Body Fluids & Basics - Wet & Wild World
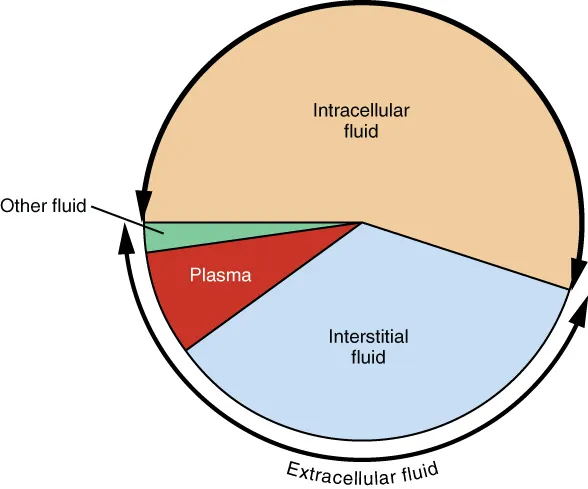
- TBW: 60% of body weight. 📌 60-40-20 Rule:
- ICF: 40% (K+ dominant)
- ECF: 20% (Na+ dominant); Interstitial 15%, Intravascular 5%.
- Electrolyte Distribution:
- ICF: High K+, Mg2+, Phosphates.
- ECF: High Na+, Cl-, HCO3-.
- Osmolality: Solute concentration per kg solvent. Plasma Osmolality $ \approx (2 \times Na^+) + (Glucose/18) + (BUN/2.8) $. Normal: 280-295 mOsm/kg.
- Tonicity: Describes ECF effect on cells. Solutions: Isotonic (e.g., NS, RL), Hypotonic (0.45% NaCl), Hypertonic (3% NaCl).
- Starling Forces: Capillary hydrostatic & oncotic pressures govern fluid exchange.
⭐ The primary determinant of ECF osmolality is Sodium concentration.
IV Fluids - Drip Drop Hydrate
- Classification:
- Crystalloids: Small molecules (NS, RL, D5W). Types: Isotonic (0.9%NS, RL), Hypotonic (0.45%NS, D5W in-vivo), Hypertonic (3%NaCl).
- Colloids: Large molecules (Albumin, HES).
- Common Fluids Overview:
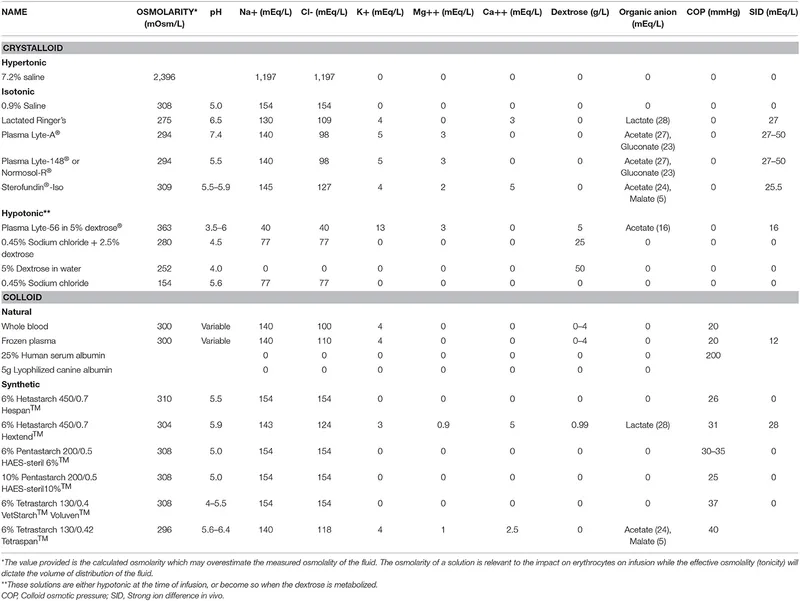
- 0.9% NS (Normal Saline): 154 Na+/Cl- mEq/L. Osm 308 mOsm/L. Use: Resuscitation. Risk: Hyperchloremic acidosis.
- RL (Ringer's Lactate): 130 Na+, 4 K+, 28 Lactate (mEq/L). Osm 273 mOsm/L. Use: Burns, trauma. ⚠️ Liver failure, hyperK+.
- D5W (5% Dextrose in Water): Osm 278 mOsm/L (in bag). Use: Free water. ⚠️ ↑ICP, hyperglycemia.
- Complications: Fluid overload, electrolyte imbalance (e.g., NS → hyperchloremic acidosis), colloid reactions.
⭐ Ringer's Lactate is most physiological but contains K+ (4 mEq/L); use cautiously in hyperkalemia or severe renal failure.
Fluid Therapy - Quenching the Thirst
- Maintenance Fluids:
- Holliday-Segar (daily): $100 \text{ml/kg for first } 10\text{kg} + 50 \text{ml/kg for next } 10\text{kg} + 20 \text{ml/kg for rest} \text{ per day}$. (4/2/1 rule hourly).
- Adults: 25-35 ml/kg/day.
- Daily Electrolytes (mEq/kg/day): Na⁺ 1-2, K⁺ 0.5-1, Cl⁻ 1-2.
- Assess Fluid Status:
- Clinical: HR, BP, UO (target 0.5-1 ml/kg/hr), JVP, skin turgor, capillary refill.
- Labs: Hct, BUN/Cr ratio.
- Deficit Replacement: Correct 50% in first 8h, remaining over next 16h.
- Insensible Losses: Approx. 400-600 ml/day; ↑ with fever, tachypnea.
⭐ In surgical patients, third space losses can significantly increase fluid requirements.
Electrolyte Imbalances - Ion Power Play
| Ion | Hypo (Sx, ECG, Mgmt, Formula) | Hyper (Sx, ECG, Mgmt) |
|---|---|---|
| Na+ | <135 mEq/L (Sev <120). CNS sx. 3% NaCl if severe. Na Deficit: $(TargetNa - ActualNa) \times TBW_L$. | >145 mEq/L. CNS sx. D5W / free H2O. |
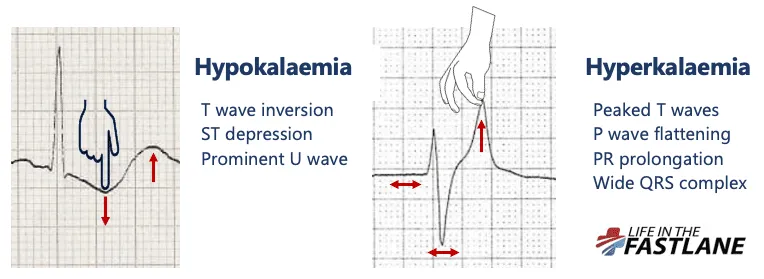
| K+ | <3.5 mEq/L. Weakness, U wave, flat T. Oral/IV KCl (Rate ≤10-20 mEq/hr). | >5.0 mEq/L. 📌 MURDER. Peaked T, ↑QRS. Ca-gluc, Insulin+D50, Salbutamol, Loop diuretic. |
| Ca2+ | <8.5 mg/dL. 📌 CATS go numb. ↑QT. IV Ca-gluc. $Ca_{corr} = Ca_{measured} + 0.8 \times (4.0 - Alb_{g/dL})$. | >10.5 mg/dL. "Stones, bones, groans...". ↓QT. Hydration, bisphosphonates. |
HyperK Mgmt
Acid-Base Balance - pH Balance Act
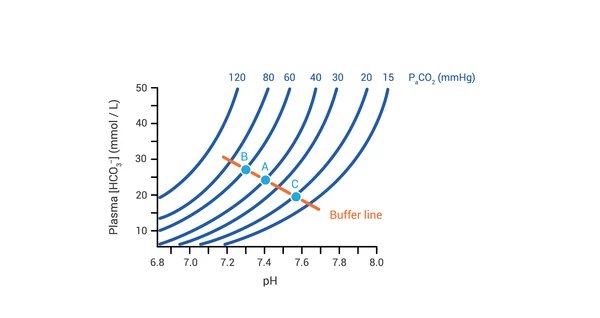
- Normal: pH 7.35-7.45, PaCO2 35-45 mmHg, HCO3 22-26 mEq/L.
- Steps: 1. pH? (Acidemia/Alkalemia) 2. Primary? (Resp/Metab) 3. Compensated?
- Anion Gap: $AG = Na^+ - (Cl^- + HCO_3^-)$ (Normal: 8-12). 📌 MUDPILES/GOLDMARK for ↑AG.
| Disorder | pH | PaCO2 | HCO3 | Key Cause(s) |
|---|---|---|---|---|
| Metabolic Acidosis | ↓ | ↓ | ↓ | DKA, Lactic Acid |
| Metabolic Alkalosis | ↑ | ↑ | ↑ | Vomiting, Diuretics |
| Respiratory Acidosis | ↓ | ↑ | ↑ | Hypoventilation |
| Respiratory Alkalosis | ↑ | ↓ | ↓ | Hyperventilation |
| %%{init: {'flowchart': {'htmlLabels': true}}}%% | ||||
| flowchart TD |
Start["🔬 ABG Analysis
• Arterial blood gas• Check acid-base"]
PHVal{"📋 pH Value?
• Measure serum pH• Identify acidity"}
Normal["✅ Normal/Comp
• pH 7.35 to 7.45• Homeostasis met"]
AcidCause{"📋 Acid Cause?
• pH < 7.35• Acidemia state"}
AlkCause{"📋 Alk Cause?
• pH > 7.45• Alkalemia state"}
RespAcid["🩺 Resp Acidosis
• ⬆️ PaCO2 levels• Hypoventilation"]
MetAcid["🩺 Metab Acidosis
• ⬇️ HCO3 levels• Renal/metabolic"]
RespAlk["🩺 Resp Alkalosis
• ⬇️ PaCO2 levels• Hyperventilation"]
MetAlk["🩺 Metab Alkalosis
• ⬆️ HCO3 levels• GI/renal loss"]
AnionGap{"📋 Anion Gap?
• Na - Cl + HCO3• Gap calculation"}
HAGMA["⚠️ HAGMA
• High Gap Acidosis• MUDPILES causes"]
NAGMA["🩺 NAGMA
• Normal Gap Acid• Diarrhea/RTA"]
Comp["👁️ Compensation
• Assess response• Winters formula"]
Start --> PHVal PHVal -->|Normal| Normal PHVal -->|Acidemia| AcidCause PHVal -->|Alkalemia| AlkCause
AcidCause -->|⬆️ PaCO2| RespAcid AcidCause -->|⬇️ HCO3| MetAcid
AlkCause -->|⬇️ PaCO2| RespAlk AlkCause -->|⬆️ HCO3| MetAlk
MetAcid --> AnionGap AnionGap -->|High| HAGMA AnionGap -->|Normal| NAGMA
RespAcid --> Comp MetAcid --> Comp RespAlk --> Comp MetAlk --> Comp
style Start fill:#FFF7ED, stroke:#FFEED5, stroke-width:1.5px, rx:12, ry:12, color:#C2410C style PHVal fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style Normal fill:#F6F5F5, stroke:#E7E6E6, stroke-width:1.5px, rx:12, ry:12, color:#525252 style AcidCause fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style AlkCause fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style RespAcid fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style MetAcid fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style RespAlk fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style MetAlk fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style AnionGap fill:#FEF8EC, stroke:#FBECCA, stroke-width:1.5px, rx:12, ry:12, color:#854D0E style HAGMA fill:#FDF4F3, stroke:#FCE6E4, stroke-width:1.5px, rx:12, ry:12, color:#B91C1C style NAGMA fill:#F7F5FD, stroke:#F0EDFA, stroke-width:1.5px, rx:12, ry:12, color:#6B21A8 style Comp fill:#EEFAFF, stroke:#DAF3FF, stroke-width:1.5px, rx:12, ry:12, color:#0369A1
> ⭐ Winters' formula ($Expected PaCO_2 = 1.5 \times [HCO_3^-] + 8 \pm 2$) for resp. compensation in metabolic acidosis.
## High‑Yield Points - ⚡ Biggest Takeaways
> * **Daily fluid needs**: **25-35 mL/kg**; Na+ **1-2 mmol/kg**, K+ **0.5-1 mmol/kg**.
> * **Isotonic crystalloids** (NS, RL) are primary for initial **volume resuscitation**.
> * **Hypokalemia** is the most common surgical electrolyte imbalance, often from **GI losses**.
> * Monitor **urine output (>0.5 mL/kg/hr)** as a key indicator of adequate **hydration & perfusion**.
> * **Hypertonic saline (3% NaCl)** for severe symptomatic **hyponatremia** or ↑ICP; administer cautiously.
> * **Third space fluid loss** is a major contributor to **postoperative hypovolemia**.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

