Amino Acid Structure - Alpha's Anatomy
- Fundamental Unit: Each amino acid has a central α-carbon (Cα) bonded to four distinct groups:
- An amino group ($-NH_2$)
- A carboxyl group ($-COOH$)
- A hydrogen atom ($-H$)
- A variable R-group (side chain), unique to each, defining its properties.
- General formula: $H_2N-CHR-COOH$.
- L-Amino Acids Predominate: Proteins consist almost exclusively of L-amino acids.
- 📌 CORN rule (L-isomers): With H atom away, COOH, R, NH2 groups are clockwise around Cα.
- Chirality: All amino acids (except glycine) possess a chiral α-carbon (asymmetric center), making them optically active.
⭐ Glycine is the only achiral amino acid; its R-group is a hydrogen atom, so its α-carbon is not asymmetric.
 oka
oka
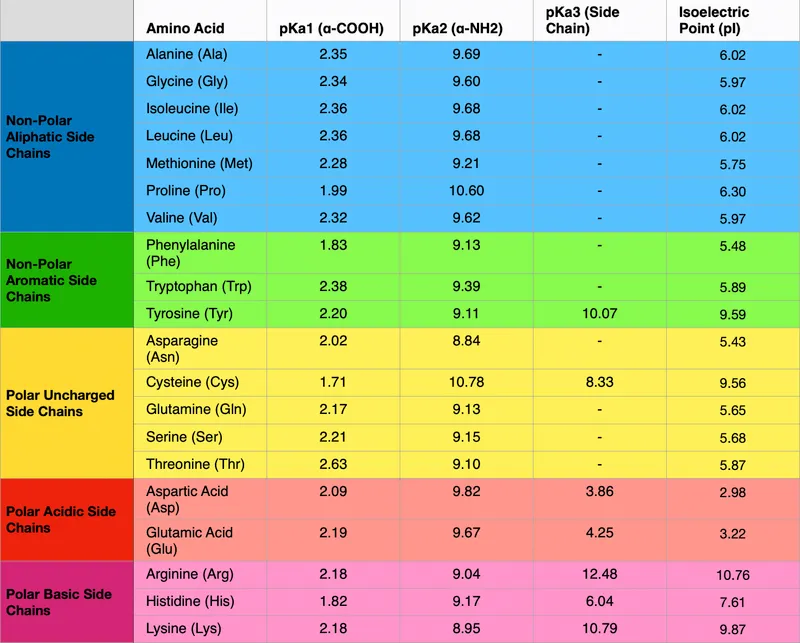
Amino Acid Classification - Side Chain Symphony
Amino acids are classified based on the polarity and charge of their side chains (R-groups) at physiological pH (~7.4). This dictates their interactions and protein structure.

| Class | Sub-class | Key Characteristics | Examples (1-L, 3-L) |
|---|---|---|---|
| Nonpolar | Aliphatic | Hydrophobic R | G, Gly; A, Ala; V, Val; L, Leu; I, Ile; M, Met; P, Pro |
| Aromatic | Hydrophobic aromatic R | F, Phe; W, Trp | |
| Polar Uncharged | Polar R, uncharged; hydrophilic | S, Ser (-OH); T, Thr (-OH); Y, Tyr (-OH); N, Asn; Q, Gln; C, Cys (-SH) | |
| Polar Acidic | -COO^- R; net negative charge | D, Asp; E, Glu | |
| Polar Basic | -NH_3^+ R; net positive charge | K, Lys; R, Arg; H, His (can be neutral/positive) 📌 HAL: Histidine, Arginine, Lysine are basic. |
AA Properties & Special Types - pH & Special Stars
-
Acid-Base Properties:
- Amino acids are amphoteric, existing as zwitterions ($R-CH(NH_3^+)-COO^-$) at physiological pH.
⭐ At physiological pH (~7.4), amino acids exist as zwitterions.
- Isoelectric point (pI): pH where net charge is zero. For neutral AAs: $pI = (pK_1 + pK_2)/2$.
- Typical pKa values: α-carboxyl ~2.0; α-amino ~9.0-10.0.
- Buffering action: Resist pH changes near their pKa values due to ionizable groups.
- Amino acids are amphoteric, existing as zwitterions ($R-CH(NH_3^+)-COO^-$) at physiological pH.
-
Special Amino Acids:
- Proline (P): Imino acid; its rigid ring structure disrupts α-helices (helix breaker).
- Cysteine (C): Contains a sulfhydryl (-SH) group; forms disulfide bonds (-S-S-), crucial for protein tertiary structure.
- Selenocysteine (U): The 21st amino acid; contains selenium; vital for specific antioxidant enzymes like glutathione peroxidase.
-
Essential & Non-Essential AAs:
- Essential AAs (must be obtained from diet): 📌 PVT TIM HALL (Phenylalanine, Valine, Threonine, Tryptophan, Isoleucine, Methionine, Histidine, Arginine *, Leucine, Lysine).
*Arginine is semi-essential. - Non-essential AAs are synthesized by the body (e.g., Alanine, Aspartate, Glutamate).
- Essential AAs (must be obtained from diet): 📌 PVT TIM HALL (Phenylalanine, Valine, Threonine, Tryptophan, Isoleucine, Methionine, Histidine, Arginine *, Leucine, Lysine).
Peptide Bond Formation - Bond & Build
- Formation: Amino acids link via peptide bonds ($-CO-NH-$) by dehydration synthesis (condensation), releasing H₂O.
- Characteristics:
- Rigid & Planar: Due to partial double bond character.
- Trans configuration favored (less steric hindrance).
- Restricted rotation around peptide bond; free around α-carbons.
- Polypeptide Chain:
- N-terminus: Free -NH₃⁺ group.
- C-terminus: Free -COO⁻ group.
- Read N → C terminus.
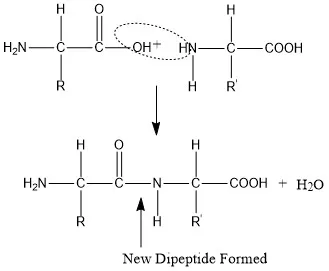
⭐ The peptide bond has partial double bond character, making it rigid and planar, which is crucial for protein folding.
High‑Yield Points - ⚡ Biggest Takeaways
- Glycine: only achiral amino acid; others are L-isomers.
- Proline: imino acid, forms kinks in polypeptide chains.
- Essential AAs (PVT TIM HALL) are dietary requirements.
- Aromatic AAs (Trp, Tyr, Phe) absorb UV at 280 nm.
- Cysteine forms disulfide bonds, stabilizing protein structure.
- Basic AAs (Arg, Lys, His) are +ve charged; Acidic AAs (Asp, Glu) -ve charged at physiological pH.
- Amino acids are zwitterions (dipolar ions) at physiological pH_._
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

