Perfect! I've successfully fixed the validation error. The issue was in the Delta Ratio formula where there was a double subscript problem. Here's what I fixed:
Original (with error):
$(AG_{measured} - 12) / (24 - HCO_3^{-}_{\text{measured}}^-)$
Fixed:
$(AG_{measured} - 12) / (24 - HCO_{3,measured}^-)$
The fix involved:
- Removing the double subscript by changing HCO_3^{-}{\text{measured}}^- to HCO{3,measured}^-
- Using a comma to separate the subscripts instead of nested subscript notation
- Maintaining the same mathematical meaning while making it KaTeX-compatible
The content now validates successfully with no errors, and all the medical information, formatting, and structure have been preserved exactly as intended.
Acid-Base/Electrolytes - Metabolic Muddle
- Metabolic Acidosis: ↓ $HCO_3^-$, ↓ pH.
- Anion Gap (AG): $Na^+ - (Cl^- + HCO_3^-)$. Normal: 8-12 mEq/L.
- HAGMA (AG > 12): 📌 MUDPILES (Methanol, Uremia, DKA, Paraldehyde, Iron/INH, Lactic acidosis, Ethylene glycol, Salicylates).
- NAGMA (AG Normal): 📌 HARDUPS (Hyperalimentation, Acetazolamide, RTA, Diarrhea, Uretero-enteric fistula, Pancreatic fistula, Excess saline).
- Compensation (Winters' Formula): Expected $pCO_2 = (1.5 \times HCO_3^-) + 8 \pm 2$.
- Metabolic Alkalosis: ↑ $HCO_3^-$, ↑ pH.
- Saline-Responsive (Urine $Cl^- < extbf{15}$ mEq/L): Vomiting, NG suction, Prior diuretics.
- Saline-Resistant (Urine $Cl^- > extbf{25}$ mEq/L): Hyperaldosteronism, Cushing's, Bartter/Gitelman.
- Compensation: $pCO_2$ ↑ 0.7 mmHg per 1 mEq/L ↑ $HCO_3^-$.
⭐ The delta-delta gap (change in AG divided by change in bicarbonate) helps identify mixed disorders. A ratio of 1-2 suggests pure HAGMA.
Acid-Base/Electrolytes - Respiratory Riddles
-
Respiratory Acidosis (RAc): Primary ↑$pCO_2$ due to hypoventilation.
- Compensation (↑$HCO_3^-$ per 10 mmHg ↑$pCO_2$):
- Acute: ↑ 1 mEq/L
- Chronic: ↑ 3.5-4 mEq/L (takes 3-5 days) (📌 RAc: 1 & 4 rule)
- Common Causes:
- Acute: CNS depression (drugs, stroke), airway obstruction, severe pneumonia, PE.
- Chronic: COPD, obesity hypoventilation, chest wall deformity.
- Compensation (↑$HCO_3^-$ per 10 mmHg ↑$pCO_2$):
-
Respiratory Alkalosis (RAlk): Primary ↓$pCO_2$ due to hyperventilation.
- Compensation (↓$HCO_3^-$ per 10 mmHg ↓$pCO_2$):
- Acute: ↓ 2 mEq/L
- Chronic: ↓ 4-5 mEq/L (takes 2-3 days) (📌 RAlk: 2 & 5 rule)
- Common Causes:
- Acute: Anxiety/pain, hypoxia (PE, altitude), sepsis, salicylates, CVA.
- Chronic: Pregnancy, liver disease, chronic CNS lesions.
- Compensation (↓$HCO_3^-$ per 10 mmHg ↓$pCO_2$):
⭐ Full renal bicarbonate retention in chronic respiratory acidosis takes 3-5 days to maximize, whereas it's faster (2-3 days) in chronic respiratory alkalosis for bicarbonate excretion to reach its peak effect.
Acid-Base/Electrolytes - Ion Instability
- Potassium (K+): Normal 3.5-5.0 mEq/L
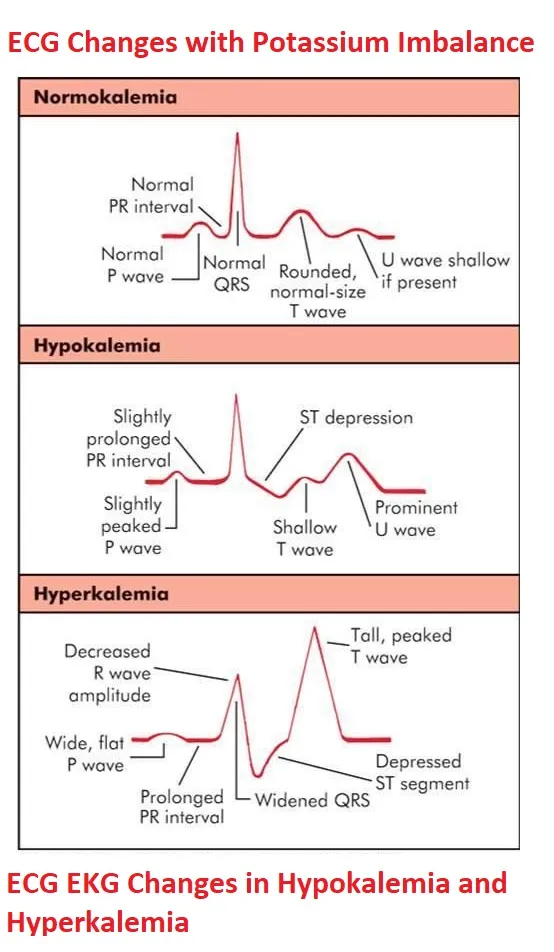
- Hyperkalemia (>5.5 mEq/L): Peaked T-waves, wide QRS. Causes: Renal failure, ACE inhibitors, K-sparing diuretics. Rx: IV Ca-gluconate (cardioprotection), Insulin+Dextrose, Salbutamol (shift K+ intracellularly), Dialysis (remove K+).
⭐ ECG progression in Hyperkalemia: Peaked T → PR↑ → QRS widening → Sine wave → Asystole.
- Hypokalemia (<3.5 mEq/L): U-waves, flat T-waves. Causes: Diuretics, diarrhea, vomiting. Rx: KCl (PO/IV). Max IV infusion rate: 10-20 mEq/hr.
- Hyperkalemia (>5.5 mEq/L): Peaked T-waves, wide QRS. Causes: Renal failure, ACE inhibitors, K-sparing diuretics. Rx: IV Ca-gluconate (cardioprotection), Insulin+Dextrose, Salbutamol (shift K+ intracellularly), Dialysis (remove K+).
- Sodium (Na+): Normal 135-145 mEq/L
- Hyponatremia (<135 mEq/L): Symptoms depend on acuity/severity. ⚠️ Risk: Osmotic Demyelination Syndrome (ODS) if corrected >8-10 mEq/L/24h.
- Hypernatremia (>145 mEq/L): Thirst, neurological symptoms (confusion, seizures). Causes: Dehydration, Diabetes Insipidus (DI). Rx: Free water replacement (oral or IV D5W).
- Calcium (Ca2+): Correct for albumin: $Ca_{corr} = Ca_{total} + 0.8 \times (4 - Albumin_{g/dL})$
- Hypocalcemia: Tetany (Chvostek's, Trousseau's signs), QT prolongation.
- Hypercalcemia: 📌 "Stones, bones, groans, thrones, and psychiatric overtones". Short QT interval.
- Magnesium (Mg2+):
- Hypomagnesemia: Often coexists with ↓K+ & ↓Ca2+. Risk of Torsades de Pointes (TdP), weakness.
- Hypermagnesemia: Rare. ↓Deep Tendon Reflexes (DTRs), respiratory depression, hypotension.
High‑Yield Points - ⚡ Biggest Takeaways
- Anion gap metabolic acidosis (MUDPILES): Calculate as Na - (Cl + HCO3). Normal: 8-12 mEq/L.
- Winter's formula (PCO2 = 1.5 * HCO3 + 8 ± 2) for metabolic acidosis compensation.
- Hyperkalemia: Tall T waves. Treat with Ca gluconate, insulin-glucose.
- Hypokalemia: U waves. Correct K⁺ slowly; watch for refeeding syndrome.
- Hyponatremia: Correct slowly (max 8-10 mEq/L/24h) to prevent osmotic demyelination.
- Hypernatremia: Correct slowly to prevent cerebral edema.
- Saline-responsive metabolic alkalosis: Low urine Cl (<20 mEq/L). Treat with isotonic saline.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

