Genetics and Disease

On this page
🧬 The Genetic Foundation: Decoding Life's Blueprint
Every cell in your body carries the same genetic code, yet mutations in just one gene can trigger devastating multi-system disease while others remain silent for decades. This lesson takes you from DNA's molecular architecture through the clinical patterns that distinguish hundreds of genetic conditions, then equips you with the diagnostic frameworks and therapeutic strategies to recognize, differentiate, and manage inherited disorders. You'll master how single-gene defects create recognizable phenotypes, why some diseases skip generations while others don't, and which interventions can alter outcomes. By integrating molecular mechanisms with bedside pattern recognition, you'll transform genetic principles into clinical decision-making power.
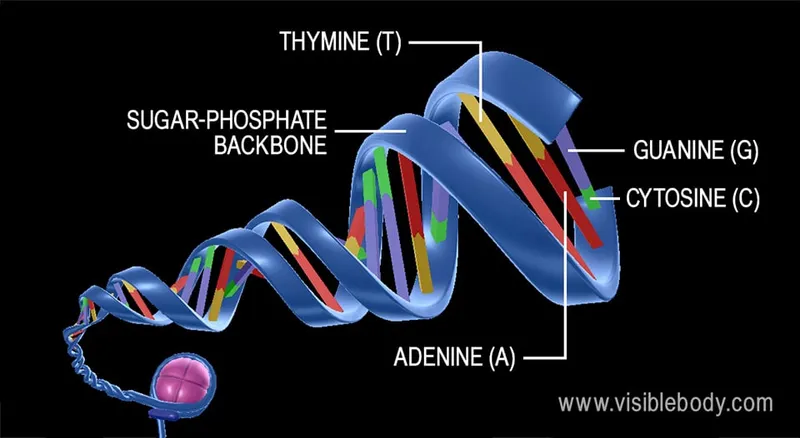
📌 Remember: ATGC - Adenine pairs with Thymine (2 hydrogen bonds), Guanine pairs with Cytosine (3 hydrogen bonds). This differential bonding strength explains why GC-rich regions have higher melting temperatures (Tm) and greater stability.
The human genome contains approximately 20,000-25,000 protein-coding genes distributed across 23 chromosome pairs. Each cell maintains 6 billion base pairs of DNA, with 99.9% sequence identity between individuals. That remaining 0.1% variation-roughly 3 million differences-accounts for all human genetic diversity and disease susceptibility.
- Chromosomal Organization
- Autosomes: 22 pairs (chromosomes 1-22)
- Sex chromosomes: 1 pair (XX or XY)
- X chromosome: 155 million base pairs, ~1,000 genes
- Y chromosome: 59 million base pairs, ~200 genes
- Mitochondrial DNA: 16,569 base pairs, 37 genes
⭐ Clinical Pearl: Mitochondrial diseases affect 1 in 4,000 births, with maternal inheritance patterns since sperm contribute virtually no mitochondria during fertilization. Each cell contains 100-1,000 mitochondria with 2-10 copies of mtDNA each.
| Genetic Element | Size | Gene Count | Clinical Significance | Inheritance Pattern | Disease Examples |
|---|---|---|---|---|---|
| Nuclear DNA | 3.2 billion bp | ~20,000 | Most genetic diseases | Mendelian patterns | Cystic fibrosis, Huntington's |
| Mitochondrial DNA | 16,569 bp | 37 | Energy metabolism disorders | Maternal only | MELAS, MERRF |
| X chromosome | 155 million bp | ~1,000 | X-linked disorders | Sex-linked | Hemophilia, DMD |
| Y chromosome | 59 million bp | ~200 | Male development | Paternal only | Male infertility |
| Chromosome 21 | 48 million bp | ~300 | Trisomy syndromes | Variable | Down syndrome |
Gene expression follows the central dogma: DNA → RNA → Protein, with multiple regulatory checkpoints. Approximately 98% of the human genome consists of non-coding sequences, including regulatory elements, introns, and repetitive sequences that influence gene expression patterns.
📌 Remember: TFIID complex components - TATA-binding protein, Factors for Initiation, Including Downstream promoter elements. This complex recognizes TATA box sequences located 25-30 base pairs upstream of transcription start sites.
Connect this genetic foundation through molecular mechanisms to understand how single nucleotide variations create the disease patterns explored in inheritance analysis.
🧬 The Genetic Foundation: Decoding Life's Blueprint
⚙️ The Molecular Machinery: Genetic Mechanisms in Action
⭐ Clinical Pearl: Alternative splicing defects cause 15% of genetic diseases. The CFTR gene has 27 exons with >1,900 disease-causing mutations, where 70% result from the ΔF508 deletion affecting protein folding rather than splicing.
Translation efficiency depends on ribosome density and codon usage bias. Human cells contain ~10 million ribosomes, each capable of synthesizing 3-5 amino acids per second. The genetic code's degeneracy provides protection against mutations, with 18 amino acids having multiple codons.
- Translation Control Mechanisms
- 5' UTR secondary structures regulate ribosome binding
- Kozak sequence (GCCRCCATGG) optimizes translation initiation
- R = purine (A or G) at positions -3 and +4
- Optimal context increases translation efficiency 10-fold
- 3' UTR elements control mRNA stability and localization
- AU-rich elements (AREs) promote mRNA degradation
- microRNA binding sites fine-tune protein levels
📌 Remember: START-STOP codon relationships - Start with AUG (methionine), Terminate with Amber (UAG), Red (UAA), Teal (UGA). 64 total codons encode 20 amino acids plus 3 stop signals.
| Mutation Type | Frequency | Protein Effect | Clinical Severity | Examples | Treatment Approach |
|---|---|---|---|---|---|
| Missense | 45% | Amino acid change | Variable | Sickle cell (Glu→Val) | Protein therapy |
| Nonsense | 12% | Premature stop | Severe | DMD, CF | Gene therapy |
| Frameshift | 20% | Reading frame shift | Severe | Tay-Sachs | Enzyme replacement |
| Splice site | 15% | Aberrant splicing | Moderate-severe | Thalassemia | Splicing modulators |
| Regulatory | 8% | Expression changes | Variable | FGFR3 promoter | Transcriptional control |
💡 Master This: Homologous recombination repairs double-strand breaks with high fidelity, while non-homologous end joining provides rapid but error-prone repair. BRCA1/BRCA2 deficiencies impair homologous recombination, increasing breast cancer risk by 45-65% and ovarian cancer risk by 11-39%.

Connect these molecular mechanisms through pattern recognition frameworks to understand how genetic variations manifest as recognizable clinical phenotypes.
⚙️ The Molecular Machinery: Genetic Mechanisms in Action
🎯 The Pattern Recognition Matrix: Clinical Genetic Signatures
📌 Remember: FACES approach to dysmorphology - Facial features, Anomalies of limbs, Cardiac defects, Eye abnormalities, Skeletal malformations. Document measurements, proportions, and asymmetries with standardized terminology.
Recognition patterns enable rapid syndrome identification through gestalt diagnosis. Down syndrome presents with >80 described features, but core recognition pattern includes upslanting palpebral fissures, epicanthal folds, flat nasal bridge, protruding tongue, and single palmar creases in 45% of cases.
- High-Yield Syndrome Recognition Patterns
- Marfan syndrome: Arm span-to-height ratio >1.05, arachnodactyly, lens dislocation
- Ghent criteria require major criteria in ≥2 organ systems
- FBN1 mutations in >90% of cases
- Noonan syndrome: Webbed neck, pectus deformity, cryptorchidism
- PTPN11 mutations in 50% of cases
- Pulmonary stenosis in 50-62% of patients
- Williams syndrome: Elfin facies, supravalvular aortic stenosis, cocktail party personality
- 7q11.23 deletion including ELN gene
- Hypercalcemia in 15% during infancy
- Marfan syndrome: Arm span-to-height ratio >1.05, arachnodactyly, lens dislocation
⭐ Clinical Pearl: Three or more minor anomalies in a newborn indicates 90% probability of associated major malformation. Single minor anomaly occurs in 14% of normal newborns, while ≥3 minor anomalies occur in <1% of normal population.
| Syndrome | Incidence | Key Features | Genetic Cause | Cardiac Involvement | Developmental Delay |
|---|---|---|---|---|---|
| Down syndrome | 1:700 births | Hypotonia, characteristic facies | Trisomy 21 | 40-50% (AV canal) | Mild-moderate |
| Turner syndrome | 1:2,500 females | Short stature, webbed neck | 45,X or variants | 25% (coarctation) | Normal IQ |
| Klinefelter | 1:500 males | Tall stature, gynecomastia | 47,XXY | Minimal | Learning difficulties |
| Fragile X | 1:4,000 males | Macrocephaly, long face | FMR1 CGG expansion | 20% (MVP) | Moderate-severe |
| Prader-Willi | 1:15,000 | Hypotonia, hyperphagia | 15q11-q13 deletion | Minimal | Mild-moderate |
💡 Master This: Anticipation occurs when disease severity increases or age of onset decreases in successive generations, typically due to trinucleotide repeat expansions. Huntington disease shows paternal anticipation with CAG repeats expanding from 6-26 normal to >40 pathogenic.

Connect these recognition patterns through systematic discrimination frameworks to understand how similar presentations require careful differential diagnosis approaches.
🎯 The Pattern Recognition Matrix: Clinical Genetic Signatures
🔬 The Differential Diagnosis Engine: Systematic Genetic Discrimination
📌 Remember: MILD intellectual disability classification - Mild (IQ 50-70, 85% of cases), Intermediate/Moderate (IQ 35-50, 10%), Low/Severe (IQ 20-35, 4%), Deep/Profound (IQ <20, 1%). Early intervention improves outcomes across all severity levels.
Connective tissue disorders present overlapping features requiring systematic evaluation of cardiovascular, ocular, skeletal, and dermatologic manifestations. Marfan syndrome, Ehlers-Danlos syndromes, and Loeys-Dietz syndrome share aortic dilatation risk but differ in inheritance patterns and management protocols.
- Connective Tissue Disorder Discrimination
- Marfan syndrome (FBN1 mutations)
- Aortic root Z-score ≥2 in adults
- Lens dislocation (typically superotemporal)
- Systemic score ≥7 using revised Ghent criteria
- Ehlers-Danlos Classical (COL5A1/COL5A2 mutations)
- Hyperextensible skin (>1.5cm on forearm)
- Atrophic scarring over pressure points
- Joint hypermobility (Beighton score ≥5)
- Loeys-Dietz syndrome (TGFBR1/TGFBR2 mutations)
- Bifid uvula or cleft palate
- Arterial tortuosity throughout vascular tree
- Aggressive aortic dilatation (earlier onset than Marfan)
- Marfan syndrome (FBN1 mutations)
⭐ Clinical Pearl: Beighton hypermobility score uses 9-point scale assessing passive thumb-to-forearm apposition, passive fifth finger hyperextension >90°, elbow hyperextension >10°, knee hyperextension >10°, and forward flexion with palms flat on floor. Score ≥5 indicates generalized joint hypermobility.
| Disorder | Gene | Cardiac Risk | Ocular Features | Skin Involvement | Inheritance | Management Priority |
|---|---|---|---|---|---|---|
| Marfan | FBN1 | Aortic root dilatation | Lens dislocation | Striae, hernias | AD | Aortic surveillance |
| EDS Classical | COL5A1/A2 | Minimal | Myopia | Hyperextensible | AD | Wound care |
| EDS Vascular | COL3A1 | Arterial rupture | Minimal | Thin, translucent | AD | Vascular monitoring |
| Loeys-Dietz | TGFBR1/2 | Aggressive aortic | Strabismus | Velvety | AD | Early intervention |
| Stickler | COL2A1 | Minimal | Retinal detachment | Minimal | AD | Ophthalmologic care |
💡 Master This: Tandem mass spectrometry enables simultaneous analysis of amino acids, acylcarnitines, and organic acids from single blood spot. False positive rate of 0.1-0.5% requires confirmatory testing, while false negative rate of <0.01% provides excellent sensitivity for screened conditions.

Connect these discrimination frameworks through evidence-based treatment algorithms to understand how accurate diagnosis guides optimal therapeutic interventions.
🔬 The Differential Diagnosis Engine: Systematic Genetic Discrimination
💊 The Therapeutic Algorithm Matrix: Evidence-Based Genetic Interventions
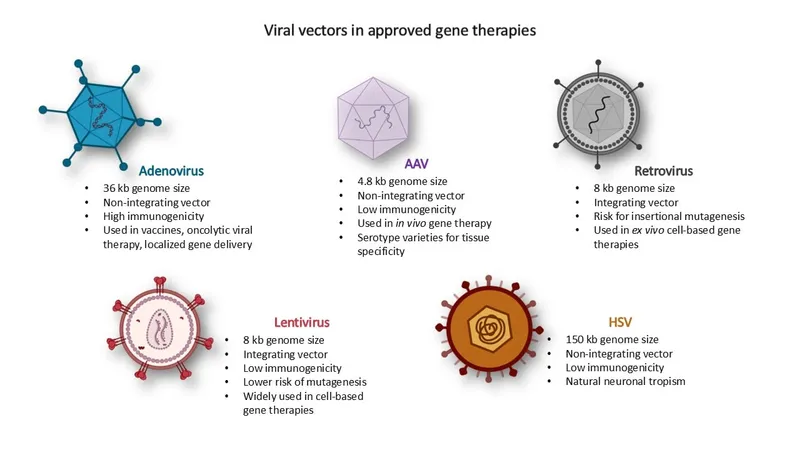
📌 Remember: VECTOR gene therapy considerations - Viral tropism for target tissues, Expression duration and levels, Capacity for therapeutic gene size, Toxicity and immunogenicity, Off-target effects, Regulatory approval status. AAV vectors show excellent safety but limited capacity (<5kb), while lentiviral vectors accommodate larger genes but require careful integration site analysis.
Enzyme replacement therapy (ERT) provides substrate reduction for lysosomal storage disorders affecting >50 conditions. Gaucher disease treatment with imiglucerase reduces hepatosplenomegaly by >50% and bone pain by >80% within 6-12 months, with dosing of 15-60 units/kg every 2 weeks.
- Targeted Therapy Success Rates by Mechanism
- Gene therapy (vector-based): 85-95% response in monogenic disorders
- Luxturna: 93% improvement in mobility testing
- Zolgensma: 100% event-free survival at 2 years
- Enzyme replacement: 70-90% symptom improvement
- Fabry disease: 50-70% reduction in neuropathic pain
- Pompe disease: Improved survival from <2 years to >10 years
- Small molecule therapy: 60-80% biochemical correction
- Cystic fibrosis (ivacaftor): >50% improvement in FEV1
- Spinal muscular atrophy (nusinersen): 40% achieve motor milestones
- Gene therapy (vector-based): 85-95% response in monogenic disorders
⭐ Clinical Pearl: Pharmacogenomic testing guides drug selection and dosing for >200 medications. CYP2D6 poor metabolizers (7% of Caucasians) require 50-90% dose reduction for codeine, tramadol, and tricyclic antidepressants, while ultrarapid metabolizers (1-2%) may need dose increases or alternative medications.
| Condition | Treatment Type | Mechanism | Response Rate | Cost (Annual) | Monitoring Required |
|---|---|---|---|---|---|
| SMA Type 1 | Gene therapy | SMN1 replacement | 100% survival | $2.1M (one-time) | Motor function |
| Gaucher Type 1 | Enzyme replacement | β-glucocerebrosidase | 90% improvement | $200K-400K | Biomarkers |
| Cystic fibrosis | Small molecule | CFTR modulation | 50-70% FEV1 | $300K | Pulmonary function |
| Fabry disease | Enzyme replacement | α-galactosidase A | 70% pain reduction | $200K-300K | Cardiac/renal |
| Hemophilia A | Gene therapy | Factor VIII expression | 95% bleed reduction | $2.5M (one-time) | Factor levels |
💡 Master This: CRISPR-Cas9 gene editing achieves >90% efficiency in ex vivo applications but faces delivery challenges for in vivo therapy. CTX001 for sickle cell disease shows >95% reduction in vaso-occlusive crises through ex vivo editing of patient hematopoietic stem cells followed by autologous transplantation.

Connect these treatment algorithms through multi-system integration approaches to understand how genetic interventions affect multiple organ systems simultaneously.
💊 The Therapeutic Algorithm Matrix: Evidence-Based Genetic Interventions
🌐 The Systems Integration Network: Multi-Organ Genetic Orchestration
📌 Remember: MELAS clinical criteria - Mitochondrial myopathy, Encephalopathy, Lactic acidosis, Stroke-like episodes. Diagnosis requires ≥2 major criteria: stroke-like episodes <40 years, encephalopathy with seizures/dementia, mitochondrial myopathy with ragged red fibers, plus ≥2 minor criteria: normal early development, recurrent headache, recurrent vomiting.
Connective tissue disorders demonstrate structural protein integration across multiple organ systems. Marfan syndrome affects fibrillin-1 distribution in aortic media, lens zonules, bone matrix, and skin elasticity, creating coordinated manifestations requiring multidisciplinary surveillance protocols.
- Multi-System Surveillance Protocols
- Marfan syndrome monitoring schedule
- Echocardiography: Annual if aortic root <40mm, every 6 months if 40-45mm
- Ophthalmologic exam: Annual with slit-lamp examination
- Orthopedic assessment: Every 2-3 years during growth, as needed in adults
- Neurofibromatosis type 1 surveillance
- Annual examination for neurofibromas, café-au-lait spots, blood pressure
- Ophthalmologic screening annually until age 8, then every 2 years
- MRI brain if neurological symptoms or learning difficulties
- Tuberous sclerosis complex monitoring
- Renal ultrasound every 1-3 years for angiomyolipomas
- Echocardiography every 1-3 years for cardiac rhabdomyomas
- Pulmonary function every 2-3 years in adult females for LAM
- Marfan syndrome monitoring schedule
⭐ Clinical Pearl: Genetic anticipation in trinucleotide repeat disorders shows parent-of-origin effects. Huntington disease demonstrates paternal anticipation with CAG expansion during spermatogenesis, while myotonic dystrophy shows maternal anticipation with congenital forms transmitted exclusively through affected mothers.
| Disorder | Primary System | Secondary Effects | Surveillance Interval | Life Expectancy | Management Team |
|---|---|---|---|---|---|
| Marfan syndrome | Connective tissue | Aortic, ocular, skeletal | 6-12 months | Near normal | Cardiology, ophthalmology |
| MELAS | Mitochondrial | Neurologic, cardiac, endocrine | 3-6 months | Variable (20-40 years) | Neurology, cardiology, endocrine |
| NF1 | Nervous system | Skeletal, vascular, malignancy | 12 months | Near normal | Genetics, neurology, oncology |
| TSC | Multiple systems | Neurologic, renal, pulmonary | 6-12 months | Variable | Neurology, nephrology, pulmonary |
| Duchenne MD | Muscular | Cardiac, respiratory | 3-6 months | 20-30 years | Neurology, cardiology, pulmonary |
💡 Master This: Polygenic risk scores integrate thousands of genetic variants to predict disease susceptibility and treatment response. Coronary artery disease PRS incorporating >6 million variants identifies 8% of population with >3-fold increased risk, enabling targeted prevention with statins and lifestyle interventions that reduce cardiovascular events by 40-50%.
Recent multi-omics integration combines genomics, transcriptomics, proteomics, and metabolomics to understand disease mechanisms and therapeutic targets. Single-cell RNA sequencing reveals cell-type-specific gene expression patterns in disease tissues, identifying novel therapeutic targets and biomarkers for treatment monitoring.
Connect this systems integration through rapid mastery frameworks to develop practical clinical tools for immediate genetic medicine application.
🌐 The Systems Integration Network: Multi-Organ Genetic Orchestration
🎯 The Clinical Genetics Arsenal: Rapid Mastery Toolkit
📌 Remember: GENETIC testing progression - General screening (karyotype, microarray), Exome sequencing for unknown syndromes, Newborn screening for treatable conditions, Ethnic-specific panels (Ashkenazi, Mediterranean), Targeted testing for known familial mutations, Immunodeficiency panels for recurrent infections, Cancer predisposition for family history.
| Test Type | Diagnostic Yield | Cost Range | Turnaround Time | Best Clinical Application | Coverage Limitations |
|---|---|---|---|---|---|
| Karyotype | 3-5% | $200-400 | 7-14 days | Suspected chromosomal disorder | Large abnormalities only |
| Microarray | 15-20% | $500-800 | 7-10 days | ID/MCA first-tier | CNVs >50kb typically |
| Exome sequencing | 25-30% | $1,000-3,000 | 2-4 weeks | Unknown genetic syndrome | Coding regions only |
| Genome sequencing | 35-40% | $3,000-5,000 | 2-6 weeks | Negative exome, structural | Interpretation challenges |
| Gene panels | 10-40% | $300-2,000 | 1-3 weeks | Specific phenotype | Limited gene coverage |
Hardy-Weinberg Equilibrium: $$p^2 + 2pq + q^2 = 1$$
- Carrier frequency for autosomal recessive: $$2pq \approx 2\sqrt{disease\ frequency}$$
- Cystic fibrosis (1:2,500 births): Carrier frequency = $$2\sqrt{1/2500} = 1:25$$
Recurrence Risk Calculations:
- Autosomal dominant: 50% each pregnancy
- Autosomal recessive: 25% if both parents carriers
- X-linked recessive: 50% for male offspring of carrier mothers
⭐ Clinical Pearl: Empiric recurrence risks apply when genetic testing is uninformative. Neural tube defects show 3-4% recurrence after one affected child, 10-12% after two affected children, reduced to <1% with folic acid supplementation (4-5mg daily) starting one month before conception.
- High-Yield Genetic Counseling Points
- Consanguinity increases autosomal recessive risk
- First cousins: 6.25% shared genes, 2-3x increased birth defect risk
- Double first cousins: 12.5% shared genes, 4-6x increased risk
- Advanced maternal age chromosomal risks
- Age 35: 1:385 Down syndrome risk
- Age 40: 1:106 Down syndrome risk
- Age 45: 1:30 Down syndrome risk
- Paternal age effects for new dominant mutations
- >40 years: 2-3x increased risk for achondroplasia, Apert syndrome
- >50 years: 5-6x increased risk for new mutations
- Consanguinity increases autosomal recessive risk
💡 Master This: Variant classification follows ACMG guidelines with 5 categories: Pathogenic (>99% certainty), Likely pathogenic (90-99%), Uncertain significance (10-90%), Likely benign (90-99% benign), Benign (>99% benign). Variants of uncertain significance occur in 40-50% of exome sequencing results, requiring family studies and functional analysis for reclassification.
Essential genetic medicine reference values for immediate clinical application:
- Normal karyotype resolution: 5-10 Mb deletions/duplications
- Microarray resolution: 50-100 kb CNVs genome-wide
- Exome coverage: ~85% of coding sequences at >20x depth
- Genome coverage: >95% of genome at >30x depth
- Variant frequency thresholds: <1% in population databases for rare disease
- Penetrance estimates: Age-dependent for most dominant conditions
This clinical genetics arsenal provides the systematic framework and quantitative tools necessary for evidence-based genetic medicine practice, enabling rapid assessment, accurate diagnosis, and optimal patient outcomes across diverse clinical presentations.
🎯 The Clinical Genetics Arsenal: Rapid Mastery Toolkit
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























