Radiation Interaction with Cells - Target Hit!
- Cellular damage mechanisms:
- Direct Action: Radiation directly ionizes critical cellular target (DNA). Predominant for high-LET radiation (α-particles, neutrons).
- Indirect Action: Radiation ionizes water (main cell constituent), creating free radicals that diffuse to damage DNA.
- Primary mechanism for low-LET radiation (X-rays, γ-rays).
- Key radical: $\cdot OH$ (hydroxyl) - most damaging.
- Water radiolysis yields: $\cdot OH, H\cdot, e_{aq}^-$.
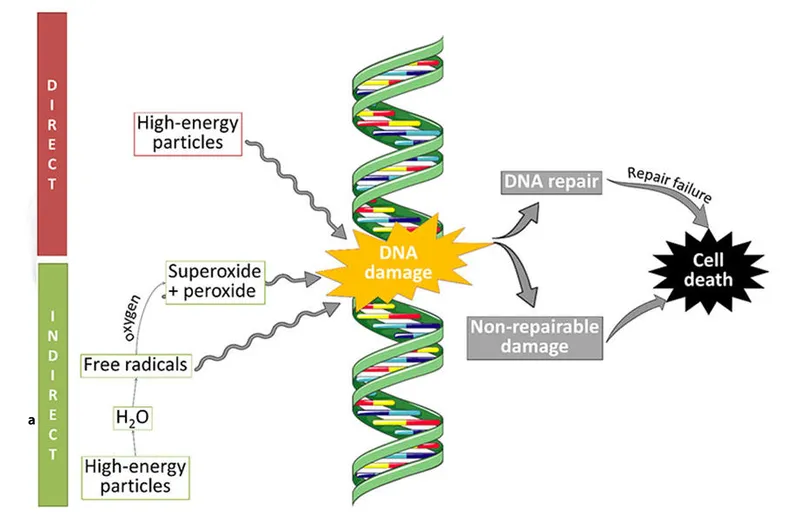
⭐ For X & γ-rays, ~2/3 DNA damage is indirect, mainly via $\cdot OH$ radicals.
Types of DNA Lesions - Molecular Scars
- Single-Strand Breaks (SSBs):
- Frequent, efficiently repaired (e.g., by BER).
- Lower biological significance than DSBs.
- Common from indirect action of low-LET radiation.
- Double-Strand Breaks (DSBs):
- Most lethal; primary cause of cell killing, chromosomal aberrations, mutations.
- Repair is complex (NHEJ, HRR).
- Caused by direct action or clustered damage (high-LET).
- Base Damage:
- Oxidation (e.g., 8-oxoguanine), alkylation.
- Repaired by BER; mutagenic if misrepaired.
- Crosslinks:
- DNA-Protein Crosslinks (DPCs).
- Intra-strand & Inter-strand crosslinks (ICLs).
- Block replication & transcription.
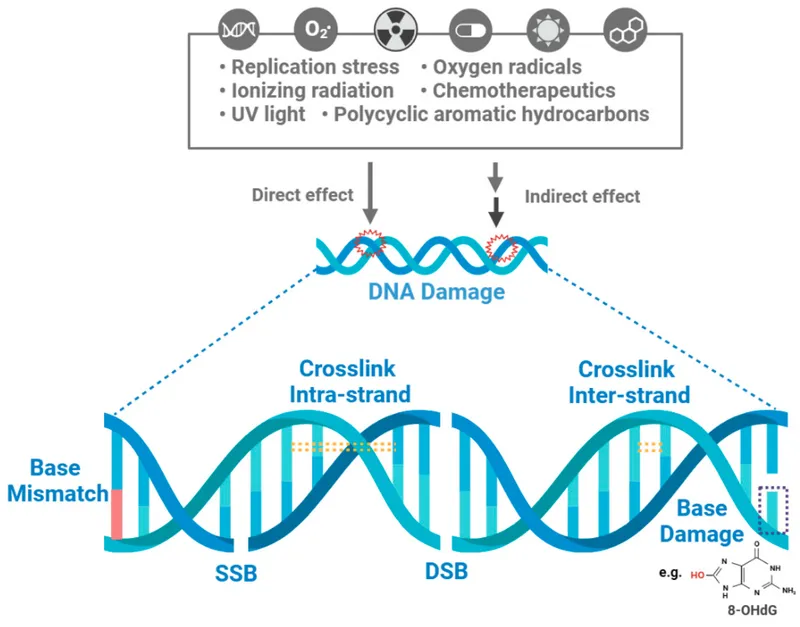
⭐ Double-Strand Breaks (DSBs) are the most critical lesions for radiation-induced cell death and genomic instability. Misrepair leads to mutations and chromosomal aberrations.
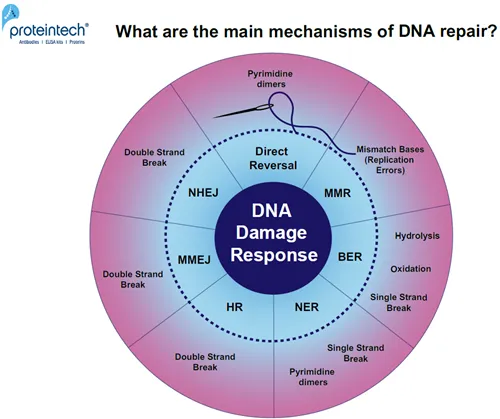
DNA Repair Pathways - Cellular ER
- Cells use diverse pathways to mend DNA damage, vital for survival.
- Base Excision Repair (BER):
- Repairs single base damage (oxidation, alkylation).
- Key enzymes: Glycosylases, AP endonuclease, DNA Pol β, Ligase.
- High fidelity.
- Nucleotide Excision Repair (NER):
- Removes bulky adducts (e.g., UV-induced pyrimidine dimers).
- Excises damaged DNA segment.
- High fidelity.
- Double-Strand Break (DSB) Repair:
- Homologous Recombination (HR):
- Error-free; uses sister chromatid as template.
- Predominant in S/G2 phase.
- Key proteins: BRCA1/2, RAD51.
- Non-Homologous End Joining (NHEJ):
- Error-prone; directly ligates broken ends.
- Active throughout cell cycle (esp. G0/G1).
- Key proteins: Ku70/80, DNA-PKcs.
- 📌 Mnemonic: "No Homie? Just Join!"
- Homologous Recombination (HR):
⭐ NHEJ is the predominant pathway for repairing DNA double-strand breaks in mammalian cells, especially outside of S/G2 phase.
Cellular Fates & Modifiers - Damage Outcomes
-
Cellular Fates (Post-Irradiation):
- Cell Cycle Arrest: Checkpoints (G1/S, G2/M) for DNA repair.
- Apoptosis: Programmed cell death (e.g., via p53).
- Senescence: Irreversible growth arrest.
- Mitotic Catastrophe: Cell death due to aberrant mitosis.
- Genomic Instability: ↑Mutations, chromosomal aberrations → carcinogenesis.
-
Modifiers of Radiation Effect:
- LET (Linear Energy Transfer): ↑LET → ↑RBE (Relative Biological Effectiveness).
- Dose: ↑Dose → ↑Effect.
- Dose Rate: ↓Dose rate → ↓Effect (more time for repair).
- Oxygen Effect: O₂ "fixes" free radical damage, making it permanent.
- OER (Oxygen Enhancement Ratio) = $D_{hypoxia} / D_{aerated}$ for same biological effect.
- Value: ~2.5-3.5 for X-rays/γ-rays (low-LET).
- ↓OER with ↑LET (approaches 1 for high-LET like α-particles).
⭐ Oxygen is the most potent chemical radiosensitizer; the OER is maximal for low-LET radiation and significantly reduced for high-LET radiation.
High‑Yield Points - ⚡ Biggest Takeaways
- Indirect action (via •OH radicals) is primary for X-rays/gamma rays.
- Direct action dominates for high-LET radiation (alpha, neutrons).
- DSBs are most biologically significant/lethal DNA lesions.
- SSBs are the most frequent DNA damage.
- Repair: BER for SSBs; NHEJ & HR for DSBs.
- Most radiosensitive: M & G2 phases; most radioresistant: late S phase.
- Oxygen enhances damage (OER ~2.5-3.5 for low-LET).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

