Drug Discovery & Preclinical - Molecule Mission
- Discovery:
- Target identification (key receptor/enzyme in disease).
- Screening (HTS, virtual) for initial 'hits'.
- Lead optimization (↑potency, ↑selectivity, favorable ADMET).
- Preclinical (Animal models; ≥2 species; Good Laboratory Practice - GLP):
- Pharmacokinetics (ADME profile).
- Pharmacodynamics (efficacy, mechanism of action - MOA).
- Toxicology:
- Acute toxicity (single dose, LD50).
- Subacute/Chronic (repeated dose effects).
- Reproductive tox. (teratogenicity, Segment II).
- Genotoxicity (mutagenic potential, Ames test).
- Carcinogenicity (long-term, 2-year rodent bioassay).
⭐ NOAEL (No Observed Adverse Effect Level) from preclinical studies is critical for determining the safe starting dose in Phase 1 human trials.
- 📌 An IND (Investigational New Drug) application to regulatory authorities (CDSCO-India, FDA-USA) is mandatory before human trials.
Clinical Trials - Human Test Voyage
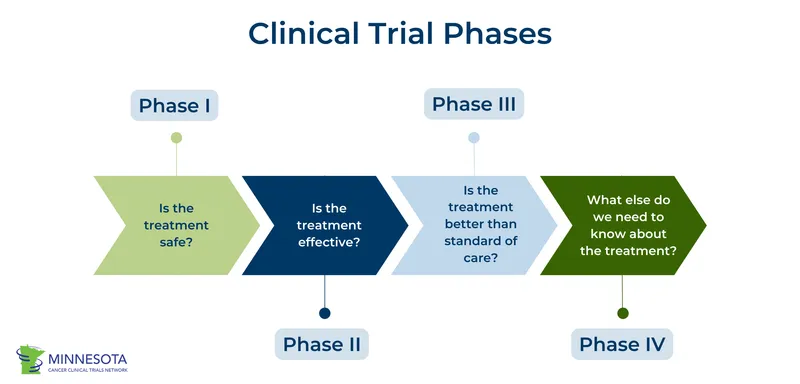
- Investigational New Drug (IND) Application: Submitted to regulatory authority (e.g., CDSCO in India) before human trials.
- Phase 0 (Exploratory/Microdosing):
- Participants: ~10-15 healthy volunteers.
- Purpose: Preliminary Pharmacokinetics (PK)/Pharmacodynamics (PD) data, not therapeutic.
- Phase I (Human Pharmacology & Safety):
- Participants: 20-80 healthy volunteers (or patients for specific conditions like cancer).
- Purpose: Assess safety, tolerability, Maximum Tolerated Dose (MTD), PK/PD.
- 📌 Mnemonic: "S" for Safety, Smallest group.
- Phase II (Therapeutic Exploratory):
- Participants: 100-300 patients with the target disease.
- Purpose: Evaluate efficacy, determine optimal dose range, identify common short-term side effects.
- Phase III (Therapeutic Confirmatory):
- Participants: 1000-3000+ diverse patients.
- Purpose: Confirm efficacy in a larger population, monitor adverse reactions, compare with standard treatment (Randomized Controlled Trials - RCTs). Forms basis for New Drug Application (NDA).
- New Drug Application (NDA): Submitted to regulatory authority for marketing approval.
- Phase IV (Post-Marketing Surveillance):
- Participants: Large patient population using the approved drug.
- Purpose: Monitor long-term safety, detect rare side effects, explore new indications, assess drug interactions. Pharmacoepidemiology.
⭐ Declaration of Helsinki: A set of ethical principles regarding human experimentation developed for the medical community. Essential for guiding clinical trial conduct globally and in India.
Indian Drug Regulation - Desi Rule Roadmap
- Primary Legislation: Drugs & Cosmetics Act, 1940 & Rules, 1945.
- Apex Body: Central Drugs Standard Control Organization (CDSCO).
- Headed by: Drug Controller General of India (DCGI).
- Functions: New drug approval, clinical trial oversight, import/export control, drug quality assurance.
- Key Supporting Bodies:
- Indian Pharmacopoeia Commission (IPC): Publishes Indian Pharmacopoeia (IP), setting drug standards.
- National Pharmaceutical Pricing Authority (NPPA): Fixes/revises drug prices via Drug Price Control Orders (DPCO).
- Crucial Schedules:
- Schedule Y: Detailed guidelines for clinical trials.
- Schedule M: Good Manufacturing Practices (GMP) requirements.
- Schedule H & H1: Prescription-only drugs; H1 requires separate register.

⭐ The Drugs and Cosmetics Act, 1940, and its subsequent Rules, 1945, form the bedrock of drug regulation in India, governing import, manufacture, distribution, and sale of drugs and cosmetics.
📌 Mnemonic: "Central Drugs Standard Control Organization Controls Drugs Safely Country Over" (CDSCO functions).
Ethical & GCP Standards - Fair Play Pharma
- Declaration of Helsinki: Guiding ethical principles for medical research involving human subjects.
-
⭐ Key ethical pillars (Belmont Report): Respect for Persons, Beneficence, Justice.
-
- Informed Consent (IC): Voluntary, informed, documented participant agreement. Essential before trial procedures.
- Institutional Review Board (IRB)/Independent Ethics Committee (IEC): Reviews & approves trial protocols; ensures subject safety & rights.
- Good Clinical Practice (GCP): ICH-GCP international standard for trial conduct, data quality, ethical compliance.
- Clinical Trial Registry - India (CTRI): Mandatory public recording of all clinical trials conducted in India.
High‑Yield Points - ⚡ Biggest Takeaways
- Clinical trial phases: Phase I (safety), Phase II (efficacy), Phase III (confirmatory), Phase IV (post-marketing).
- CDSCO, led by DCGI, is India's primary drug regulatory authority.
- Schedule Y dictates clinical trial guidelines in India.
- Orphan drugs are for rare diseases, often fast-tracked.
- IND (Investigational New Drug) application precedes human trials.
- PvPI (Pharmacovigilance Programme of India) monitors ADRs.
- NDA (New Drug Application) is for marketing approval after successful trials.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more


