O2 Carriage: Basics - The O2 Taxi Service
- $O_2$ transported in blood in two forms:
- Dissolved $O_2$ (~2%): Directly in plasma; $PaO_2 \times \mathbf{0.003}$. Contributes to partial pressure.
- Bound to Hemoglobin (Hb) (~98%): Main transport mechanism.
- Hemoglobin (Hb):
- Protein in Red Blood Cells (RBCs); 4 heme groups, each binds one $O_2$ molecule.
- Normal Hb: M: 13-17 g/dL, F: 12-15 g/dL.
- $1g \text{ Hb binds } \mathbf{1.34 mL} O_2$.
- $O_2$ Carrying Capacity: Maximum $O_2$ blood can carry if Hb is 100% saturated.
- $O_2$ Content ($CaO_2$): Total $O_2$ in arterial blood.
- Formula: $CaO_2 = (Hb \times \mathbf{1.34} \times SaO_2/100) + (PaO_2 \times \mathbf{0.003})$.
- $SaO_2$: % of Hb saturated with $O_2$.
- $PaO_2$: Partial pressure of $O_2$ in arterial blood.
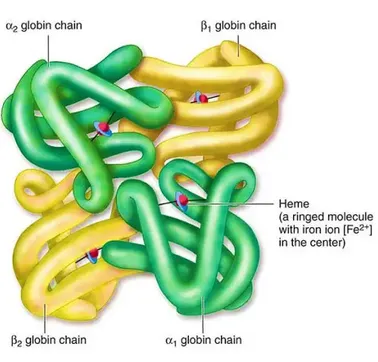
⭐ Each Hb molecule can bind up to four $O_2$ molecules, exhibiting cooperative binding. The binding of one $O_2$ molecule increases the affinity of Hb for subsequent $O_2$ molecules, facilitating efficient oxygen uptake in the lungs.
O2 Dissociation Curve - Letting Go Gracefully
The Oxygen-Hemoglobin Dissociation Curve (OHDC) is a sigmoid-shaped graph crucial for understanding $O_2$ transport. It plots hemoglobin's $O_2$ saturation against the partial pressure of oxygen ($PO_2$), detailing Hb's $O_2$ binding in lungs and release in tissues.
- P50 Value: The specific $PO_2$ at which hemoglobin is 50% saturated with $O_2$. This value inversely reflects Hb's affinity for oxygen.
- Normal adult P50: 26.6 mmHg.
- An ↑P50 indicates ↓ $O_2$ affinity (right shift of the curve), enhancing $O_2$ delivery to tissues.
- A ↓P50 indicates ↑ $O_2$ affinity (left shift of the curve), promoting $O_2$ retention by Hb.
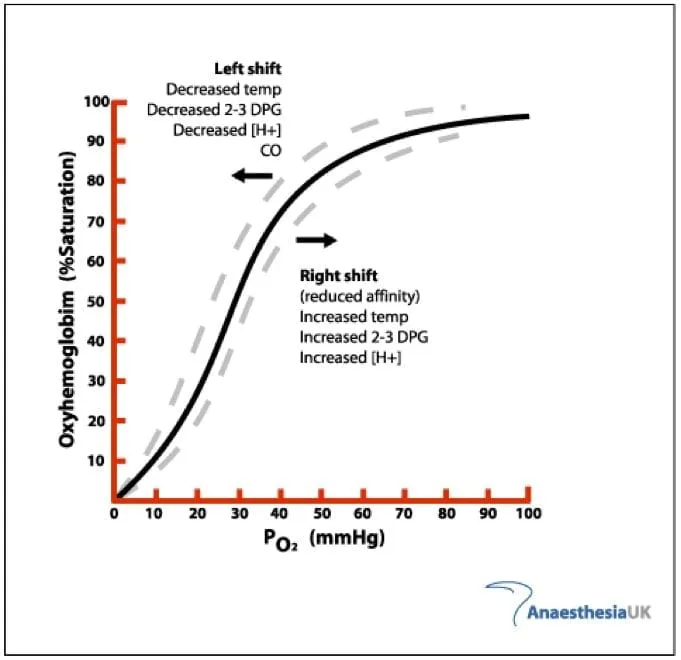
The mermaid diagram below details factors causing these shifts:
⭐ The OHDC's sigmoid shape is vital: its steep portion allows for substantial $O_2$ unloading to tissues with minor $PO_2$ drops, while the flat upper portion ensures high $O_2$ saturation in lungs despite $PO_2$ fluctuations.
CO2 Transport Mechanisms - The Waste Disposal Unit
-
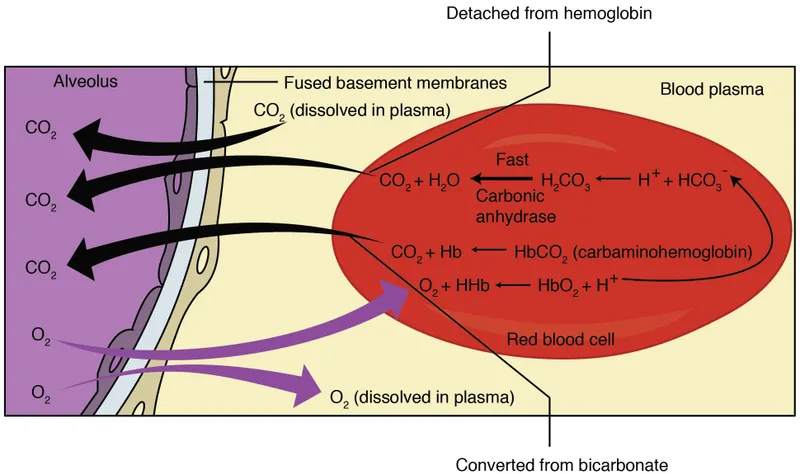
CO2 is transported in blood in three forms:
- As Bicarbonate ions ($HCO_3^-$): ~70%
- $CO_2 + H_2O \xrightarrow{\text{Carbonic Anhydrase (in RBC)}} H_2CO_3 \leftrightarrow H^+ + HCO_3^-$
- $HCO_3^-$ moves from RBC to plasma.
- As Carbaminohemoglobin: ~23%
- $CO_2$ binds to globin part of Hb: $CO_2 + Hb \cdot NH_2 \leftrightarrow Hb \cdot NH \cdot COOH$
- Favored by deoxygenated Hb.
- Dissolved $CO_2$: ~7%
- Directly in plasma, contributes to $PCO_2$.
- As Bicarbonate ions ($HCO_3^-$): ~70%
-
Chloride Shift (Hamburger's Phenomenon) 📌
- To maintain electrical neutrality, $Cl^-$ enters RBC as $HCO_3^-$ exits.
- Occurs in tissues; reverse process in lungs.
- Haldane Effect
- Deoxygenated Hb has a higher affinity for $CO_2$ and is a better buffer for $H^+$.
- Oxygenation of Hb in lungs (↑ $PO_2$) promotes $CO_2$ release.
⭐ Haldane Effect: Deoxygenation of hemoglobin (e.g., in tissues) ↑ its ability to carry $CO_2$; conversely, oxygenated Hb (e.g., in lungs) has a ↓ affinity for $CO_2$ and releases it.
High‑Yield Points - ⚡ Biggest Takeaways
- Oxygen primarily binds hemoglobin (97%); 3% dissolved.
- Oxygen-Hb curve right shift (↑O₂ release): ↑PCO₂, ↑H⁺ (Bohr effect), ↑2,3-DPG, ↑Temp.
- CO₂ transport: Mainly bicarbonate (70%), carbaminoHb (23%), dissolved CO₂ (7%).
- Haldane effect: Deoxy-Hb has ↑affinity for CO₂ & H⁺, aiding CO₂ tissue loading.
- Chloride shift: HCO₃⁻ exits RBCs in tissues; Cl⁻ influx maintains electroneutrality.
- P₅₀ (~26.6 mmHg) reflects Hb-O₂ affinity; ↑P₅₀ means ↓affinity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

