Antimicrobial Arsenal - Class & Action Central
- Cell Wall Synthesis Inhibitors:
- Beta-lactams (Penicillins, Cephalosporins, Carbapenems): Inhibit transpeptidases (Penicillin-Binding Proteins, PBPs).
- Vancomycin: Binds D-Ala-D-Ala terminus; blocks transglycosylation & transpeptidation.
- Protein Synthesis Inhibitors: (📌 "Buy AT 30, CCELL at 50")
- 30S Subunit: Aminoglycosides (cause mRNA misreading), Tetracyclines (block tRNA attachment).
- 50S Subunit: Macrolides, Clindamycin, Linezolid (block peptide chain translocation/initiation complex formation). Chloramphenicol (inhibits peptidyl transferase).
- Nucleic Acid Synthesis/Function Inhibitors:
- Fluoroquinolones: Inhibit DNA gyrase (topoisomerase II) & topoisomerase IV.
- Rifampin: Inhibits DNA-dependent RNA polymerase.
- Sulfonamides & Trimethoprim: Sequentially inhibit folate synthesis.
- Cell Membrane Integrity Disruptors:
- Polymyxins (e.g., Colistin): Disrupt Gram-negative bacterial outer membrane.
- Daptomycin: Depolarizes Gram-positive bacterial cell membrane.

⭐ Beta-lactams exhibit time-dependent killing (Time > MIC is critical), while Aminoglycosides show concentration-dependent killing (Peak/MIC ratio) and a post-antibiotic effect (PAE).
Drug Dynamics - PK/PD & Dosing Dance
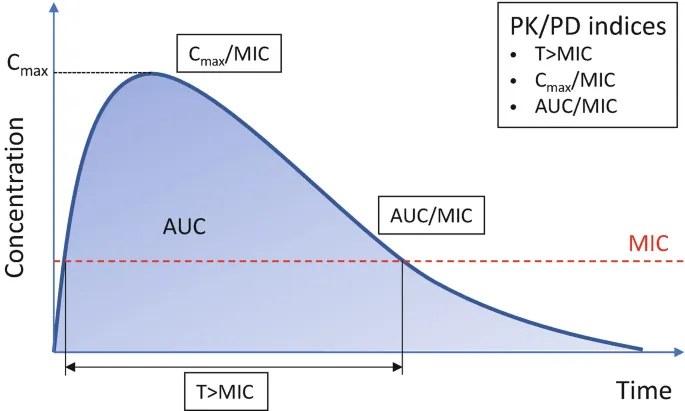
- PK/PD Indices & Dosing: Core goal is to optimize exposure to achieve bacterial kill & prevent resistance.
- Cmax/MIC (Concentration-dependent):
- Drugs: Aminoglycosides, Fluoroquinolones, Daptomycin.
- Goal: Peak conc. (Cmax) 8-10x MIC.
- Dosing: Large doses, less frequent intervals.
- T > MIC (Time-dependent):
- Drugs: Beta-lactams, Clindamycin, Linezolid.
- Goal: Drug conc. above MIC for 40-70% of dosing interval.
- Dosing: Frequent smaller doses or continuous/prolonged infusion.
- AUC/MIC (Exposure-dependent):
- Drugs: Vancomycin (target AUC/MIC > 400), Azithromycin, Fluoroquinolones, Tetracyclines.
- Goal: Optimize total drug exposure over 24h.
- Cmax/MIC (Concentration-dependent):
- Post-Antibiotic Effect (PAE): Persistent bacterial suppression after drug levels fall below MIC (e.g., Aminoglycosides, Fluoroquinolones).
- Dosing Adjustments: Crucial in renal impairment (e.g., Beta-lactams, Vancomycin) & hepatic dysfunction (e.g., Macrolides, Metronidazole).
⭐ For Vancomycin, trough concentrations (15-20 mg/L for severe infections like MRSA pneumonia/endocarditis; 10-15 mg/L for less severe infections) are often used as a practical surrogate for achieving the target AUC/MIC > 400.
Resistance Rampage - Bugs Fight Back!

-
Primary Mechanisms of Resistance:
- Altered Target Site: Modification of drug binding site. e.g., PBPs (mecA in MRSA → Methicillin resistance), Ribosomes (rRNA methylation by erm genes → Macrolide resistance).
- Enzymatic Inactivation: Bacterial enzymes destroy/modify antibiotic. e.g., β-lactamases (ESBLs, AmpC, KPC, NDM-1), Aminoglycoside-modifying enzymes.
- Decreased Permeability/Increased Efflux:
- Reduced drug entry: Porin loss (e.g., Pseudomonas vs Carbapenems).
- Active drug removal: Efflux pumps (e.g., Tetracyclines, Fluoroquinolones). 📌 MDRugs Pumped Out!
-
Acquisition & Spread:
- Intrinsic Resistance: Natural to the organism.
- Acquired Resistance:
- Mutations: Spontaneous changes in bacterial DNA.
- Horizontal Gene Transfer (HGT): Sharing resistance genes.
⭐ Carbapenemases like NDM-1 (New Delhi Metallo-β-lactamase) confer broad β-lactam resistance, including carbapenems, limiting therapeutic options significantly.
Therapy Tactics - Smart Drug Choices
- Selection Factors: Host (allergy, organ function), Drug (PK/PD, spectrum, cost), Site, Local Antibiogram.
- Combination Therapy Uses:
- Synergy (e.g., endocarditis: β-lactam + aminoglycoside).
- Polymicrobial infections.
- Prevent resistance (e.g., TB, HIV).
- Empirical for severe sepsis/neutropenic fever.
- Antimicrobial Prophylaxis:
- Surgical: Give <60 min pre-incision. Re-dose if surgery >2 drug half-lives or major blood loss.
- Medical: Specific high-risk groups (e.g., PCP in HIV, rheumatic fever recurrence).
- PK/PD Insights: Time-dependent (e.g., β-lactams: $T > MIC$) vs. Concentration-dependent (e.g., Aminoglycosides: $C_{max}/MIC$).
⭐ Surgical site infection (SSI) prophylaxis: Cefazolin is a common choice. For high MRSA risk, consider adding Vancomycin. Prophylaxis is typically discontinued within 24h post-operatively for most procedures.

High‑Yield Points - ⚡ Biggest Takeaways
- Bactericidal drugs (e.g., Beta-lactams) kill bacteria; Bacteriostatic (e.g., Macrolides) inhibit growth.
- Time-dependent killing (e.g., Beta-lactams) needs levels above MIC; Concentration-dependent (e.g., Aminoglycosides) needs high peaks.
- Post-Antibiotic Effect (PAE) allows extended dosing intervals for drugs like Aminoglycosides.
- Major resistance mechanisms: Enzymatic inactivation (β-lactamases), target modification (MRSA), efflux pumps.
- Empirical therapy guides initial choice; de-escalate based on culture and sensitivity.
- Combination therapy for synergy, broad-spectrum cover, or preventing resistance.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

