Cardiology

On this page
🗺️ Cardiovascular Architecture: The Hemodynamic Blueprint
The cardiovascular system operates as an integrated pressure-volume network where structural integrity determines functional capacity. Understanding cardiac anatomy through hemodynamic principles reveals why specific lesions produce characteristic clinical syndromes and guides therapeutic interventions.
Cardiac Chamber Geometry & Pressure Dynamics
The heart functions as two pumps in series, each generating distinct pressure profiles that determine blood flow patterns and failure mechanisms:
-
Right Heart System (low-pressure circuit)
- Right atrial pressure: 0-8 mmHg (elevated >10 mmHg signals volume overload)
- RV systolic pressure: 15-30 mmHg (matches pulmonary artery systolic)
- RV diastolic pressure: 0-8 mmHg (thin-walled chamber, volume-sensitive)
- RV stroke work: ~10% of LV due to lower afterload
- RV failure threshold: PA systolic >55 mmHg chronically
- Pulmonary capillary wedge pressure (PCWP): 6-12 mmHg (reflects LA pressure)
-
Left Heart System (high-pressure circuit)
- Left atrial pressure: 4-12 mmHg (elevated >18 mmHg causes pulmonary edema)
- LV systolic pressure: 90-140 mmHg (generates systemic perfusion)
- LV end-diastolic pressure (LVEDP): 8-12 mmHg (>16 mmHg indicates diastolic dysfunction)
- LV ejection fraction: 55-70% (preserved systolic function)
- LV mass index: Men <115 g/m², Women <95 g/m²
- Aortic pressure: Systolic 90-140 mmHg, Diastolic 60-90 mmHg
- Pulse pressure: 30-50 mmHg (widened in aortic regurgitation, stiffened vessels)

📌 Remember: "RAP-RV-PA-PCWP-LA-LV-AO" traces the pressure cascade from 0→8→30→12→12→140 mmHg-each jump reveals chamber function and identifies failure points.
Valvular Orifice Areas & Flow Dynamics
Valve stenosis severity correlates directly with orifice area reduction and transvalvular pressure gradients:
| Valve | Normal Area | Mild Stenosis | Moderate Stenosis | Severe Stenosis | Critical Threshold |
|---|---|---|---|---|---|
| Aortic | 3.0-4.0 cm² | 1.5-2.0 cm² | 1.0-1.5 cm² | <1.0 cm² | <0.6 cm² (symptomatic) |
| Mitral | 4.0-6.0 cm² | 1.5-2.5 cm² | 1.0-1.5 cm² | <1.0 cm² | <0.5 cm² (critical) |
| Tricuspid | 7.0-9.0 cm² | 3.0-5.0 cm² | 2.0-3.0 cm² | <2.0 cm² | Rare isolated disease |
| Pulmonary | 5.0-7.0 cm² | Rarely stenotic | Usually congenital | Variable | Post-surgical only |
| Mean Gradient | <5 mmHg | 5-15 mmHg | 15-40 mmHg | >40 mmHg | Intervention threshold |
⭐ Clinical Pearl: Aortic valve area <1.0 cm² with mean gradient >40 mmHg predicts 75% mortality at 3 years without intervention-valve replacement indicated regardless of symptoms.
- Regurgitant Lesions (volume overload patterns)
- Regurgitant fraction: Mild <30%, Moderate 30-50%, Severe >50%
- Regurgitant volume: Severe aortic regurgitation >60 mL/beat
- Effective regurgitant orifice area (EROA): Severe mitral regurgitation >0.4 cm²
- Acute vs chronic: Acute MR causes pulmonary edema with normal LA size
- Chronic compensation: LA dilates to >60 mL/m², preventing symptoms until decompensation

💡 Master This: Stenotic lesions create pressure overload (concentric hypertrophy, preserved EF until late), while regurgitant lesions cause volume overload (eccentric hypertrophy, dilated chambers, early EF decline)-this distinction predicts surgical timing.
Coronary Perfusion Territories & Dominance Patterns
Coronary anatomy determines infarct location, arrhythmia risk, and mechanical complications:
-
Left Anterior Descending (LAD) - 45-55% of myocardial mass
- Anterior wall, anteroseptum, apex
- Diagonal branches: Anterolateral wall
- Septal perforators: Anterior 2/3 of interventricular septum
- Proximal LAD occlusion: Anterior STEMI, high risk cardiogenic shock (15-20%)
- Septal involvement: Complete heart block risk (5-10% of anterior MI)
-
Left Circumflex (LCx) - 15-25% of myocardial mass
- Lateral wall, posterior wall (in 15% left-dominant systems)
- Obtuse marginal branches: Lateral LV
- "Silent culprit": Posterior MI often lacks ST elevation in standard leads
-
Right Coronary Artery (RCA) - 30-40% of myocardial mass
- Inferior wall, RV free wall, posterior wall (85% right-dominant)
- Posterior descending artery (PDA): Inferior septum
- AV nodal artery (90% from RCA): Conduction system
- Inferior STEMI: 30-40% develop bradycardia/AV block (usually transient)
- RV infarction: 30-50% of inferior MI, avoid preload reduction
📌 Remember: "LAD = Pump, LCx = Lateral, RCA = Rhythm"-LAD controls contractility, LCx is electrically silent, RCA governs conduction and preload-sensitive RV.
- Coronary Dominance Patterns
- Right-dominant (85%): RCA gives PDA + posterolateral branches
- Left-dominant (8%): LCx gives PDA + posterolateral branches
- Co-dominant (7%): Both contribute to inferior/posterior supply
- Clinical impact: Left-dominant systems have worse prognosis with LCx occlusion
- Collateral circulation: Develops over weeks-months with chronic stenosis >70%

Understanding these pressure-volume relationships, valvular thresholds, and perfusion territories transforms abstract anatomy into predictive clinical frameworks-every hemodynamic derangement follows these architectural rules.
🗺️ Cardiovascular Architecture: The Hemodynamic Blueprint
📚 The Cardiac Cycle Engine: Systolic & Diastolic Machinery
The cardiac cycle integrates electrical activation, mechanical contraction, and valve function into synchronized pressure-volume work. Mastering these temporal relationships reveals why specific pathologies produce characteristic clinical presentations and guides hemodynamic interventions.
Systolic Function: The Ejection Phase
Systole transforms electrical depolarization into mechanical work through calcium-mediated actin-myosin cross-bridging:
-
Isovolumetric Contraction (50-80 ms)
- All valves closed, LV pressure rises from 8→80 mmHg
- No volume change, maximum dP/dt (contractility marker)
- Peak dP/dt: 1200-1800 mmHg/sec (reduced <1000 in systolic dysfunction)
- Prolonged in aortic stenosis: Pressure must exceed >100 mmHg to open valve
- Abbreviated in mitral regurgitation: Early ejection into LA at lower pressure
-
Rapid Ejection (100-120 ms)
- Aortic valve opens when LV pressure exceeds aortic diastolic (~80 mmHg)
- 70% of stroke volume ejected in first half of systole
- Peak LV pressure: 120-140 mmHg at mid-ejection
- Stroke volume: 60-100 mL/beat (cardiac output = SV × HR)
- Ejection fraction = (EDV - ESV) / EDV × 100 = 55-70%
-
Reduced Ejection (120-150 ms)
- Aortic flow decelerates, pressure equalizes LV-aorta
- Aortic valve closes when LV pressure < aortic pressure
- Dicrotic notch on aortic pressure tracing marks closure
⭐ Clinical Pearl: Pulsus parvus et tardus (weak, delayed carotid upstroke) in severe aortic stenosis reflects prolonged ejection time >300 ms and reduced stroke volume-sensitivity 85% for valve area <0.8 cm².
Diastolic Function: The Filling Phase
Diastole determines preload through active relaxation and passive compliance-often the first function to fail:
-
Isovolumetric Relaxation (60-100 ms)
- All valves closed, LV pressure drops from 80→8 mmHg
- Active energy-dependent process (ATP-dependent calcium reuptake)
- Tau (relaxation time constant): 30-40 ms (prolonged >50 ms in diastolic dysfunction)
- Impaired in LV hypertrophy, ischemia, aging
- Mitral valve opens when LV pressure < LA pressure (~8 mmHg)
-
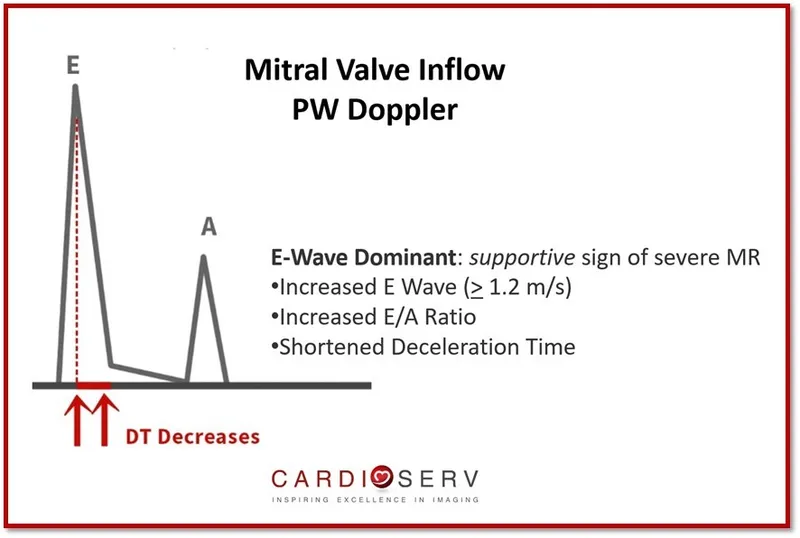
Rapid Filling (120-150 ms)
- 70-80% of ventricular filling occurs passively
- E-wave velocity on echo Doppler: 0.6-1.0 m/sec
- LV suction creates negative pressure gradient LA→LV
- E/A ratio: Normally 1.0-2.0 (reversed <0.8 in impaired relaxation)
- Deceleration time: 160-240 ms (shortened <140 ms in restriction)
-
Diastasis (variable duration)
- Minimal flow, pressure equilibration LA-LV
- Shortens with tachycardia (heart rate >100 bpm reduces filling time)
- Disappears entirely at heart rate >180 bpm
-
Atrial Systole (80-100 ms)
- Atrial kick contributes 20-30% of ventricular filling
- A-wave velocity: 0.4-0.8 m/sec (normally < E-wave)
- Loss in atrial fibrillation reduces cardiac output 15-25%
- Critical in diastolic dysfunction: Atrial contribution increases to 40%
- Mitral stenosis: Atrial systole generates LA pressure >25 mmHg to overcome obstruction
📌 Remember: "E-A-Tau-DT" captures diastolic assessment-E-wave (early filling), A-wave (atrial kick), Tau (relaxation), DT (deceleration time)-each abnormality defines specific dysfunction patterns.
| Diastolic Pattern | E/A Ratio | Deceleration Time | E/e' Ratio | LA Pressure | Clinical Significance |
|---|---|---|---|---|---|
| Normal | 1.0-2.0 | 160-240 ms | <8 | Normal | Preserved function |
| Impaired Relaxation | <0.8 | >240 ms | <8 | Normal | Grade I, early HFpEF |
| Pseudonormal | 0.8-2.0 | 160-200 ms | 9-14 | Elevated | Grade II, requires tissue Doppler |
| Restrictive | >2.0 | <140 ms | >14 | High | Grade III-IV, poor prognosis |
| Irreversible | >2.5 | <120 ms | >20 | Very high | Transplant consideration |
💡 Master This: Diastolic dysfunction progresses through four grades as relaxation impairment worsens and LA pressure rises-Grade I (impaired relaxation) is reversible, but restrictive patterns (Grade III-IV) carry 50% 5-year mortality.
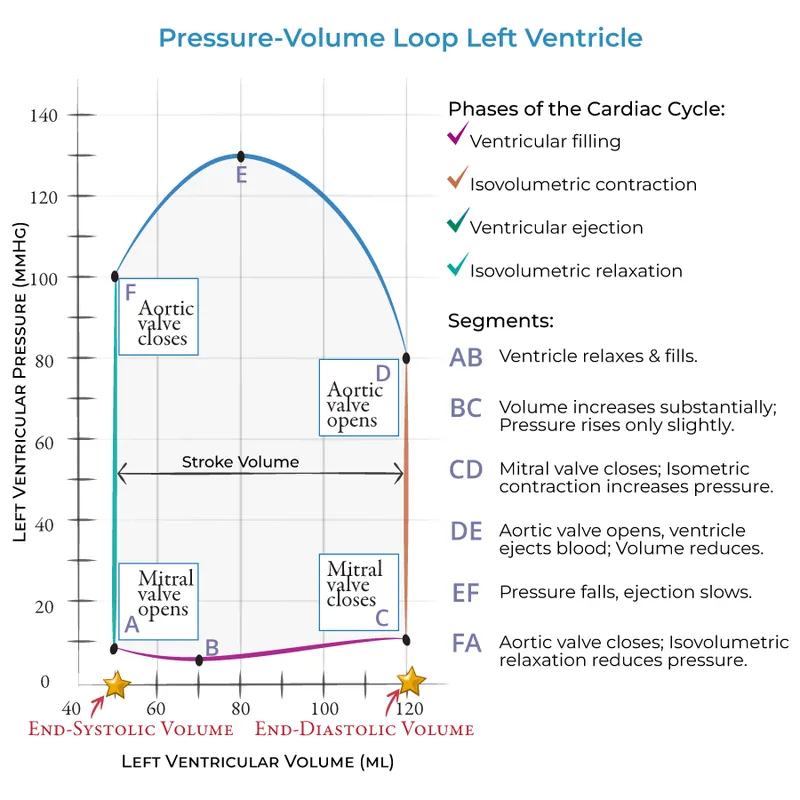
Pressure-Volume Loop Integration
The PV loop graphically represents the cardiac cycle's work and efficiency:
-
Loop Components
- Width = stroke volume (60-100 mL)
- Height = developed pressure (~120 mmHg)
- Area = stroke work (~10,000 mmHg·mL or ~1.3 J)
- End-systolic pressure-volume relationship (ESPVR): Contractility index (slope)
- End-diastolic pressure-volume relationship (EDPVR): Compliance curve (exponential)
-
Pathologic Loop Alterations
- Aortic stenosis: Increased height (afterload), normal width, increased area (work)
- Mitral regurgitation: Increased width (preload), normal height, leftward shift
- Dilated cardiomyopathy: Rightward shift (↑ EDV), reduced height (↓ contractility), wide loop
- Restrictive cardiomyopathy: Narrow loop, steep EDPVR (↓ compliance), normal ESPVR early
⭐ Clinical Pearl: Cardiac power output (mean arterial pressure × cardiac output / 451) <0.6 W predicts in-hospital mortality >40% in cardiogenic shock-integrates both pressure and flow failure.
These mechanical principles explain why tachycardia disproportionately impairs diastolic dysfunction (reduced filling time), why atrial fibrillation destabilizes mitral stenosis (loss of atrial kick), and why beta-blockers improve heart failure (prolonged diastole, reduced oxygen demand).
📚 The Cardiac Cycle Engine: Systolic & Diastolic Machinery
🔬 Electrical Conduction: The Cardiac Pacemaker Hierarchy
The heart's intrinsic conduction system generates and propagates electrical impulses through specialized tissues with distinct automaticity rates and refractory periods. Understanding this hierarchy predicts arrhythmia mechanisms, localizes conduction blocks, and guides antiarrhythmic therapy.
Pacemaker Hierarchy & Escape Rhythms
Automaticity decreases progressively from SA node to Purkinje fibers, creating backup pacemakers:
-
Sinoatrial (SA) Node - Primary pacemaker
- Intrinsic rate: 60-100 bpm (autonomic modulation: 40-180 bpm)
- Location: Right atrial junction with SVC
- Blood supply: SA nodal artery (RCA 60%, LCx 40%)
- Sinus node recovery time: <1500 ms (prolonged in sick sinus syndrome)
- P-wave morphology: Upright II, III, aVF; biphasic V1
-
Atrioventricular (AV) Node - Gatekeeper & delay
- Intrinsic rate: 40-60 bpm if SA node fails
- AV nodal delay: 50-120 ms (PR interval 120-200 ms)
- Blood supply: AV nodal artery from RCA 90%
- Dual AV nodal pathways: Fast (long refractory) + Slow (short refractory) = AVNRT substrate
- Wenckebach point: Heart rate where 1:1 conduction fails (~180-200 bpm)
-
Bundle of His & Bundle Branches
- His-Purkinje rate: 30-40 bpm (escape rhythm if AV block)
- Right bundle: Thin, anterior, vulnerable to RV pressure overload
- Left bundle: Bifascicular (anterior + posterior), thick
- Intraventricular conduction time: <120 ms (QRS duration)
- RBBB: QRS >120 ms, rSR' in V1-V2 (M-shaped)
- LBBB: QRS >120 ms, broad R in I, aVL, V5-V6 (loss of septal q-waves)
-
Purkinje Network
- Fastest conduction: 2-4 m/sec (rapid ventricular activation)
- Intrinsic rate: 20-40 bpm (idioventricular rhythm)
- Extensive arborization: Synchronous contraction
- Purkinje cells vulnerable to ischemia: VT/VF substrate
- Refractory period: 200-400 ms (protects against rapid atrial rates)
📌 Remember: "SA-60, AV-50, His-40, Purkinje-30"-memorize intrinsic rates to identify escape rhythms and predict hemodynamic tolerance of bradycardia.

⭐ Clinical Pearl: Inferior MI with AV block is usually nodal (narrow QRS, escape rate 40-60 bpm, transient, resolves in 2-7 days)-anterior MI with AV block is infranodal (wide QRS, escape rate <40 bpm, permanent pacemaker needed in 80%).
Action Potential Phases & Refractory Periods
Ionic currents shape action potential morphology and determine excitability:
-
Phase 0 - Rapid Depolarization
- Fast Na+ channels open: -90 → +20 mV in <2 ms
- Responsible for QRS complex
- Blocked by Class I antiarrhythmics (Na+ channel blockers)
- Conduction velocity: Atrial 1 m/sec, Purkinje 2-4 m/sec
- Reduced phase 0 slope: Widened QRS, slowed conduction (hyperkalemia, Class Ic drugs)
-
Phase 1 - Early Repolarization
- Transient K+ outward current
- Creates J-point notch on ECG
- Exaggerated in Brugada syndrome, early repolarization patterns
-
Phase 2 - Plateau Phase
- L-type Ca2+ influx balances K+ efflux: 200-400 ms duration
- Generates ST segment
- Prolonged by Class III antiarrhythmics (K+ channel blockers)
- Shortened in hypercalcemia: QTc <340 ms
- Lengthened in hypokalemia, hypocalcemia: QTc >500 ms (TdP risk)
-
Phase 3 - Repolarization
- K+ efflux dominates: Voltage returns to -90 mV
- Generates T-wave
- Determines refractory period duration
- Absolute refractory period: No stimulus can depolarize (Phase 0-3 early)
- Relative refractory period: Strong stimulus may depolarize (Phase 3 late) = vulnerable period for R-on-T phenomenon
-
Phase 4 - Resting Potential
- Na+/K+-ATPase restores ionic gradients
- Pacemaker cells show spontaneous diastolic depolarization (automaticity)
- Slope determines firing rate: Steep in SA node, flat in ventricles
💡 Master This: QT interval represents ventricular action potential duration (Phase 0-3)-QTc >500 ms increases torsades de pointes risk exponentially (2-3% per 10 ms prolongation above 500 ms), while QTc <340 ms predicts ventricular fibrillation in Brugada syndrome.
Autonomic Modulation & Arrhythmia Substrates
Sympathetic and parasympathetic tone dynamically regulate heart rate, conduction, and refractoriness:
-
Parasympathetic (Vagal) Effects
- Acetylcholine → M2 muscarinic receptors
- SA node: ↓ firing rate to 40-50 bpm (sinus bradycardia)
- AV node: ↑ refractory period, ↓ conduction velocity (↑ PR interval)
- Vagal maneuvers: Carotid massage, Valsalva → terminate AVNRT/AVRT
- Vagal tone dominant at rest: Explains sinus arrhythmia (rate varies with respiration)
-
Sympathetic (Adrenergic) Effects
- Norepinephrine → β1 receptors
- SA node: ↑ firing rate to 100-180 bpm (sinus tachycardia)
- AV node: ↓ refractory period, ↑ conduction velocity (↓ PR interval)
- Exercise, stress, catecholamines: Unmask conduction disease
- Beta-blockers: Slow rate, prolong AV conduction (treat SVT, rate control AF)
-
Arrhythmia Mechanisms
- Automaticity: Enhanced (triggered by ischemia, catecholamines) or suppressed (sick sinus)
- Triggered Activity: Early afterdepolarizations (EADs) in long QT, delayed afterdepolarizations (DADs) in digoxin toxicity
- Reentry: Requires unidirectional block + slow conduction + recovered excitability
- Reentry circuit length = conduction velocity × refractory period
- AVNRT: Dual AV nodal pathways with different refractory periods
- Atrial flutter: Macro-reentry around tricuspid annulus (cycle length ~200 ms, rate ~300 bpm)
| Arrhythmia Mechanism | Substrate | Trigger | Clinical Example | Treatment Target |
|---|---|---|---|---|
| Enhanced Automaticity | Ischemia, stretch | Catecholamines | Sinus tachycardia | Beta-blockers |
| Triggered Activity (EAD) | Prolonged QT | Pause-dependent | Torsades de pointes | Shorten QT, Mg2+, pacing |
| Triggered Activity (DAD) | Ca2+ overload | Digoxin, ischemia | Ventricular ectopy | Stop digoxin, beta-blockers |
| Reentry (Micro) | Scar, fibrosis | PVC | Ventricular tachycardia | Ablation, antiarrhythmics |
| Reentry (Macro) | Anatomic circuit | Atrial stretch | Atrial flutter | Cavotricuspid isthmus ablation |
📌 Remember: "Auto-Trigger-Reentry" covers all arrhythmia mechanisms-Automaticity (ectopic focus), Triggered (afterdepolarizations), Reentry (circus movement)-identifying mechanism guides therapy selection.
Understanding these electrical principles explains why adenosine terminates AVNRT (blocks AV node transiently), why amiodarone prolongs QT (blocks K+ channels), and why ablation targets slow pathways in AVNRT or isthmus in atrial flutter.
🔬 Electrical Conduction: The Cardiac Pacemaker Hierarchy
🏥 Hemodynamic Profiles: Matching Physiology to Clinical Syndromes
Integrating cardiac output, filling pressures, and systemic vascular resistance creates hemodynamic profiles that categorize heart failure phenotypes and guide targeted therapy. The Forrester classification and Stevenson profiles transform invasive measurements into actionable clinical frameworks.
Forrester Classification: Tissue Perfusion × Congestion Matrix
Originally developed for acute MI, this 2×2 matrix stratifies patients by cardiac index and pulmonary capillary wedge pressure:
-
Subset I - Warm & Dry (Compensated)
- Cardiac index: >2.2 L/min/m²
- PCWP: <18 mmHg
- Clinical: No rales, no peripheral hypoperfusion
- Mortality: 3% in-hospital
- Management: Standard post-MI care, no specific hemodynamic intervention
-
Subset II - Warm & Wet (Pulmonary Congestion)
- Cardiac index: >2.2 L/min/m²
- PCWP: >18 mmHg
- Clinical: Rales, orthopnea, elevated JVP
- Mortality: 9% in-hospital
- Management: Diuretics, vasodilators (nitroglycerin, nitroprusside)
-
Subset III - Cold & Dry (Peripheral Hypoperfusion)
- Cardiac index: <2.2 L/min/m²
- PCWP: <18 mmHg
- Clinical: Cool extremities, narrow pulse pressure, hypovolemia
- Mortality: 23% in-hospital
- Management: Cautious fluid challenge, identify cause (RV infarction, hypovolemia)
-
Subset IV - Cold & Wet (Cardiogenic Shock)
- Cardiac index: <2.2 L/min/m²
- PCWP: >18 mmHg
- Clinical: Pulmonary edema + hypoperfusion + hypotension
- Mortality: 51% in-hospital without mechanical support
- Management: Inotropes (dobutamine), mechanical circulatory support (IABP, Impella), urgent revascularization
⭐ Clinical Pearl: Forrester Subset IV (cold & wet) with cardiac index <1.8 L/min/m² and PCWP >25 mmHg defines cardiogenic shock-mortality approaches 80% without mechanical support or revascularization within 90 minutes.

📌 Remember: "Warm-Cold = Perfusion, Wet-Dry = Congestion"-assess perfusion (skin temperature, mentation, urine output) and congestion (rales, JVP, edema) to assign profile and therapy.
Stevenson Hemodynamic Profiles: Bedside Assessment
Simplified clinical profiles guide outpatient heart failure management without invasive monitoring:
-
Profile A - Warm & Dry
- Well-compensated, euvolemic
- Clinical: No rales, JVP <8 cm, no edema
- Target profile for chronic management
- Maintain with guideline-directed medical therapy (GDMT)
- Uptitrate ACE-I/ARB/ARNI, beta-blockers, MRA
-
Profile B - Warm & Wet
- Congested but perfused
- Clinical: Rales, JVP >8 cm, edema, orthopnea
- Most common decompensation pattern (70-80% of admissions)
- Management: IV diuretics (furosemide 2.5× home dose), consider ultrafiltration if diuretic resistance
- Target: Net negative 3-5 L over 3-5 days, hemoconcentration (↑ Hct, ↑ albumin)
-
Profile C - Cold & Dry
- Hypoperfused without congestion
- Clinical: Low pulse pressure (<25% SBP), cool extremities, fatigue
- Uncommon (<10% of admissions), often overdiuresed
- Management: Reduce diuretics, cautious fluids, address low output (inotropes if severe)
-
Profile L - Cold & Wet
- Congested AND hypoperfused
- Clinical: Profile B signs + hypotension, worsening renal function, narrow pulse pressure
- Advanced heart failure, 20-30% of admissions
- Management: Inotropes (dobutamine, milrinone), consider MCS, transplant evaluation
- Mortality: 15-20% in-hospital, 40-50% at 1 year
| Profile | Congestion | Perfusion | JVP | Rales | SBP | Pulse Pressure | Therapy | 1-Year Mortality |
|---|---|---|---|---|---|---|---|---|
| A (Dry-Warm) | No | Adequate | <8 cm | No | >90 | >25% | Optimize GDMT | <10% |
| B (Wet-Warm) | Yes | Adequate | >8 cm | Yes | >90 | >25% | IV diuretics | 10-20% |
| C (Dry-Cold) | No | Low | <8 cm | No | <90 | <25% | Reduce diuretics | 20-30% |
| L (Wet-Cold) | Yes | Low | >8 cm | Yes | <90 | <25% | Inotropes, MCS | 40-50% |
💡 Master This: Pulse pressure (SBP - DBP) <25% of SBP identifies low cardiac output with 90% specificity-a systolic 90 mmHg with diastolic 70 mmHg (pulse pressure 20 mmHg, 22%) signals Profile C or L requiring inotropic support.
Invasive Hemodynamic Parameters & Swan-Ganz Interpretation
Pulmonary artery catheter measurements quantify cardiac performance and guide advanced therapy:
-
Cardiac Output & Index
- Cardiac output (CO): 4-8 L/min (stroke volume × heart rate)
- Cardiac index (CI): 2.5-4.0 L/min/m² (CO / BSA)
- Stroke volume: 60-100 mL/beat
- CI <2.2 L/min/m²: Hypoperfusion, consider inotropes
- CI <1.8 L/min/m²: Cardiogenic shock, mechanical support indicated
-
Filling Pressures
- Right atrial pressure (RAP): 0-8 mmHg
- PCWP: 6-12 mmHg (LA pressure surrogate)
- LVEDP: Usually PCWP + 2-4 mmHg
- PCWP >18 mmHg: Pulmonary congestion (dyspnea threshold)
- PCWP >25 mmHg: Pulmonary edema (alveolar flooding)
-
Vascular Resistances
- Systemic vascular resistance (SVR): 800-1200 dyne·sec/cm⁵
- SVR = (MAP - RAP) / CO × 80
- Elevated SVR >1500: Afterload mismatch, use vasodilators (nitroprusside)
- Pulmonary vascular resistance (PVR): <250 dyne·sec/cm⁵ (<3 Wood units)
- PVR = (mPAP - PCWP) / CO × 80
- Elevated PVR >480 (>6 Wood units): Pulmonary hypertension, evaluate reversibility
- Systemic vascular resistance (SVR): 800-1200 dyne·sec/cm⁵
-
Mixed Venous Oxygen Saturation (SvO2)
- Normal: 65-75% (from PA catheter)
- Low SvO2 <60%: ↑ oxygen extraction, ↓ cardiac output
- High SvO2 >75%: Sepsis (impaired extraction), left-to-right shunt, high output states
⭐ Clinical Pearl: SvO2 <50% with CI <2.0 L/min/m² defines refractory cardiogenic shock-mortality >70% without mechanical circulatory support (Impella, ECMO) or urgent transplant.
Understanding these hemodynamic profiles transforms undifferentiated "heart failure" into specific phenotypes with targeted therapies-warm-wet patients need diuresis, cold-wet patients need inotropes, and cold-dry patients need cautious rehydration.
🏥 Hemodynamic Profiles: Matching Physiology to Clinical Syndromes
🔍 Valvular Hemodynamics: Stenosis vs. Regurgitation Physiology
Valvular lesions impose distinct hemodynamic burdens-stenosis creates pressure overload with concentric remodeling, while regurgitation causes volume overload with eccentric dilation. Understanding these compensatory mechanisms and decompensation thresholds guides surgical timing.
Aortic Stenosis: The Pressure-Overloaded Ventricle
Progressive valve narrowing forces the LV to generate supranormal pressures, triggering concentric hypertrophy:
-
Severity Grading by Hemodynamics
- Mild AS: Valve area 1.5-2.0 cm², mean gradient <25 mmHg, peak velocity 2.6-3.0 m/sec
- Moderate AS: Valve area 1.0-1.5 cm², mean gradient 25-40 mmHg, peak velocity 3.0-4.0 m/sec
- Severe AS: Valve area <1.0 cm², mean gradient >40 mmHg, peak velocity >4.0 m/sec
- Critical AS: Valve area <0.6 cm², mean gradient >50 mmHg, peak velocity >5.0 m/sec
- Low-flow, low-gradient AS: Valve area <1.0 cm², mean gradient <40 mmHg, EF <50%-requires dobutamine stress echo to assess true severity
-
Compensatory Mechanisms
- Concentric LV hypertrophy: Wall thickness ↑ to normalize wall stress
- Preserved EF: 55-70% maintained until late decompensation
- Prolonged ejection time: >300 ms (normal 200-250 ms)
- LV mass index: Often >150 g/m² (normal <115 g/m² men, <95 g/m² women)
- Diastolic dysfunction: Impaired relaxation from hypertrophy, ↑ LVEDP
-
Decompensation Triggers
- Symptom onset: Angina, syncope, dyspnea (average survival 2-3 years without AVR)
- EF decline: <50% signals irreversible myocardial damage
- Mean gradient drop: Paradoxically decreases as contractility fails (pseudo-severity)
- Sudden death risk: 1-2% per year in asymptomatic severe AS
- Rapid progression: Velocity increases 0.3 m/sec per year, valve area decreases 0.1 cm² per year
⭐ Clinical Pearl: Severe AS with symptoms (angina, syncope, dyspnea) carries 50% 2-year mortality without valve replacement-symptom onset is absolute indication for AVR regardless of age.

📌 Remember: "CAVE" for AS symptoms-Chest pain (angina, 5-year survival), Activity intolerance (dyspnea, 3-year survival), **
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app



















