue spread (necrotizing fasciitis) | - |
Understanding virulence mechanisms enables prediction of clinical syndromes, guides diagnostic approaches, and reveals therapeutic vulnerabilities. Connect these pathogenic strategies through clinical presentations to recognize disease patterns.
🎯 Virulence Arsenal: Pathogenic Mechanisms Decoded
🔬 Laboratory Identification: Diagnostic Detective Work
Rapid and accurate bacterial identification requires systematic application of culture techniques, biochemical tests, and molecular methods. Master the diagnostic algorithms, and you predict organism identity before final culture results-enabling targeted empiric therapy.
Culture Media Selection Strategy
-
Enriched Media: Supporting Fastidious Growth
- Blood agar: Supports most bacteria, demonstrates hemolysis patterns
- α-hemolysis (partial, green): S. pneumoniae, viridans streptococci
- β-hemolysis (complete, clear): S. pyogenes, S. agalactiae, L. monocytogenes
- γ-hemolysis (none): Enterococcus, S. epidermidis
- Chocolate agar: Heated blood releases factors X (hemin) and V (NAD)
- Required for Haemophilus influenzae, N. gonorrhoeae, N. meningitidis
- Löffler medium: Corynebacterium diphtheriae (metachromatic granules)
- Blood agar: Supports most bacteria, demonstrates hemolysis patterns
-
Selective Media: Isolating Specific Pathogens
- MacConkey agar: Selects Gram-negative, differentiates lactose fermenters
- Lactose+ (pink): E. coli, Klebsiella, Enterobacter
- Lactose- (colorless): Salmonella, Shigella, Proteus, Pseudomonas
- Mannitol salt agar (MSA): Selects Staphylococcus (7.5% NaCl)
- Mannitol+ (yellow): S. aureus (>90% of strains)
- Mannitol- (red): Coagulase-negative staphylococci
- Thiosulfate-citrate-bile salts-sucrose (TCBS): Vibrio species (yellow colonies)
- Xylose-lysine-deoxycholate (XLD): Salmonella (black centers from H₂S), Shigella (red)
- Bordet-Gengou agar: Bordetella pertussis (requires 3-7 days)
- Löwenstein-Jensen medium: Mycobacterium tuberculosis (requires 3-8 weeks)
- MacConkey agar: Selects Gram-negative, differentiates lactose fermenters
📌 Remember: SELECTIVE MEDIA = INHIBIT COMPETITION - High salt (MSA for staph), bile salts (MacConkey for Gram-negatives), antibiotics (Thayer-Martin for Neisseria) create hostile environments that only target organisms tolerate, enabling isolation from mixed flora.

Biochemical Test Algorithms
-
Catalase Test: Staph vs Strep
- Detects catalase enzyme (converts H₂O₂ to H₂O + O₂)
- Positive (bubbles): Staphylococcus, Listeria, Bacillus
- Negative: Streptococcus, Enterococcus
- Mechanism: Catalase protects aerobes/facultative anaerobes from oxidative damage
-
Coagulase Test: S. aureus vs CoNS
- Bound coagulase (clumping factor): Slide test, detects fibrinogen binding (5 minutes)
- Free coagulase: Tube test, converts fibrinogen to fibrin (4 hours)
- Positive: S. aureus (>99% sensitivity/specificity)
- Negative: Coagulase-negative staphylococci (S. epidermidis, S. saprophyticus)
-
Oxidase Test: Key for Gram-Negatives
- Detects cytochrome c oxidase
- Positive (purple within 10 seconds): Pseudomonas, Neisseria, Vibrio, Campylobacter, Aeromonas, HACEK
- Negative: Enterobacteriaceae, Stenotrophomonas
- Clinical utility: Narrows Gram-negative rod differential immediately
💡 Master This: The oxidase test achieves 100% negative predictive value for Enterobacteriaceae-any oxidase-positive Gram-negative rod is NOT E. coli, Salmonella, Shigella, or Klebsiella. This single test immediately redirects diagnostic thinking toward Pseudomonas, Vibrio, or non-fermenting Gram-negatives.
Advanced Identification Methods
-
Enterobacteriaceae Differentiation
- Triple sugar iron (TSI) agar: Glucose, lactose, sucrose fermentation + H₂S production
- Indole test: Tryptophan metabolism (E. coli positive, Klebsiella negative)
- Urease test: Proteus (rapid, <6 hours), Klebsiella (slower)
- Citrate utilization: Klebsiella, Enterobacter, Citrobacter positive
- Motility: Proteus (swarming), Salmonella (motile), Shigella (non-motile)
-
Streptococcal Classification
- Lancefield grouping: Carbohydrate C antigen (Groups A, B, C, D, G)
- Group A: S. pyogenes (GAS) - bacitracin sensitive
- Group B: S. agalactiae (GBS) - CAMP test positive, hippurate hydrolysis positive
- Group D: Enterococcus - bile esculin positive, grows in 6.5% NaCl
- Optochin sensitivity: S. pneumoniae (sensitive), viridans strep (resistant)
- Bile solubility: S. pneumoniae (positive), viridans strep (negative)
- Lancefield grouping: Carbohydrate C antigen (Groups A, B, C, D, G)
| Organism | Hemolysis | Catalase | Coagulase | Oxidase | Special Test | Key Clinical Clue |
|---|---|---|---|---|---|---|
| S. aureus | β (variable) | + | + | - | Mannitol+ | Golden colonies, abscesses |
| S. pyogenes (GAS) | β | - | - | - | Bacitracin sensitive | Pharyngitis, ASO+ |
| S. pneumoniae | α | - | - | - | Optochin sensitive | Pneumonia, meningitis |
| Enterococcus | γ or α | - | - | - | Bile esculin+, 6.5% NaCl+ | UTI, endocarditis |
| E. coli | - | + | - | - | Indole+, lactose+ | UTI, diarrhea |
| Pseudomonas | - | + | - | + | Oxidase+, pyocyanin | Blue-green pus, CF |
⭐ Clinical Pearl: S. pneumoniae demonstrates >95% sensitivity to optochin (ethylhydrocupreine) while viridans streptococci are resistant-this single disk test differentiates the leading cause of community-acquired pneumonia from oral commensals. Bile solubility provides 100% specificity as a confirmatory test.
Molecular and Rapid Diagnostics
-
Nucleic Acid Amplification Tests (NAATs)
- PCR-based methods: Detect bacterial DNA/RNA within 1-4 hours
- Multiplex panels: Identify 15-30 pathogens simultaneously from blood, CSF, respiratory samples
- N. gonorrhoeae/C. trachomatis: Urine NAAT sensitivity >95% (vs culture 80-85%)
- GeneXpert MTB/RIF: M. tuberculosis + rifampin resistance in 2 hours (sensitivity 88%, specificity 99%)
- Blood culture PCR panels: Reduce time to pathogen ID by 24-48 hours
-
MALDI-TOF Mass Spectrometry
- Matrix-assisted laser desorption/ionization time-of-flight
- Identifies bacteria to species level in <5 minutes from colonies
- Accuracy >95% for common pathogens
- Cost-effective, replacing many biochemical tests
- Limitations: Requires pure culture, database-dependent
-
Antigen Detection
- Rapid strep test: GAS pharyngitis (sensitivity 85-90%, specificity >95%)
- S. pneumoniae urinary antigen: Sensitivity 70-80% for bacteremic pneumonia
- Legionella urinary antigen: Detects serogroup 1 (causes 80-90% of cases)
- Cryptococcal antigen: CSF/serum for meningitis (sensitivity >95%)

💡 Master This: Blood culture positivity occurs in 10-30% of community-acquired pneumonia, 30-50% of hospital-acquired pneumonia, and >90% of endocarditis cases. Time to positivity predicts bacteremia severity-detection within 12 hours suggests high-grade bacteremia (>100 CFU/mL), while >48 hours indicates low-grade or contaminant. Understanding these kinetics guides clinical interpretation.
Systematic application of these diagnostic tools enables rapid organism identification, guides empiric therapy selection, and predicts antimicrobial susceptibility patterns. Connect laboratory findings through clinical presentations to recognize infection patterns.
🔬 Laboratory Identification: Diagnostic Detective Work
🏥 Clinical Syndromes: Pattern Recognition Framework
Bacterial infections produce characteristic clinical syndromes determined by organism virulence, tissue tropism, and host immune response. Mastering "see X, think Y" correlations enables rapid diagnosis and appropriate empiric therapy before culture confirmation.
Respiratory Tract Infections
- Community-Acquired Pneumonia (CAP) Patterns
- S. pneumoniae (30-50% of CAP)
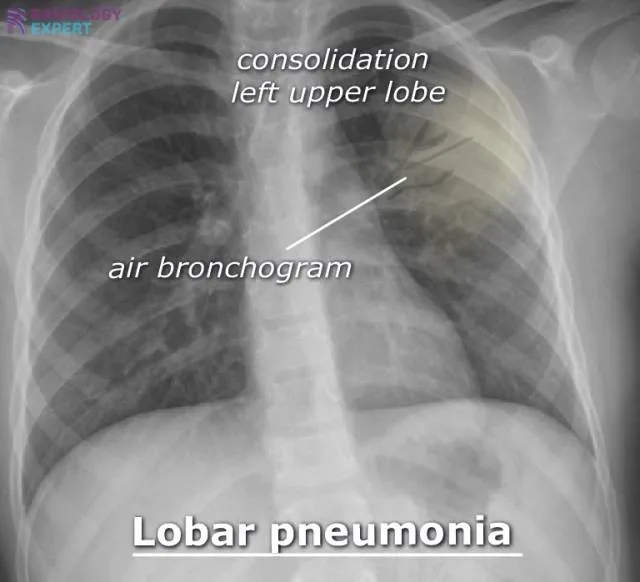
- Acute onset, single shaking chill, rust-colored sputum
- Lobar consolidation on CXR, air bronchograms
- Mortality 5-15% overall, 30-40% if bacteremic
- H. influenzae (5-20% of CAP)
- COPD patients, smokers
- Bronchopneumonia pattern, purulent sputum
- Non-typeable strains (lacking capsule) most common
- Mycoplasma pneumoniae (10-20% of CAP)
- Young adults, gradual onset, dry cough
- "Walking pneumonia," extrapulmonary manifestations (25%)
- Cold agglutinins positive (50% of cases)
- Legionella pneumophila (2-15% of CAP)
- Hyponatremia (<130 mEq/L in 50%), elevated transaminases
- Diarrhea (30-50%), confusion, relative bradycardia
- Mortality 10-15%, higher in nosocomial cases
- S. pneumoniae (30-50% of CAP)
📌 Remember: CURB-65 PREDICTS PNEUMONIA MORTALITY - Confusion, Uremia (BUN >20 mg/dL), Respiratory rate ≥30, Blood pressure <90/60, age ≥65. Score 0-1: <3% mortality (outpatient), 2: 9% (consider admission), 3-5: 15-40% (ICU consideration).
- Pertussis: The 100-Day Cough
- Bordetella pertussis
- Three stages: Catarrhal (1-2 weeks), paroxysmal (2-8 weeks), convalescent (weeks-months)
- Paroxysmal cough with inspiratory whoop, post-tussive emesis
- Lymphocytosis >20,000/μL (sometimes >100,000/μL)
- Complications: Pneumonia, seizures, encephalopathy (infants <6 months)
- PCR sensitivity >90% in first 3 weeks, culture requires Bordet-Gengou agar
Central Nervous System Infections
- Bacterial Meningitis: The Time-Critical Emergency
- S. pneumoniae (50% of cases, adults)
- Mortality 20-30%, neurologic sequelae 30-50%
- CSF: WBC 1000-10,000/μL, glucose <40 mg/dL, protein >100 mg/dL
- Risk factors: Pneumococcal pneumonia, otitis, sinusitis, CSF leak, asplenia
- N. meningitidis (25% of cases, adolescents/young adults)
- Petechial/purpuric rash (50-80%), fulminant sepsis (Waterhouse-Friderichsen)
- Mortality 10-15% with treatment, >90% without
- Close contacts require prophylaxis (rifampin, ciprofloxacin, ceftriaxone)
- L. monocytogenes (5-10% of cases)
- Neonates, elderly >50 years, immunocompromised
- Brainstem involvement (rhombencephalitis), cranial nerve palsies
- Ampicillin required (cephalosporins ineffective)
- H. influenzae type b (<5% since Hib vaccine)
- Unvaccinated children, immunocompromised adults
- Vaccine efficacy >95%
- S. pneumoniae (50% of cases, adults)
⭐ Clinical Pearl: Antibiotic administration within 1 hour of ED arrival reduces bacterial meningitis mortality by 30-40%. Blood cultures should NOT delay antibiotics-obtain cultures, then immediately give ceftriaxone 2 g IV (plus vancomycin if pneumococcus suspected, plus ampicillin if Listeria risk). LP can follow within 2-4 hours without compromising CSF diagnostic yield.
Gastrointestinal Infections
-
Diarrheal Disease Classification
- Secretory (watery, non-inflammatory)
- V. cholerae: Rice-water stools, 10-20 L/day, mortality 50% untreated (now <1% with ORT)
- Enterotoxigenic E. coli (ETEC): Traveler's diarrhea, heat-labile (LT) and heat-stable (ST) toxins
- C. perfringens: Food poisoning, 8-16 hour incubation, self-limited
- Inflammatory (bloody, dysentery)
- Shigella: Bloody diarrhea, tenesmus, 10-100 organisms sufficient for infection
- Enterohemorrhagic E. coli (EHEC O157:H7): Shiga toxin, HUS risk 5-10% (children)
- Salmonella enteritidis: Inflammatory diarrhea, fever, usually self-limited (3-7 days)
- Campylobacter jejuni: Most common bacterial diarrhea (US), Guillain-Barré risk 1:1000
- Secretory (watery, non-inflammatory)
-
Enteric Fever: Systemic Salmonellosis
- Salmonella typhi (typhoid fever)
- Stepwise fever increase over 3-4 days, relative bradycardia
- Rose spots (faint salmon-colored macules on trunk, 30% of cases)
- Complications: Intestinal perforation (3%), hemorrhage, chronic carrier state (1-5%)
- Blood culture positive in first week (90%), stool culture positive after first week (>90%)
💡 Master This: Hemolytic uremic syndrome (HUS) develops in 5-10% of EHEC O157:H7 infections, typically 5-10 days after diarrhea onset. Triad: Microangiopathic hemolytic anemia, thrombocytopenia <150,000/μL, acute kidney injury. Antibiotics may INCREASE HUS risk by 2-3× (promote toxin release), so avoid in suspected EHEC-supportive care only.
Skin and Soft Tissue Infections
-
Cellulitis and Erysipelas
- S. pyogenes (GAS): Erysipelas (superficial, raised borders, facial)
- S. aureus: Cellulitis (deeper, indistinct borders, extremities)
- MRSA: Community-acquired (CA-MRSA) often presents as recurrent abscesses
- PVL toxin in >90% of CA-MRSA
- Risk factors: Contact sports, incarceration, MSM, IV drug use
-
Necrotizing Fasciitis: The Surgical Emergency
- Type I: Polymicrobial (anaerobes + Enterobacteriaceae)
- Diabetes, post-surgical, perirectal
- Mortality 25-35%
- Type II: Monomicrobial S. pyogenes (GAS)
- "Flesh-eating bacteria," rapid progression (hours)
- Streptococcal toxic shock syndrome in 50%
- Mortality 30-70% despite treatment
- LRINEC score: Lab risk indicator for necrotizing fasciitis
- Score ≥6: >50% probability, immediate surgical exploration
- CRP, WBC, hemoglobin, sodium, creatinine, glucose
- Type I: Polymicrobial (anaerobes + Enterobacteriaceae)

⭐ Clinical Pearl: Pain out of proportion to physical findings is the hallmark of necrotizing fasciitis, present in >90% of cases. Crepitus (subcutaneous gas) is specific but only present in **30-40%
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app

















