CRS Fundamentals - Defining the Debulk
- CRS: Surgical removal of all visible tumor (peritoneal metastases).
- Aim: Maximal tumor debulking for optimal outcome.
- Significance: Enhances adjuvant therapy efficacy (e.g., HIPEC), ↑ survival.
- Completeness of Cytoreduction (CC) Score:
- CC-0: No visible disease (Goal).
- CC-1: Nodules < 2.5 mm.
- CC-2: Nodules 2.5 mm-2.5 cm.
- CC-3: Nodules > 2.5 cm.
⭐ Achieving CC-0 (no visible residual disease) in CRS is the most crucial factor for long-term survival.
Patient Selection - Who Gets CRS?
- Goal: Achieve complete cytoreduction (CC-0/CC-1).
- Key Considerations:
- Good performance status (ECOG 0-1).
- No major comorbidities precluding major surgery.
- Disease biology: Low-grade tumors respond better.
- Limited extra-peritoneal disease.
- Possibility of achieving complete cytoreduction.
⭐ The Peritoneal Cancer Index (PCI) score is a critical preoperative assessment tool to quantify disease burden and determine resectability in peritoneal surface malignancies. A PCI score < 20 is often a favorable prognostic factor.
The CRS Procedure - Surgical Showdown
- Objective: Achieve complete macroscopic tumor removal.
- Core Components:
- Peritonectomy: Removal of diseased peritoneum.
- Visceral Resections: Targeted removal of involved organs (e.g., omentum, spleen, bowel segments).
- Completeness of Cytoreduction (CC) Score:
- CC-0: No visible disease.
- CC-1: Residual tumor < 2.5 mm.
- CC-2: Residual tumor 2.5 mm - 2.5 cm.
- CC-3: Residual tumor > 2.5 cm.
⭐ CC-0 (no macroscopic residual disease) is the single most important prognostic factor for survival in CRS patients.
HIPEC & Adjuvants - The Chemo Cocktail
- HIPEC (Hyperthermic Intraperitoneal Chemotherapy):
- Heated chemo (e.g., Mitomycin C, Cisplatin, Oxaliplatin) circulated in peritoneum. 📌 My Cousin Owes.
- Temperature: 41-43°C; Duration: 60-90 minutes.
- Goal: Eradicate microscopic residual tumor cells post-CRS.
- Mechanism: Hyperthermia ↑ drug penetration & cytotoxicity.
- Other Adjuvants:
- EPIC: Early Postoperative Intraperitoneal Chemotherapy (non-heated).
- NIPEC: Normothermic Intraperitoneal Chemotherapy.
- Systemic Chemotherapy: Neoadjuvant or adjuvant.
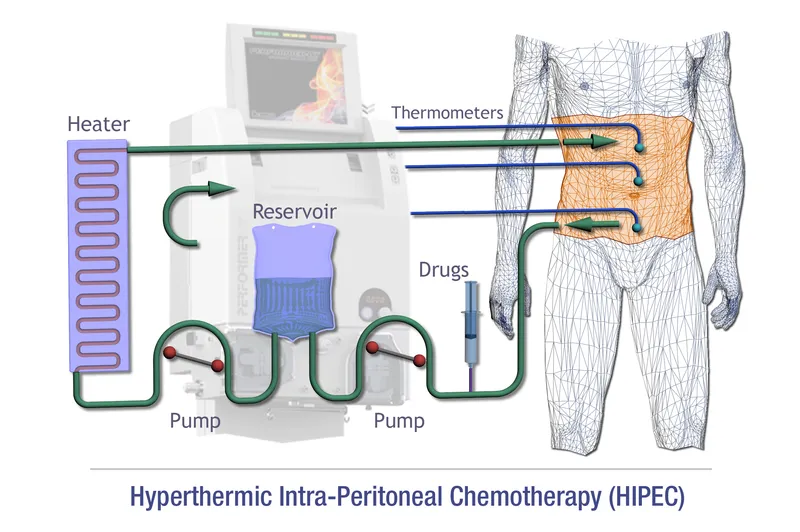
⭐ Hyperthermia (typically 41-43°C) during HIPEC enhances the penetration and cytotoxic effect of chemotherapeutic agents like Mitomycin C or Cisplatin in the peritoneal cavity.
Outcomes & Complications - The Aftermath
- Outcomes:
- ↑ Median survival & DFS in select patients (e.g., appendiceal, ovarian, mesothelioma).
- Prognostic Factors:
- Completeness of Cytoreduction (CC-0/1 ideal).
- Peritoneal Cancer Index (PCI <20 is favorable).
- Tumor histology, grade, & careful patient selection.
- Complications:
- Morbidity: Significant, reported 30-50%; Mortality: <5% in experienced, high-volume centers.
- Common: Anastomotic leak, prolonged ileus, infections (SSI, pneumonia), myelosuppression (esp. post-HIPEC), DVT/PE, fistulas.
- Quality of Life (QoL): Initially ↓, typically improves over several months.
⭐ Major morbidity after CRS and HIPEC can be significant (up to 30-50%), with common complications including anastomotic leaks, infections, and myelosuppression; however, in experienced centers, mortality is <5%.
High‑Yield Points - ⚡ Biggest Takeaways
- CRS (Cytoreductive Surgery) targets no gross residual disease (CC-0) in peritoneal malignancies, often combined with HIPEC.
- Main indications: Appendiceal, colorectal, ovarian cancers, and peritoneal mesothelioma.
- Peritoneal Cancer Index (PCI) is vital for selection; PCI >20 often excludes patients.
- Completeness of Cytoreduction (CC score) is the strongest predictor of survival.
- HIPEC uses heated chemotherapy (e.g., Mitomycin C, Cisplatin) to target microscopic cells post-CRS.
- Associated with significant morbidity; requires specialized, high-volume centers for optimal outcomes.
- Sugarbaker's principles often guide the peritonectomy techniques employed during CRS.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

