Oncology

On this page
🧬 Cancer Biology: The Cellular Rebellion Decoded
Cancer begins when a single cell breaks free from the body's regulatory controls, accumulating mutations that transform it from cooperative citizen to invasive threat. You'll trace this cellular rebellion from its genetic origins through the hallmarks that define malignancy, understanding how oncogenes accelerate growth while tumor suppressors fail, how cancer cells metastasize and evade immune surveillance, and how modern oncology translates these mechanisms into targeted therapies. By integrating molecular biology with clinical pattern recognition, you'll build the diagnostic and therapeutic framework that transforms oncology from memorization into strategic medicine.

📌 Remember: HANGERS for Cancer Hallmarks - Hangiogenesis, Apoptosis resistance, No growth inhibition, Growth signals sustained, Energy metabolism reprogrammed, Replicative immortality, Spreading/invasion. Each represents a fundamental capability that normal cells lack but cancer cells systematically acquire.
The transformation from normal to malignant involves 6-10 driver mutations accumulated over 10-30 years, with each mutation conferring specific survival advantages. Cancer cells demonstrate 100-1000x higher mutation rates than normal cells, creating genetic instability that drives continued evolution and treatment resistance.
- Sustained Proliferative Signaling
- Growth factor independence: >90% of cancers show autonomous growth signals
- Oncogene activation: RAS mutations in 30% of cancers, MYC amplification in 15%
- Normal cells require external growth signals every 12-24 hours
- Cancer cells generate internal signals continuously
- Evading Growth Suppressors
- p53 mutations: Present in >50% of human cancers
- RB pathway disruption: >90% of cancers lose G1/S checkpoint control
- Normal cells arrest at checkpoints for 2-6 hours for DNA repair
- Cancer cells bypass checkpoints in <30 minutes
⭐ Clinical Pearl: p53 status predicts chemotherapy response-wild-type p53 tumors show 60-80% response rates to DNA-damaging agents, while mutant p53 tumors respond in only 20-30% of cases. This explains why TP53 sequencing guides treatment selection in many protocols.
| Hallmark | Normal Cell Response | Cancer Cell Adaptation | Clinical Significance | Therapeutic Target |
|---|---|---|---|---|
| Growth Signals | Requires external factors | Autonomous production | Predicts aggressive behavior | Growth factor receptors |
| Growth Inhibition | Responds to contact inhibition | Ignores density signals | Enables local invasion | Cell cycle checkpoints |
| Apoptosis | Activates with DNA damage | Resists death signals | Chemotherapy resistance | Pro-apoptotic pathways |
| Replicative Immortality | 50-70 divisions max | Unlimited divisions | Metastatic potential | Telomerase inhibitors |
| Angiogenesis | Quiescent vasculature | Continuous vessel formation | Tumor size >2mm | Anti-VEGF therapy |
The "two-hit hypothesis" governs tumor suppressor inactivation, requiring both alleles to be lost for complete functional disruption. This explains the 10-20 year latency period for many hereditary cancers and why germline mutation carriers develop tumors 2-3 decades earlier than sporadic cases.
Connect these fundamental cellular changes through carcinogenesis mechanisms to understand how environmental factors trigger the transformation cascade that creates clinical cancer.
🧬 Cancer Biology: The Cellular Rebellion Decoded
⚡ Carcinogenesis: The Transformation Timeline
📌 Remember: IPP for Carcinogenesis Phases - Initiation (DNA damage, irreversible, seconds-minutes), Promotion (clonal expansion, reversible, months-years), Progression (malignant transformation, irreversible, years-decades). Each phase requires different interventions and has distinct clinical implications.
The initiation phase occurs within minutes-hours of carcinogen exposure, creating permanent DNA alterations in <0.1% of exposed cells. However, initiated cells remain dormant without promotional stimuli, explaining why single exposures rarely cause cancer but repeated exposures over 10-20 years dramatically increase risk.
- Initiation Phase Characteristics
- DNA damage occurs within 2-6 hours of carcinogen exposure
- Affects 1 in 10,000-100,000 exposed cells
- Irreversible genetic alterations in critical genes
- p53 mutations: UV radiation causes C→T transitions at dipyrimidine sites
- KRAS mutations: Chemical carcinogens create G→T transversions
- Requires metabolic activation for >80% of chemical carcinogens
- Promotion Phase Dynamics
- Clonal expansion increases initiated cell population 100-1000x
- Requires continuous exposure for months-years
- Reversible if promotional stimuli removed
- Smoking cessation: Lung cancer risk decreases 50% within 10 years
- Alcohol reduction: Liver cancer risk drops 30% within 5 years
⭐ Clinical Pearl: The "field cancerization" concept explains why 20-30% of patients develop second primary tumors-entire tissue fields acquire pre-malignant changes from chronic carcinogen exposure. This drives surveillance protocols requiring lifelong monitoring of the entire at-risk anatomical region.
| Carcinogen Type | Mechanism | Latency Period | Cancer Association | Prevention Strategy |
|---|---|---|---|---|
| UV Radiation | Direct DNA damage | 10-30 years | Skin cancer (90% cases) | Sunscreen, protective clothing |
| Tobacco | Metabolic activation | 20-40 years | Lung cancer (85% cases) | Smoking cessation programs |
| Alcohol | Acetaldehyde formation | 15-25 years | Liver cancer (60% cases) | Consumption reduction |
| HPV | Viral oncoproteins | 10-20 years | Cervical cancer (99% cases) | Vaccination, screening |
| Asbestos | Chronic inflammation | 20-50 years | Mesothelioma (80% cases) | Occupational protection |
💡 Master This: Tumor heterogeneity emerges during progression as different cell clones acquire distinct mutations. This explains why single-agent therapies fail in >90% of advanced cancers-multiple resistant clones exist before treatment begins, requiring combination approaches targeting different pathways simultaneously.
Epigenetic modifications contribute equally to genetic mutations in carcinogenesis, with DNA methylation silencing tumor suppressors in >70% of cancers. Unlike genetic changes, epigenetic alterations remain potentially reversible, creating therapeutic opportunities through demethylating agents and histone deacetylase inhibitors.
Connect carcinogenesis understanding through oncogenes and tumor suppressors to master how specific genes control the transformation process and guide targeted therapeutic interventions.
⚡ Carcinogenesis: The Transformation Timeline
🎯 Oncogenes & Tumor Suppressors: The Genetic Gatekeepers

📌 Remember: POWER for Oncogene Functions - Proliferation signals, Organelle biogenesis, Wound healing, Energy metabolism, Repair mechanisms. When activated inappropriately, these normal cellular functions become malignant drivers requiring single allele alteration for transformation.
Oncogenes function as cellular accelerators-normally promoting growth when appropriate, but causing uncontrolled proliferation when mutated or overexpressed. Unlike tumor suppressors requiring both alleles lost, oncogenes need only single allele activation to contribute to malignancy, explaining their dominant inheritance pattern in familial cancer syndromes.
- Major Oncogene Categories
- Growth Factor Receptors: EGFR amplified in 30% of lung cancers
- Signal Transduction: RAS mutations in 30% of all cancers
- Transcription Factors: MYC amplification in 15% of cancers
- HER2 overexpression: 20-25% of breast cancers, predicts trastuzumab response
- BCR-ABL fusion: >95% of CML cases, target for imatinib therapy
- Cell Cycle Regulators: Cyclin D1 amplification in 50% of breast cancers
- Tumor Suppressor Mechanisms
- p53: "Guardian of genome" - >50% of cancers show mutations
- RB: Controls G1/S checkpoint - >90% of cancers lose function
- APC: Regulates Wnt signaling - >80% of colorectal cancers
- BRCA1/2: DNA repair genes - 5-10% of breast/ovarian cancers
- VHL: Hypoxia response - >90% of clear cell renal cancers
⭐ Clinical Pearl: p53 status determines chemotherapy sensitivity patterns-wild-type p53 tumors respond to DNA-damaging agents in 70-80% of cases through intact apoptotic pathways, while p53-mutant tumors show <30% response rates but may be more sensitive to cell cycle inhibitors and immunotherapy.
| Gene | Type | Normal Function | Cancer Alteration | Clinical Significance | Targeted Therapy |
|---|---|---|---|---|---|
| RAS | Oncogene | Growth signal transduction | Point mutations (30%) | Poor prognosis marker | KRAS G12C inhibitors |
| p53 | Tumor Suppressor | DNA damage checkpoint | Loss of function (50%) | Chemotherapy resistance | MDM2 inhibitors |
| EGFR | Oncogene | Growth factor response | Amplification/mutation | Treatment target | Erlotinib, cetuximab |
| BRCA1/2 | Tumor Suppressor | DNA repair | Germline mutations | PARP inhibitor sensitivity | Olaparib, rucaparib |
| MYC | Oncogene | Cell cycle progression | Amplification (15%) | Aggressive behavior | BET inhibitors |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | |||||
| flowchart TD |
Start["🧫 Normal Cell
• Baseline state• Homeostasis kept"]
Onco["🧬 Oncogene (+)
• Gene activation• Mutational event"]
IncrPro["📈 ⬆️ Proliferation
• Rapid division• Clonal growth"]
SuppIntact["🛡️ TS Intact?
• Protein active• Checkpoint check"]
Senescence["⏹️ Senescence
• Growth arrest• Programmed death"]
Malignant["⚠️ Malignancy
• Transformation• Cancer cell"]
SuppLoss["📉 TS Loss?
• Gene deletion• Loss of brakes"]
GrowthLoss["🚫 Control Loss
• Unchecked growth• Genomic damage"]
NormalCycle["✅ Normal Cycle
• Regular mitosis• No pathology"]
Start --> Onco Onco -->|Yes| IncrPro Onco -->|No| SuppLoss IncrPro --> SuppIntact SuppIntact -->|Yes| Senescence SuppIntact -->|No| Malignant SuppLoss -->|Yes| GrowthLoss SuppLoss -->|No| NormalCycle GrowthLoss --> Malignant
style Start fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252 style Onco fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style IncrPro fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style SuppIntact fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Senescence fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style Malignant fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style SuppLoss fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style GrowthLoss fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style NormalCycle fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534
The **"two-hit hypothesis"** explains tumor suppressor inactivation requiring **both alleles** lost for complete functional disruption. **Germline mutation carriers** inherit **one defective copy**, requiring only **single somatic event** for complete loss, explaining **earlier onset** and **higher penetrance** in hereditary cancer syndromes.
> 💡 **Master This**: **Synthetic lethality** exploits cancer-specific genetic defects-**BRCA-deficient** tumors cannot survive **PARP inhibition** because they lack **homologous recombination** backup for DNA repair. This principle enables **precision medicine** approaches targeting cancer vulnerabilities while sparing normal cells.
**Tumor suppressor loss** follows predictable patterns: **LOH (loss of heterozygosity)** occurs in **>80%** of cancers, with **chromosome 17p** (p53 locus) deleted in **>60%** of cases. **Microsatellite analysis** can detect these losses, providing prognostic information and therapeutic guidance.
**Oncogene addiction** describes cancer cell dependence on specific activated oncogenes for survival. **BCR-ABL-positive** leukemias show **>95%** response rates to **tyrosine kinase inhibitors**, while **EGFR-mutant** lung cancers respond to **erlotinib** in **70-80%** of cases, demonstrating therapeutic vulnerability created by oncogene dependence.
Connect oncogene and tumor suppressor understanding through **metastasis and immune evasion** to master how genetic alterations enable cancer spread and resistance to host defenses.
🎯 Oncogenes & Tumor Suppressors: The Genetic Gatekeepers
🌊 Metastasis & Immune Evasion: The Escape Artists
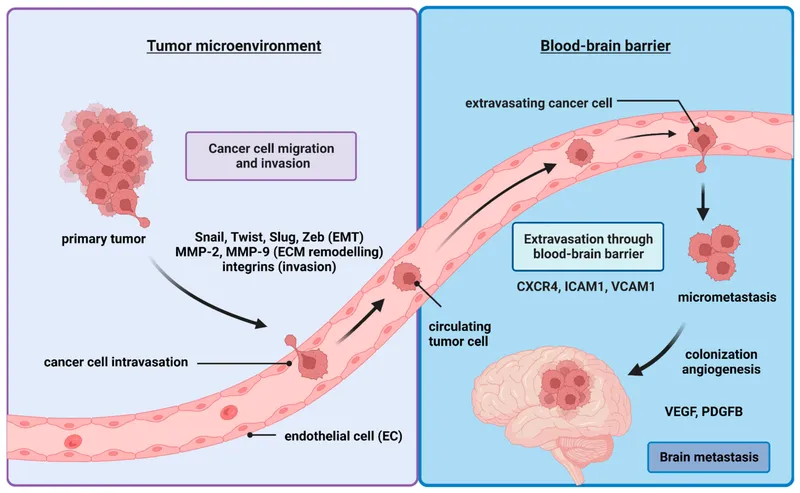
📌 Remember: ISLANDS for Metastatic Steps - Invasion through basement membrane, Survival in circulation, Lodging in capillaries, Angiogenesis induction, Niche establishment, Dormancy escape, Secondary tumor growth. Each step requires specific molecular capabilities and represents potential therapeutic intervention points.
The invasion-metastasis cascade requires systematic acquisition of capabilities that normal cells completely lack. Epithelial-mesenchymal transition (EMT) initiates the process, with cancer cells losing E-cadherin expression in >80% of carcinomas and gaining mesenchymal markers like vimentin and N-cadherin.
- Local Invasion Mechanisms
- Matrix metalloproteinase (MMP) overexpression: >90% of invasive cancers
- Basement membrane degradation: Type IV collagen breakdown
- Cell adhesion loss: E-cadherin reduction in >70% of carcinomas
- MMP-2 and MMP-9: Degrade type IV collagen and gelatin
- TIMP (tissue inhibitors): Lost in >60% of metastatic cancers
- Circulation Survival Strategies
- Anoikis resistance: Survival without matrix attachment
- Platelet aggregation: >80% of CTCs associate with platelets
- Immune evasion: PD-L1 expression protects from T-cell killing
- Circulating tumor cells (CTCs): 1-100 per 7.5mL blood in metastatic disease
- CTC survival: <0.01% establish micrometastases
⭐ Clinical Pearl: Liquid biopsies detecting CTCs and circulating tumor DNA (ctDNA) provide real-time monitoring of metastatic burden. ctDNA levels correlate with tumor volume and predict treatment response 2-8 weeks before imaging changes, enabling earlier therapeutic adjustments.
| Metastatic Site | Primary Tumor Preference | Molecular Basis | Survival Impact | Therapeutic Approach |
|---|---|---|---|---|
| Liver | Colorectal (60-70%), Pancreas | Portal circulation | 6-24 months median | Hepatic resection, ablation |
| Lung | Breast (20-30%), Sarcoma | Pulmonary circulation | 12-36 months median | Pulmonary metastasectomy |
| Bone | Breast (70%), Prostate (90%) | RANKL/OPG pathway | 24-48 months median | Bisphosphonates, denosumab |
| Brain | Lung (40%), Melanoma (60%) | Blood-brain barrier disruption | 3-12 months median | Stereotactic radiosurgery |
| Peritoneum | Ovarian (>90%), Gastric | Transcoelomic spread | 6-18 months median | Cytoreductive surgery, HIPEC |
- Immune Evasion Strategies
- Checkpoint molecule upregulation: PD-L1, CTLA-4, LAG-3
- Immunosuppressive cytokine production: TGF-β, IL-10, VEGF
- Regulatory T-cell recruitment: Tregs comprise >20% of tumor-infiltrating lymphocytes
- Tumor-associated macrophages (TAMs): M2 polarization promotes metastasis
- Myeloid-derived suppressor cells (MDSCs): Suppress T-cell and NK-cell function
- Antigen presentation defects: HLA class I loss in >50% of cancers
- Complement system activation: C5a promotes metastatic seeding
💡 Master This: Metastatic organotropism follows the "seed and soil" hypothesis-specific cancer types preferentially metastasize to particular organs based on molecular compatibility. Breast cancer bone metastases express RANKL receptors, while brain metastases upregulate blood-brain barrier penetration factors, explaining site-specific therapeutic requirements.
Tumor dormancy represents a critical metastatic phase where micrometastases remain growth-arrested for months-decades before reactivating. >30% of breast cancer patients develop late recurrences >10 years after primary treatment, highlighting the clinical importance of dormant cell populations.
Immunotherapy resistance emerges through adaptive immune resistance-initially responsive tumors acquire secondary mutations in antigen presentation machinery or interferon signaling pathways. Combination immunotherapy targeting multiple checkpoints shows improved response rates of 40-60% compared to single-agent approaches achieving 15-25% responses.
Connect metastasis and immune evasion concepts through clinical applications to master how these biological principles guide diagnostic approaches, staging systems, and therapeutic strategies in oncological practice.
🌊 Metastasis & Immune Evasion: The Escape Artists
🎪 Pattern Recognition: The Cancer Detective's Toolkit
📌 Remember: MATCH for Cancer Pattern Recognition - Morphology assessment, Architectural patterns, Tissue markers (IHC), Clinical context, Hereditary factors. Each element provides diagnostic clues that must be integrated systematically to avoid anchoring bias and premature closure.
Histological pattern recognition forms the foundation of cancer diagnosis, with architectural features and cellular morphology providing the first diagnostic clues. Well-differentiated tumors retain >70% of normal tissue architecture, while poorly differentiated cancers show <30% structural organization, directly correlating with prognosis and treatment response.
- Adenocarcinoma Recognition Patterns
- Glandular architecture: >50% of tumor shows gland formation
- Mucin production: PAS-positive intracellular or extracellular mucin
- Nuclear features: Enlarged nuclei with prominent nucleoli
- Signet ring variant: >50% cells show intracytoplasmic mucin
- Papillary pattern: Fibrovascular cores with epithelial lining
- Squamous Cell Carcinoma Patterns
- Keratinization: Intercellular bridges and keratin pearl formation
- Stratified architecture: Basal-to-surface differentiation gradient
- Desmoplastic response: >80% show stromal fibrosis
- Well-differentiated: >75% keratinization, better prognosis
- Poorly differentiated: <25% keratinization, aggressive behavior
⭐ Clinical Pearl: Immunohistochemistry panels resolve diagnostic uncertainty in >90% of cases where H&E morphology alone is insufficient. TTF-1 positive/CK7 positive suggests lung adenocarcinoma, while CK20 positive/CDX2 positive indicates colorectal origin with >95% specificity.
| Cancer Type | Key Morphological Features | Diagnostic IHC Markers | Molecular Signatures | Clinical Behavior |
|---|---|---|---|---|
| Lung Adenocarcinoma | Glandular/papillary pattern | TTF-1+, CK7+ | EGFR, KRAS mutations | 5-year survival: 15-60% |
| Breast Ductal Carcinoma | Solid nests, desmoplasia | CK7+, GATA3+ | HER2, ER/PR status | 5-year survival: 85-95% |
| Colorectal Adenocarcinoma | Cribriform glands | CK20+, CDX2+ | APC, KRAS mutations | 5-year survival: 65-90% |
| Prostate Adenocarcinoma | Small acinar pattern | PSA+, NKX3.1+ | TMPRSS2-ERG fusion | 5-year survival: >95% |
| Melanoma | Nested/single cell pattern | S-100+, Melan-A+ | BRAF, NRAS mutations | 5-year survival: 20-95% |
- Actionable Molecular Patterns
- EGFR mutations: Tyrosine kinase inhibitor sensitivity in >70% of cases
- HER2 amplification: Trastuzumab response in >80% of breast cancers
- BRAF V600E: Vemurafenib response in >90% of melanomas
- Microsatellite instability (MSI-H): Immunotherapy response in >40% of cases
- PD-L1 expression: Checkpoint inhibitor response predictor
- BRCA1/2 mutations: PARP inhibitor sensitivity in >60% of cases
- ALK rearrangements: Crizotinib response in >90% of lung cancers
💡 Master This: Tumor heterogeneity creates diagnostic challenges-primary tumors and metastases show concordant biomarkers in only 70-80% of cases. Liquid biopsies detecting circulating tumor DNA provide real-time molecular profiling that captures tumor evolution and resistance mechanisms better than single tissue samples.
Clinical pattern integration requires synthesizing patient demographics, presentation patterns, and risk factors with pathological findings. Age-specific cancer patterns show distinct distributions-pediatric cancers are >80% hematological, while adult cancers are >90% solid tumors, fundamentally altering diagnostic approaches.
Hereditary cancer syndromes create recognizable clinical patterns requiring systematic family history assessment. Lynch syndrome patients develop colorectal cancer at median age 45 versus sporadic cases at median age 70, with >90% showing microsatellite instability and increased immunotherapy sensitivity.
Connect pattern recognition mastery through systematic diagnostic approaches to understand how clinical frameworks enable rapid cancer identification and precision treatment selection in complex oncological scenarios.
🎪 Pattern Recognition: The Cancer Detective's Toolkit
⚖️ Treatment Paradigms: The Precision Medicine Revolution

📌 Remember: TARGET for Precision Treatment Selection - Tumor genetics, Anatomical staging, Response biomarkers, Germline factors, Eastern Cooperative Oncology Group (ECOG) performance status, Toxicity profile. Each factor influences treatment feasibility and expected outcomes in evidence-based therapeutic decision-making.
Biomarker-driven therapy has transformed cancer treatment, with >60% of FDA-approved cancer drugs now requiring companion diagnostics. Targeted therapies achieve response rates of 60-90% in biomarker-selected populations compared to 15-30% with traditional chemotherapy in unselected patients.
- Targeted Therapy Categories
- Tyrosine Kinase Inhibitors: EGFR, ALK, ROS1 targeting
- Monoclonal Antibodies: HER2, VEGF, CD20 targeting
- Cell Cycle Inhibitors: CDK4/6, PARP, mTOR inhibition
- Imatinib for BCR-ABL: >95% complete cytogenetic response in CML
- Trastuzumab for HER2+ breast cancer: 50% reduction in recurrence risk
- Immunotherapy Agents: PD-1/PD-L1, CTLA-4 checkpoint inhibitors
- Antibody-Drug Conjugates: Targeted delivery of cytotoxic payloads
⭐ Clinical Pearl: Combination therapy strategies overcome single-agent resistance by targeting multiple pathways simultaneously. CDK4/6 inhibitors plus endocrine therapy in HR-positive breast cancer improve progression-free survival from 14 months to 28 months, demonstrating synergistic efficacy without overlapping toxicities.
| Treatment Modality | Mechanism | Response Rate | Duration of Response | Key Toxicities | Biomarker Requirements |
|---|---|---|---|---|---|
| Targeted Therapy | Specific pathway inhibition | 60-90% | 8-24 months | Skin rash, diarrhea | Mutation testing required |
| Immunotherapy | Immune system activation | 20-40% | >24 months | Immune-related AEs | PD-L1, MSI testing |
| Chemotherapy | DNA damage/mitotic inhibition | 15-30% | 4-8 months | Myelosuppression, nausea | None typically required |
| Hormone Therapy | Hormone receptor blockade | 30-60% | 12-36 months | Hot flashes, bone loss | ER/PR expression |
| Radiation Therapy | DNA double-strand breaks | 70-90% local | Durable local control | Skin reaction, fatigue | None required |
- Immunotherapy Response Patterns
- Primary resistance: 60-70% of patients show no initial response
- Acquired resistance: 30-40% of initial responders develop progression
- Hyperprogression: <10% show accelerated tumor growth
- Pseudoprogression: Initial enlargement followed by response in 5-10%
- Delayed responses: >6 months to maximum benefit in 20% of responders
- Immune-related adverse events: >80% experience any grade toxicity
- Durable responses: >50% of responders maintain benefit >2 years
💡 Master This: Resistance mechanisms drive treatment sequencing decisions-EGFR T790M mutations develop in >60% of first-generation TKI failures, requiring third-generation inhibitors like osimertinib. Liquid biopsies detect resistance mutations 4-8 weeks before radiographic progression, enabling proactive treatment changes.

Treatment sequencing optimization requires understanding cross-resistance patterns and synergistic combinations. Neoadjuvant therapy achieves pathological complete response in 30-60% of cases, with pCR rates strongly predicting long-term survival across multiple tumor types.
Resistance management strategies include combination approaches, sequential therapy, and intermittent dosing to delay resistance and maintain sensitivity. Adaptive therapy approaches that modulate treatment intensity based on tumor response show promise in extending progression-free survival compared to continuous maximum-dose strategies.
Connect treatment paradigm understanding through integrated cancer management to master how multidisciplinary approaches coordinate surgery, radiation, systemic therapy, and supportive care for optimal patient outcomes.
⚖️ Treatment Paradigms: The Precision Medicine Revolution
🎯 Mastery Integration: The Oncology Command Center
📌 Remember: MASTER for Oncology Integration - Molecular profiling, Anatomical staging, Systemic therapy selection, Timing optimization, Emergency recognition, Resistance management. Each component requires evidence-based decision-making that adapts to tumor evolution and patient factors.
Clinical decision algorithms integrate tumor biology, staging information, and patient factors to guide treatment selection with >90% accuracy in standard scenarios. Molecular tumor boards review complex cases where standard algorithms require expert interpretation of novel biomarkers or rare genetic alterations.
- Essential Clinical Integration Points
- Staging accuracy: >95% concordance between clinical and pathological staging
- Biomarker turnaround: <14 days for actionable mutation results
- Treatment initiation: <30 days from diagnosis to first treatment
- Multidisciplinary conferences: Weekly review of complex cases
- Genetic counseling: <7 days for hereditary syndrome evaluation
- Response assessment: Every 6-12 weeks with standardized criteria
- Toxicity management: 24/7 access to oncology expertise
⭐ Clinical Pearl: Liquid biopsies enable real-time monitoring of treatment response and resistance development, with circulating tumor DNA changes preceding radiographic progression by 4-16 weeks. Serial ctDNA monitoring guides treatment modifications and early intervention for emerging resistance.
| Clinical Scenario | Key Decision Points | Biomarker Requirements | Treatment Sequence | Monitoring Strategy |
|---|---|---|---|---|
| Early-Stage Solid Tumor | Surgery timing, adjuvant therapy | Prognostic markers | Surgery → Adjuvant | Surveillance imaging |
| Locally Advanced Disease | Neoadjuvant vs concurrent | Predictive biomarkers | Neoadjuvant → Surgery | Response assessment |
| Metastatic Disease | First-line selection | Actionable mutations | Targeted → Immunotherapy | ctDNA monitoring |
| Hereditary Syndrome | Risk-reducing surgery | Germline testing | Prevention → Surveillance | Family screening |
| Treatment Resistance | Mechanism identification | Resistance mutations | Sequential targeting | Liquid biopsy |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
DX["🩺 Cancer Diagnosis
• Biopsy confirmation• Tissue analysis"]
MDT["📋 MDT Review
• Team discussion• Expert consensus"]
PROF["🔬 Molec Profile
• Genetic markers• Targeted targets"]
STG["🔬 Staging
• Imaging scans• TNM classification"]
PLAN["💊 Rx Planning
• Regimen choice• Patient goals"]
INTENT{"📋 Curative?
• Goal assessment• Outcome intent"}
MULTI["💊 Multimodal Rx
• Combined therapy• Serial treatments"]
MON["👁️ Monitoring
• Track response• Follow imaging"]
COMP{"📋 Complete?
• Check remission• Total response"}
SURV["✅ Surveillance
• Regular follow-up• Watch recurrence"]
RESIST["🧪 Resistance
• Clonal evolution• Bio-mechanism"]
MOD["💊 Rx Mod
• Adjust therapy• Second line care"]
PALL["💊 Palliative
• Quality of life• Symptom focus"]
SYMP["🩹 Symptom Mgmt
• Pain control• Support care"]
DX --> MDT --> PROF --> STG --> PLAN --> INTENT INTENT -->|Yes| MULTI INTENT -->|No| PALL MULTI --> MON --> COMP COMP -->|Yes| SURV COMP -->|No| RESIST RESIST --> MOD PALL --> SYMP
style DX fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style MDT fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style PROF fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style STG fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style PLAN fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style INTENT fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style MULTI fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style MON fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1 style COMP fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style SURV fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252 style RESIST fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style MOD fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style PALL fill:#F1FCF5, stroke:#BEF4D8, stroke-width:1.5px, rx:12, ry:12, color:#166534 style SYMP fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1
**Rapid reference frameworks** enable **point-of-care** decision-making during **urgent clinical scenarios**. **Oncologic emergencies** require **immediate recognition** and **intervention** within **<6 hours** for **optimal outcomes**, with **standardized protocols** reducing **morbidity** by **>50%**.
* **Critical Oncology Thresholds**
- **Neutropenia**: **ANC <500** requires **immediate antibiotics**
- **Hypercalcemia**: **>14 mg/dL** needs **urgent intervention**
- **Tumor lysis syndrome**: **Uric acid >8 mg/dL** with **renal dysfunction**
+ **Spinal cord compression**: **<24 hours** to **radiation therapy**
+ **Superior vena cava syndrome**: **<48 hours** to **treatment initiation**
- **Thrombocytopenia**: **<10,000** requires **platelet transfusion**
- **Febrile neutropenia**: **Temperature >100.4°F** with **ANC <1000**
> 💡 **Master This**: **Precision oncology** success depends on **systematic biomarker integration**-**>70%** of patients with **actionable mutations** achieve **clinical benefit** when matched to **appropriate targeted therapy**, compared to **<30%** response rates with **empirical treatment** selection. **Molecular tumor boards** improve **biomarker utilization** from **40%** to **>85%** in **complex cases**.
**Treatment resistance patterns** follow **predictable evolutionary pathways** that enable **proactive management strategies**. **Sequential therapy** based on **resistance mechanisms** extends **overall survival** by **6-12 months** compared to **empirical treatment** changes, highlighting the importance of **systematic resistance** analysis.
**Quality metrics** for **oncology excellence** include **>90%** **biomarker testing** rates, **<30 days** **diagnosis-to-treatment** intervals, **>80%** **clinical trial** enrollment eligibility discussion, and **<5%** **30-day readmission** rates, creating **measurable standards** for **comprehensive cancer care** delivery.
🎯 Mastery Integration: The Oncology Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app




















