Principles of Immunization - Bodyguard Basics
- Immunity: Body's specific defense against pathogens.
- Antigen (Ag): Foreign substance (e.g., microbial protein) recognized by immune system, inducing a response.
- Antibody (Ab): Protective protein (immunoglobulin) produced by B-lymphocytes against a specific Ag.
- Adjuvant: Substance enhancing immune response to a vaccine Ag (e.g., Alum salts).
- Active Immunity: Own body produces Abs; long-lasting, memory.
- Natural: Post-infection (e.g., Measles).
- Artificial: Post-vaccination (e.g., OPV).
- Passive Immunity: Readymade Abs transferred; short-lived, no memory.
- Natural: Maternal (IgG via placenta, IgA via colostrum).
- Artificial: Immunoglobulin/Antisera admin (e.g., ATS, Rabies Ig).

⭐ Herd immunity: Indirect protection of unimmunized individuals when a critical proportion of the population is immune. For measles, this is ~95% vaccination coverage needed to prevent outbreaks.
Types of Vaccines - Potion Arsenal
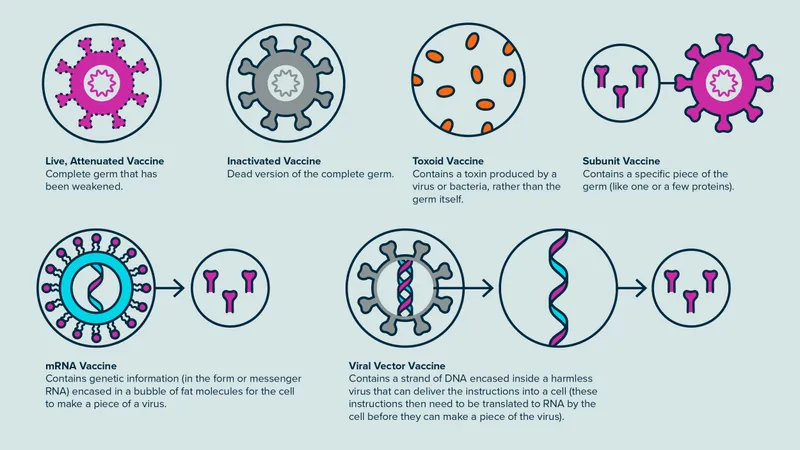
- Live-Attenuated: Weakened pathogen.
- Immunity: Strong, Cell-Mediated Immunity (CMI) & humoral, long-lasting.
- E.g., BCG, Oral Polio Vaccine (OPV), Measles, Mumps, Rubella (MMR), Varicella, Yellow Fever, Rotavirus.
- Adv: Fewer doses often sufficient.
- Disadv: Risk of reversion to virulence, contraindicated in severe immunodeficiency.
- Inactivated (Killed): Whole, killed pathogen.
- Immunity: Mainly humoral, shorter duration, requires boosters.
- E.g., Inactivated Polio Vaccine (IPV), Rabies, Influenza (injectable), Pertussis (whole-cell), Hepatitis A, Japanese Encephalitis (killed).
- Adv: Safer, no risk of reversion.
- Disadv: Weaker immune response, multiple doses needed.
- Toxoid: Inactivated bacterial toxins.
- E.g., Tetanus Toxoid (TT), Diphtheria Toxoid (DT).
- Adv: Targets toxin directly. Disadv: Boosters required.
- Subunit: Uses specific antigens (e.g., proteins, polysaccharides).
- Protein: Hepatitis B, Acellular Pertussis.
- Polysaccharide: Pneumococcal (PPSV23) - poor immunogenicity in children <2 years.
- Conjugate: Hib, Pneumococcal (PCV), Meningococcal (polysaccharide + carrier protein → T-cell dependent response).
- Viral Vector: Uses a harmless virus to deliver genetic material coding for target antigens.
- E.g., Covishield (Adenovirus vector for SARS-CoV-2).
- mRNA: Contains mRNA encoding for specific antigen(s); host cells produce the antigen.
- E.g., Pfizer-BioNTech, Moderna COVID-19 vaccines.
- Adv: Rapid development, potent immunity. Disadv: Ultra-cold chain for some.
⭐ Conjugate vaccines (e.g., Hib, PCV) convert T-independent polysaccharide antigens into T-dependent antigens by linking them to a protein carrier. This significantly enhances immunogenicity, especially in infants and young children <2 years old, and induces immunological memory.
Vaccine Admin & Cold Chain - Delivery & Chill
- Admin:
- Routes: ID (BCG), SC (Measles), IM (Penta), Oral (OPV).
- Sites: L.Upper Arm (ID), Anterolat.Thigh/Deltoid (SC/IM).
- Cold Chain: System for vaccine potency (+2°C to +8°C).
- Freezer (-15°C to -25°C): OPV, Measles, JE (lyo).
- ⚠️ Freeze-sensitive: HepB, Penta, DPT, TT, IPV, PCV, Diluents. (📌 DPT, Penta, TT, HepB).
- Equipment: WIC → DF/ILR → Cold Box → Vaccine Carrier.

- VVM: Heat-sensitive. Inner square darkens. Discard if ≥ outer circle (Stage 3/4).
- Open Vial Policy (OVP):
- Multi-dose (OPV, Penta, TT, IPV, PCV, Rota-liq): Use up to 28 days (VVM OK, cold chain).
- Reconstituted (BCG, Measles, JE): Discard: BCG 4 hrs; Measles/JE 6 hrs / end session.
⭐ Reconstituted BCG: discard after 4 hrs (prevent TSS).
AEFI & Herd Immunity - Side Quests & Superpowers
- AEFI (Adverse Events Following Immunization): Any untoward medical occurrence post-immunization; not necessarily causal.
- Classification: Vaccine product-related, Vaccine quality defect-related, Immunization error-related, Immunization anxiety-related, Coincidental event.
- Reporting is vital for pharmacovigilance.
- Herd Immunity: Indirect protection of unvaccinated individuals when a critical proportion of the population is immune.
- Threshold (HIT) for interruption of transmission: $ (1 - 1/R_0) \times 100% $.
- $R_0$ = Basic Reproduction Number. Higher $R_0$ requires higher HIT.
⭐ For measles ($R_0 \approx \mathbf{12-18}$), HIT is 92-95%.
- Vaccine Efficacy vs. Effectiveness:
- Efficacy: Protective effect under ideal trial conditions. Formula: $ (ARU - ARV) / ARU \times 100% $ (ARU=Attack Rate Unvaccinated, ARV=Attack Rate Vaccinated).
- Effectiveness: Real-world vaccine performance.
High‑Yield Points - ⚡ Biggest Takeaways
- Herd immunity threshold is crucial (e.g., Measles >94%).
- Cold chain: +2°C to +8°C; OPV, Measles need freezing ( -15°C to -25°C).
- VVM: discard if inner square is same as/darker than outer circle.
- Live vaccines (BCG, OPV, Measles): long immunity; contraindicated in pregnancy, immunosuppression.
- Killed vaccines (IPV, TT): safer, multiple doses often needed.
- Interval between two live vaccines: 4 weeks if not simultaneous.
- Anaphylaxis post-vaccination: treat with adrenaline.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

