Basics & Immunity - Vaccine 101 Kickstart
-
Vaccine: Biological agent providing active acquired immunity.
-
Vaccination: Administering a vaccine.
-
Immunization: Process of becoming protected against disease.
-
Active Immunity: Body produces its own antibodies.
- Natural: Post-infection (e.g., Measles). Long-lasting.
- Artificial: Post-vaccination (e.g., MMR vaccine). Long-lasting.
-
Passive Immunity: Pre-formed antibodies received.
- Natural: Maternal (placenta, breast milk). Short-term.
- Artificial: Immunoglobulin administration (e.g., Tetanus Immunoglobulin). Immediate, short-term.
-
Immune Response to Vaccines:
- Primary: Slow onset, lower Ab levels (IgM → IgG).
- Secondary (Booster): Rapid, ↑↑ high Ab levels (IgG), memory.
⭐ Artificially acquired passive immunity provides immediate but temporary protection (e.g., Tetanus Immunoglobulin).
Vaccine Varieties - The Immunization Arsenal
📌 LIVE vaccines mnemonic: 'BOY, This Real MMR Vaccine is Scary' (BCG, OPV, Yellow Fever, Typhoid oral, Rota, MMR, Varicella, Influenza nasal, Sabin).
| Type | Feature | Examples | Adv. | Disadv. |
|---|---|---|---|---|
| Live Atten. | Weakened pathogen | MMR, BCG, OPV | Strong, long immunity | Risk in IC; reversion |
| Inactivated | Killed pathogen | IPV, Rabies, Flu | Safer; no reversion | Weaker; multi-dose |
| Toxoid | Inactivated toxin | TT, Diphtheria | Targets toxin | Toxin diseases only |
| Subunit | Specific Ag | Hep B, PPSV23 | Fewer side effects | Weaker; needs adjuvant |
| Conjugate | Polysacch.+prot | Hib, PCV | ↑Infant response | Complex |
| mRNA | mRNA for Ag | COVID-19 | Rapid dev; non-infect. | Ultra-cold chain |
| Viral Vector | Benign virus+Ag | COVID-19, Ebola | Strong response | Pre-exist. vector immunity |
Live attenuated vaccines: robust, long-lasting immunity, often lifelong with one dose, mimic natural infection.
Delivery & Timelines - Shot Clock Science
- Routes & Examples:
- IM: Hep B, DPT, IPV, PCV (Deltoid, Thigh)
- SC: MMR, Varicella, JE (Thigh, Upper arm)
- ID: BCG (L. Upper arm), Rabies (updated regimen)
- Oral: OPV, Rotavirus
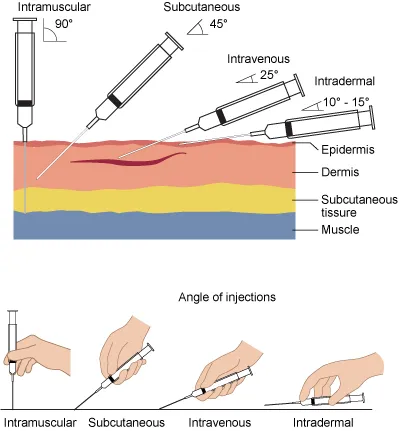
- Scheduling Principles:
- Simultaneous: Most vaccines, different sites.
- Intervals:
- Live-Live (if not same day): Min. 4 weeks.
- Live-Inactivated: Any interval.
- Inactivated-Inactivated: Any interval (often 4 weeks for same antigen).
- NIS India: Vital public health tool. Key vaccines: BCG, OPV, Hep B, Pentavalent, IPV, Rota, MMR, JE.
- Catch-up: Give missed doses ASAP per age/interval guidelines. No restart needed for most.
⭐ Most live vaccines can be given simultaneously. If not, a minimum interval of 4 weeks is required between two live vaccines_
Efficacy, Safety & Issues - Guarding the Guards
- Herd Immunity: Indirect protection for unvaccinated when many are immune; vital for community.
- Vaccine Performance:
- Efficacy: Protection in ideal trial conditions.
- Effectiveness: Protection in real-world settings.
- Adverse Events Following Immunization (AEFIs):
- Common: Local (pain, redness), systemic (fever, malaise).
- Serious: Rare (e.g., anaphylaxis, GBS); mandatory reporting.
- Contraindications:
- General: Prior anaphylaxis to vaccine/component.
- Live vaccines: Severe immunodeficiency, pregnancy (generally).
- Cold Chain: System for vaccine storage/transport at +2°C to +8°C.
⭐ The cold chain is critical for maintaining vaccine potency; most vaccines are stored between +2°C and +8°C, and freeze-sensitive vaccines (like DPT, TT, Hep B) must never be frozen.

High‑Yield Points - ⚡ Biggest Takeaways
- Live vaccines (MMR, OPV): long immunity; C/I in immunodeficiency & pregnancy.
- Killed vaccines (IPV, Rabies): multiple doses; safer for immunocompromised.
- Toxoids (Tetanus, Diphtheria): immunity against bacterial toxins.
- Cold chain: OPV (most heat-sensitive); Hep B, DPT, TT (freeze-sensitive).
- Minimum interval for two live vaccines (if not simultaneous): 4 weeks.
- Adjuvants (e.g., Alum) boost immunogenicity of killed/subunit vaccines.
- Anaphylaxis post-vaccination: treat with adrenaline.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

