Biology of Aging: Introduction & Theories - Aging Defined
- Aging (Senescence): Universal, progressive, intrinsic decline in physiological function across lifespan; results in ↑ vulnerability to disease & ↑ mortality. Natural process, not a disease.
- Key Domains of Aging:
- Chronological: Passage of time since birth.
- Biological (Physiological): Changes in body structure & function.
- Psychological: Alterations in cognition, emotion, behavior.
- Social: Shifts in societal roles, relationships, support.
- Fundamental Concepts:
- Homeostenosis: Gradual narrowing of homeostatic reserves, reducing the ability to cope with stressors.
- Cellular Senescence: State of irreversible cell cycle arrest, contributing to tissue aging.
⭐ Hayflick Limit: Normal human somatic cells have a limited replicative capacity (typically ~40-60 divisions in vitro) before entering senescence. This phenomenon underscores the finite lifespan of individual cells.
Biology of Aging: Cellular Hallmarks Pt. 1 - Microscopic Mayhem
- Genomic Instability: Increased accumulation of DNA damage.
- Sources: Exogenous (UV radiation, chemicals), Endogenous (Reactive Oxygen Species, DNA replication errors).
- Consequences: Point mutations, chromosomal rearrangements, aneuploidy.
- Declining efficiency of DNA repair mechanisms (e.g., BER, NER).
- Telomere Attrition: Progressive shortening of telomeres (protective chromosome ends).
- Composed of TTAGGG repeats; shorten with each cell division (end-replication problem).
- Reaches Hayflick limit → triggers cellular senescence or apoptosis.
⭐ Telomerase, an enzyme that adds telomeric DNA, shows high activity in germ cells and cancer cells, but is typically low in most somatic cells.
- Epigenetic Alterations: Heritable changes in gene function without altering DNA sequence.
- Includes: DNA methylation (hyper/hypo), histone modifications (acetylation, methylation), chromatin remodeling.
- Leads to "epigenetic drift," causing dysregulated gene expression patterns with age. oka
Biology of Aging: Cellular Hallmarks Pt. 2 - Cell's Inner Chaos
- Loss of Proteostasis (Protein Homeostasis):
- Impaired protein folding, ↑ aggregation (e.g., amyloid, tau).
- Reduced clearance: ↓ autophagy (cellular cleaning), ↓ proteasome activity.
- Chronic Endoplasmic Reticulum (ER) stress.
⭐ Activation of UPR (Unfolded Protein Response) is a key cellular response to loss of proteostasis.
- Deregulated Nutrient Sensing:
- Insulin/IGF-1 Signaling (IIS) pathway: ↓ activity (e.g., via caloric restriction) promotes longevity.
- mTOR (mechanistic Target of Rapamycin): ↓ activity extends lifespan, promotes autophagy.
- AMPK & Sirtuins (longevity pathways): Activation (e.g., by metformin, resveratrol) ↑ healthspan.
- Mitochondrial Dysfunction:
- ↓ ATP synthesis, ↑ Reactive Oxygen Species ($ROS$) leakage, oxidative damage.
- Accumulation of mitochondrial DNA (mtDNA) mutations.
- Defective mitophagy (clearance of damaged mitochondria).
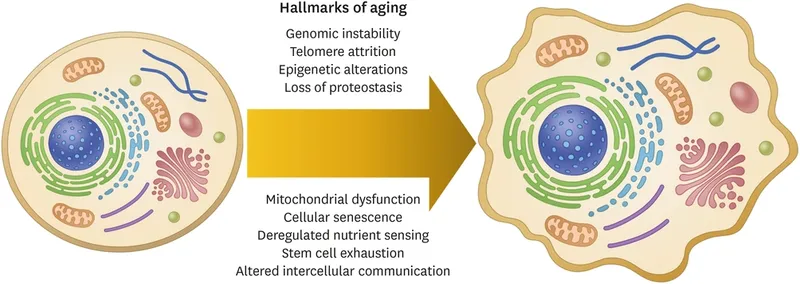
Biology of Aging: Integrative Hallmarks & Interventions - Systemic Slowdown & Fixes
- Integrative Hallmarks: Culmination of earlier damage, leading to system-level decline.
- Stem cell exhaustion: ↓ regenerative capacity, impaired tissue repair & organ maintenance.
- Altered intercellular communication:
- Inflammaging: Chronic, sterile, low-grade inflammation (↑ pro-inflammatory cytokines).
- Endocrine dysregulation: e.g., ↓ GH/IGF-1 axis, insulin resistance.
- Neurohormonal signaling changes.
- Gut dysbiosis.
- Interventions: Aim to modulate aging pathways & improve healthspan.
- Lifestyle: Caloric restriction (CR), CR mimetics (e.g., resveratrol), exercise.
- Pharmacological: Senolytics (e.g., Dasatinib + Quercetin), Metformin, Rapamycin (mTOR inhibitor).
- Emerging: Stem cell-based therapies, gene editing (e.g., telomerase activation).
⭐ Cellular senescence contributes to age-related diseases through the Senescence-Associated Secretory Phenotype (SASP), promoting inflammation and tissue damage.
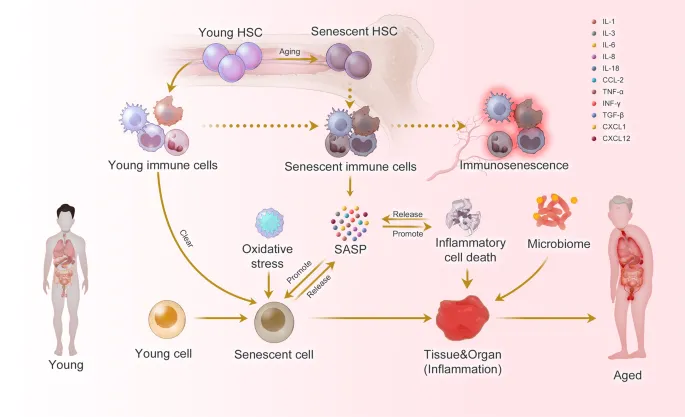
High‑Yield Points - ⚡ Biggest Takeaways
- Cellular senescence: Irreversible arrest; markers p16INK4a, SA-β-gal; drives age-related pathology.
- Telomere shortening: Limits cell divisions (Hayflick limit); low telomerase in somatic cells.
- Oxidative stress: ROS damage (DNA, proteins, lipids); mitochondrial dysfunction is key source.
- Genomic instability: ↑DNA damage, mutations; impaired DNA repair accelerates aging.
- Epigenetic alterations: DNA methylation, histone modifications alter gene expression with age.
- Loss of proteostasis: Impaired protein folding/clearance; toxic protein accumulation.
- Deregulated nutrient sensing: mTOR, Insulin/IGF-1 pathways; caloric restriction extends lifespan.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

