Bicarbonate Buffer System - pH's Main Guard
- Definition: The body's primary, rapid-acting chemical buffer system, crucial for maintaining pH homeostasis, especially in the ECF.
- Components:
- Weak Acid: Carbonic Acid ($H_2CO_3$) - formed from $CO_2$ and $H_2O$.
- Conjugate Base: Bicarbonate ion ($HCO_3^-$) - can accept $H^+$.
- Significance: Most important buffer in Extracellular Fluid (ECF).
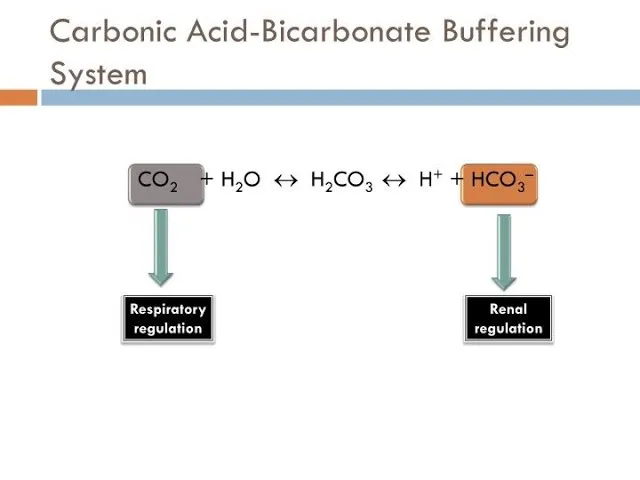
- Key Chemical Equation: $CO_2 + H_2O \rightleftharpoons H_2CO_3 \rightleftharpoons H^+ + HCO_3^-$
- This reversible reaction is catalyzed by carbonic anhydrase.
⭐ The bicarbonate buffer system is the most important buffer in the extracellular fluid (ECF), handling moment-to-moment pH changes.
Bicarbonate Buffer System - How It Buffers
The body's most important extracellular buffer, rapidly neutralizing pH shifts.
-
Neutralizing Added Acid (e.g., $H^+$ from metabolic acidosis):
- $H^+$ (acid) + $HCO_3^-$ (bicarbonate) $\rightarrow$ $H_2CO_3$ (carbonic acid).
- Effect: $H^+$ is consumed, preventing a sharp pH drop.
- $H_2CO_3$ $\xrightarrow{\text{CA or spontaneous}}$ $H_2O$ + $CO_2$ (carbon dioxide).
- Effect: $CO_2$ is exhaled by lungs, effectively removing acid.
- $H^+$ (acid) + $HCO_3^-$ (bicarbonate) $\rightarrow$ $H_2CO_3$ (carbonic acid).
-
Neutralizing Added Base (e.g., $OH^-$ from alkali ingestion):
- $H_2CO_3$ (carbonic acid) + $OH^-$ (base) $\rightarrow$ $HCO_3^-$ (bicarbonate) + $H_2O$.
- Effect: $H_2CO_3$ donates $H^+$, neutralizing the base.
- $CO_2$ + $H_2O$ $\xrightarrow{\text{Carbonic Anhydrase}}$ $H_2CO_3$.
- Effect: Replenishes $H_2CO_3$, maintaining buffer capacity.
- $H_2CO_3$ (carbonic acid) + $OH^-$ (base) $\rightarrow$ $HCO_3^-$ (bicarbonate) + $H_2O$.
-
Role of Carbonic Anhydrase (CA):
- Enzyme; massively accelerates the reversible reaction: $CO_2 + H_2O \rightleftharpoons H_2CO_3$.
- Crucial for the system's rapid response to acid-base challenges in blood and tissues.
⭐ The pKa of the carbonic acid/bicarbonate system is 6.1.
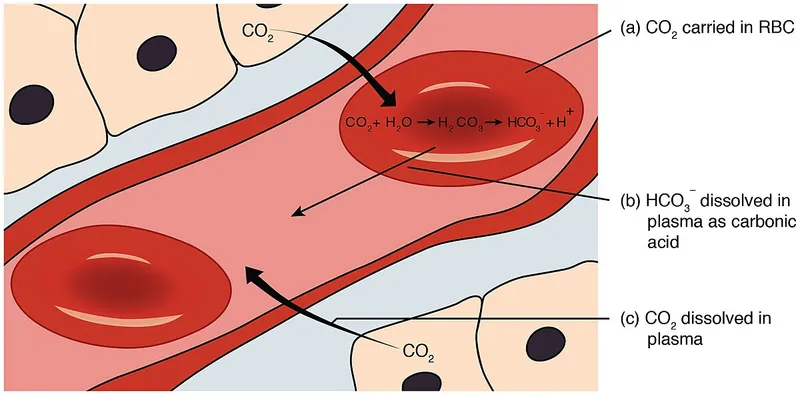
Bicarbonate Buffer System - Organ Control Crew
The body's principal ECF buffer, vital for pH homeostasis. Lungs and kidneys meticulously regulate its components: $CO_2 + H_2O \leftrightarrow H_2CO_3 \leftrightarrow H^+ + HCO_3^-$.
- Lungs (Respiratory Control): The Fast Responders
- Rapidly adjust $PCO_2$ (normal: 35-45 mmHg) by altering alveolar ventilation.
- ↑ Ventilation (blows off $CO_2$) → ↓ $PCO_2$ → ↑ pH.
- ↓ Ventilation (retains $CO_2$) → ↑ $PCO_2$ → ↓ pH.
- Kidneys (Renal Control): The Sustained Regulators
- Slowly but powerfully manage $H^+$ excretion (e.g., as $NH_4^+$) and $HCO_3^-$ (normal: 22-26 mEq/L) reabsorption/regeneration.
- Essential for long-term acid-base balance.
⭐ Respiratory compensation is rapid (minutes to hours), while renal compensation is slower (hours to days) but more powerful in restoring acid-base balance.
Bicarbonate Buffer System - Clinic & H-H Equation
- Dominant extracellular fluid buffer; maintains pH homeostasis.
- Henderson-Hasselbalch equation: $pH = pKa + \log \frac{[HCO_3^-]}{[0.03 \times PCO_2]}$
- Relates pH to bicarbonate ([HCO3-]) and PCO2.
- Clinical Application & Normal Values:
- pH: 7.35-7.45 (Overall acid-base status)
- PCO2: 35-45 mmHg (Respiratory component, regulated by lungs)
- HCO3-: 22-26 mEq/L (Metabolic component, regulated by kidneys)
- Used to diagnose acidosis/alkalosis and assess compensation.
⭐ At physiological pH 7.4, the ratio of [HCO3-] to dissolved CO2 (H2CO3) is approximately 20:1, maximizing buffer capacity.
High‑Yield Points - ⚡ Biggest Takeaways
- Most important extracellular buffer in the body.
- Composed of weak acid H₂CO₃ (carbonic acid) and conjugate base HCO₃⁻ (bicarbonate).
- Carbonic anhydrase enzyme crucial for rapid interconversion.
- An open system: CO₂ regulated by lungs, HCO₃⁻ by kidneys.
- pH determined by Henderson-Hasselbalch equation (ratio of HCO₃⁻ to dissolved CO₂).
- pKₐ is 6.1; highly effective due to high concentration and volatile CO₂ removal.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

