Renal Acid-Base: Basics - Bicarb's Big Save
- Kidneys: Long-term acid-base balance; slower than lungs.
- Primary role: Reclaims filtered $HCO_3^-$ (major buffer), preventing its urinary loss.
- $HCO_3^-$ Reabsorption:
- ~80-90% in Proximal Convoluted Tubule (PCT).
- Rest in Thick Ascending Limb (TAL), Distal Tubule (DT), Collecting Ducts (CD).
- Indirect mechanism:
- Luminal: $H^+ + HCO_3^- \rightarrow H_2CO_3; H_2CO_3 \xrightarrow{CA_{IV}} CO_2 + H_2O$.
- Cellular: $CO_2 + H_2O \xrightarrow{CA_{II}} H_2CO_3 \rightarrow H^+ + HCO_3^-$.
- $HCO_3^-$ exits to blood (e.g., NBCe1-A in PCT).
- $H^+$ secreted to lumen (NHE3, H-ATPase).
- Key Enzymes: Carbonic Anhydrase (CA-IV luminal, CA-II cytoplasmic).
⭐ Majority of $HCO_3^-$ reabsorption occurs in the Proximal Convoluted Tubule (PCT), approximately 80-90%.
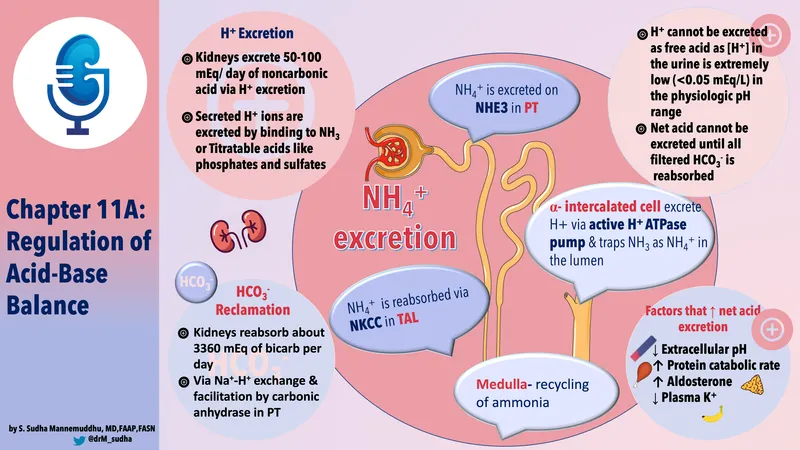
Renal Acid-Base: Acid Out - Proton Pump & Buffers
- Kidneys excrete 50-100 mEq/day of non-volatile acids, generating new $HCO_3^-$.
- H⁺ Secretion Mechanisms (Apical, Type A Intercalated Cells):
- H⁺-ATPase (Proton Pump): Directly secretes H⁺.
- H⁺-K⁺-ATPase: Secretes H⁺, reabsorbs K⁺.
- Urinary Buffers (combine with secreted H⁺ in lumen):
- Phosphate Buffer: $HPO_4^{2-} + H^+ \rightleftharpoons H_2PO_4^-$ (Titratable Acid). pKa 6.8.
- Ammonia Buffer: $NH_3 + H^+ \rightleftharpoons NH_4^+$.
- $NH_3$ from glutamine metabolism (PCT). 📌 Glutamine gives an amine.
- $NH_3$ diffuses into lumen, combines with H⁺ → $NH_4^+$ (trapped).
- Major adaptive response to acidosis.
⭐ Glutamine is the primary source of $NH_4^+$ produced by the kidneys, crucial for excreting large acid loads.
- Net Acid Excretion (NAE):
- NAE = (Titratable Acid + $NH_4^+$) - (Excreted $HCO_3^-$)
- Represents net acid eliminated & new $HCO_3^-$ gained by blood.
Renal Acid-Base: Control Knobs - Modulating Factors
Renal acid-base handling is modulated by:
- Arterial $PCO_2$:
- ↑$PCO_2$ → ↑H+ secretion, ↑$HCO_3^-$ reabsorption.
- ↓$PCO_2$ → ↓H+ secretion, ↓$HCO_3^-$ reabsorption.
- Potassium (K+):
- Hypokalemia → ↑H+ secretion, ↑$HCO_3^-$ reabsorption.
- Hyperkalemia → ↓H+ secretion, ↓$HCO_3^-$ reabsorption.
- Hormones:
- Aldosterone: ↑H+ secretion.
- Angiotensin II: ↑H+ secretion, ↑$HCO_3^-$ reabsorption (via NHE3).
- ECF Volume:
- Depletion → ↑H+ secretion, ↑$HCO_3^-$ reabsorption (via RAAS).
- Expansion → Opposite.
⭐ Hypokalemia stimulates H+ secretion and bicarbonate reabsorption, potentially leading to metabolic alkalosis.
Renal Acid-Base: Imbalance Response - Kidney to Rescue
Kidneys combat systemic acid-base disturbances by modulating three key processes: $H^+$ secretion, $HCO_3^-$ reabsorption, and $HCO_3^-$ generation (or enhanced excretion in alkalosis).
| Imbalance | Primary Defect | Renal Compensation | Expected Urine pH |
|---|---|---|---|
| Metabolic Acidosis | ↓ $HCO_3^-$ | ↑ $H^+$ secretion, ↑ $NH_4^+$ excretion, ↑ $HCO_3^-$ synthesis | < 5.5 |
| Metabolic Alkalosis | ↑ $HCO_3^-$ | ↓ $H^+$ secretion, ↓ $NH_4^+$ excretion, ↑ $HCO_3^-$ excretion | > 7.0 |
| Respiratory Acidosis | ↑ $pCO_2$ | ↑ $H^+$ secretion, ↑ $NH_4^+$ excretion, ↑ $HCO_3^-$ synthesis (slow) | < 5.5 |
| Respiratory Alkalosis | ↓ $pCO_2$ | ↓ $H^+$ secretion, ↓ $NH_4^+$ excretion, ↑ $HCO_3^-$ excretion (slow) | > 7.0 |
High‑Yield Points - ⚡ Biggest Takeaways
- Kidneys manage acid-base via H+ secretion, HCO3- reabsorption, and new HCO3- generation.
- PCT reabsorbs ~85% filtered HCO3-, aided by carbonic anhydrase.
- H+ secretion occurs in PCT, TAL, and collecting ducts (CD) via H+-ATPase & H+-K+-ATPase.
- Excretion of titratable acids (e.g., H2PO4-) and NH4+ eliminates fixed acids, regenerates HCO3-.
- Acidosis: ↑ H+ secretion, ↑ NH4+ production, ↑ HCO3- reabsorption.
- Alkalosis: ↓ H+ secretion, ↓ NH4+ production, ↑ HCO3- excretion.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more


