Enzyme Inhibition - Enzyme Slowdown
- Enzyme inhibitors are molecules that bind to enzymes and ↓ their activity.
- Crucial for metabolic regulation and drug action.
- Classified based on reversibility:
- Reversible Inhibition:
- Inhibitor binds non-covalently.
- Enzyme activity can be restored by removing inhibitor.
- Includes competitive, non-competitive, and uncompetitive types.
- Irreversible Inhibition:
- Inhibitor binds covalently (usually).
- Causes permanent enzyme inactivation.
- Examples: Aspirin (COX), organophosphates (AChE), heavy metals. ⭐ > Many drugs act as enzyme inhibitors; e.g., statins inhibit HMG-CoA reductase.
- Reversible Inhibition:
Competitive Inhibition - Active Site Battle
- Mechanism: Inhibitor (I) structurally resembles substrate (S).
- Competes directly with S for binding to the enzyme's active site (E).
- Reversible: Effect overcome by ↑ [S].
- Kinetic Impact:
- $K_m$: ↑ (Apparent $K_m$ increases; more S needed to reach 1/2 $V_{max}$ as enzyme affinity for S appears to decrease).
- $V_{max}$: Unchanged (With sufficient [S], I is outcompeted, and the original $V_{max}$ is reached).
- Lineweaver-Burk Plot: Lines intersect on the Y-axis (1/$V_{max}$ is unchanged).
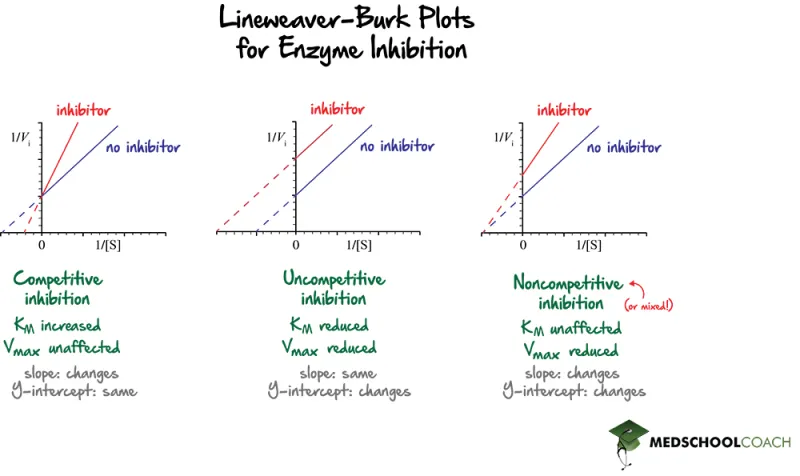
- Clinical Examples:
- Statins (e.g., Atorvastatin) inhibit HMG-CoA reductase (hypercholesterolemia treatment).
- Methotrexate inhibits Dihydrofolate reductase (cancer chemotherapy, rheumatoid arthritis).
- Ethanol for methanol poisoning (competes for alcohol dehydrogenase).
⭐ Competitive inhibitors increase the apparent $K_m$ of the enzyme for the substrate but do not alter the $V_{max}$ achievable at saturating substrate concentrations.
Non-competitive Inhibition - Allosteric Attack
- Inhibitor (I) binds to an allosteric site (distinct from the active site).
- Binds to Enzyme (E) or Enzyme-Substrate (ES) complex with equal affinity.
- Reduces catalytic efficiency; prevents product (P) formation at normal rate.
- Kinetic Effects:
- ↓$V_{max}$ (effectively ↓ concentration of functional enzyme).
- $K_m$ unchanged (substrate affinity to active site is not affected).
- Cannot be overcome by increasing substrate concentration [S].
- Lineweaver-Burk Plot:
- Lines (inhibited vs. uninhibited) intersect on the x-axis.
- Y-intercept (1/$V_{max}$) increases.
- X-intercept (-1/$K_m$) is unchanged.
- Examples: Lead (e.g., on ferrochelatase, ALA dehydratase), Alanine (on pyruvate kinase), Pepstatin (on aspartic proteases).
- 📌 Mnemonic: "Non-Compete? $K_m$ No Change, $V_{max}$ Vanishes (↓)."
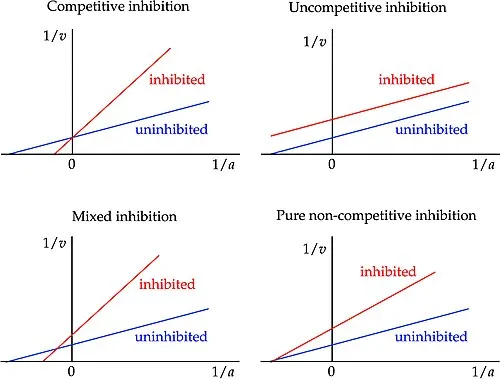
⭐ Non-competitive inhibitors effectively reduce the number of functional enzyme molecules, thus lowering $V_{max}$, but do not interfere with substrate binding to the active site ($K_m$ unchanged).
Inhibitor Comparison - Key Differences
| Feature | Competitive Inhibition | Non-competitive Inhibition |
|---|---|---|
| Binding Site | Binds Active Site; directly competes with substrate. | Binds Allosteric Site; distinct from substrate binding. |
| $K_m$ (Affinity) | ↑ (Apparent $K_m$ ↑); affinity for substrate appears ↓. | Unchanged; enzyme affinity for substrate not affected. |
| $V_{max}$ (Efficacy) | Unchanged; can be reached with high [S]. | ↓ (Decreases); enzyme efficacy reduced, cannot reach normal $V_{max}$. |
| Overcome by ↑ [S]? | Yes; high [S] displaces inhibitor. | No; inhibition effect is independent of [S]. |
High‑Yield Points - ⚡ Biggest Takeaways
- Competitive inhibitors bind active site, ↑ Km (↓ affinity), Vmax unchanged; overcome by ↑ [S].
- Non-competitive inhibitors bind allosteric site, Km unchanged, ↓ Vmax; not overcome by ↑ [S].
- Lineweaver-Burk: Competitive lines intersect on Y-axis; Pure non-competitive lines intersect on X-axis.
- Km is [S] at ½ Vmax; reflects substrate affinity.
- Vmax is maximum reaction velocity at saturating [S].
- Example (Competitive): Statins (vs HMG-CoA reductase).
- Example (Non-competitive): Lead (vs ferrochelatase).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

