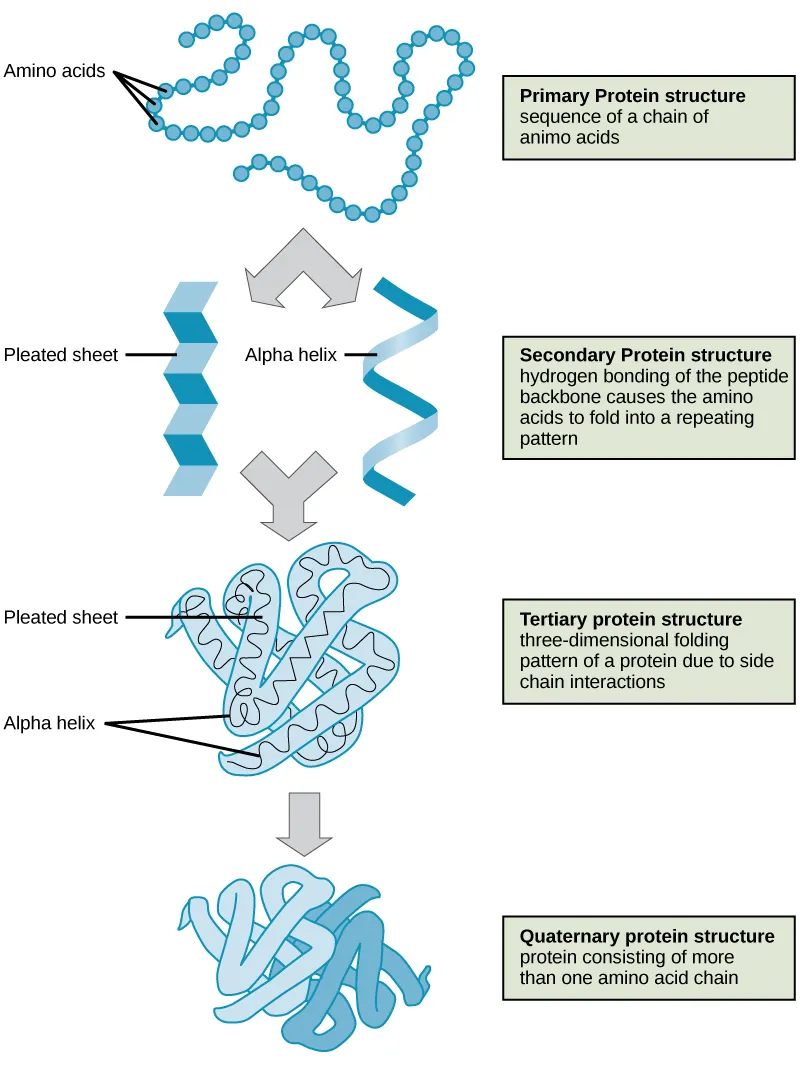
Protein Architecture - Building Blocks Basics
- Primary (1°): Linear amino acid sequence; peptide bonds. Dictates higher structures.
- Secondary (2°): Local folding.
- α-helix: Intra-chain H-bonds.
- β-pleated sheet: Inter/intra-chain H-bonds.
- Ramachandran plot: Defines allowed $ \phi $ and $ \psi $ angles.
- Tertiary (3°): Single polypeptide 3D shape. Stabilized by disulfide bonds, hydrophobic interactions, H-bonds, ionic bonds. Forms functional domains.
- Quaternary (4°): Assembly of >1 polypeptide subunits (e.g., Hemoglobin $ \alpha_2\beta_2 $).
⭐ Proline, with its rigid cyclic structure, acts as a 'helix breaker' in α-helices and is often found in β-turns.
Folding & Friends - Shaping Up Proteins
- Anfinsen's dogma: Primary structure (amino acid sequence) dictates 3D conformation.
- Driving forces for folding:
- Hydrophobic effect (major): Nonpolar residues buried.
- Hydrogen bonds, ionic interactions, van der Waals forces.
- Molecular chaperones (e.g., Hsp60, Hsp70): Assist correct folding, prevent aggregation; some require ATP.
- Denaturation: Loss of 2°, 3°, 4° structure & function. Causes: heat, extreme pH, organic solvents, urea.
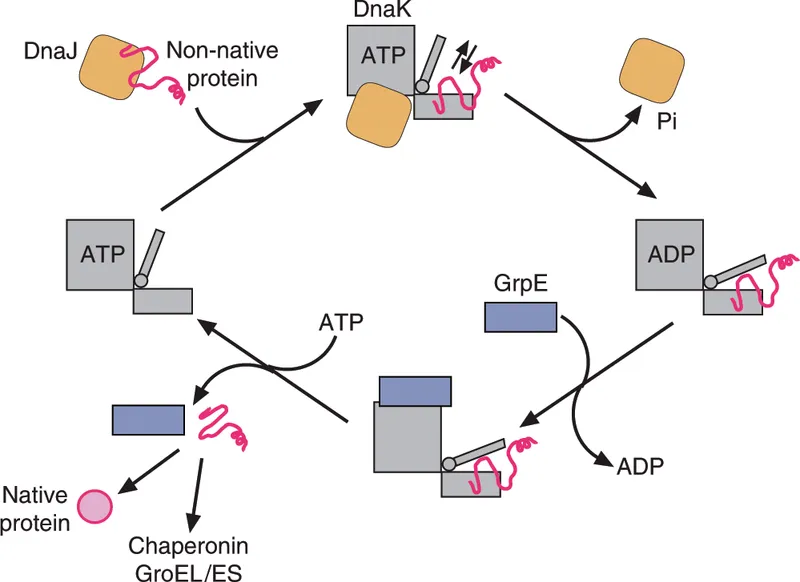
⭐ Christian Anfinsen's ribonuclease A experiment: Primary structure dictates tertiary structure.
Functional Forms - Proteins At Work
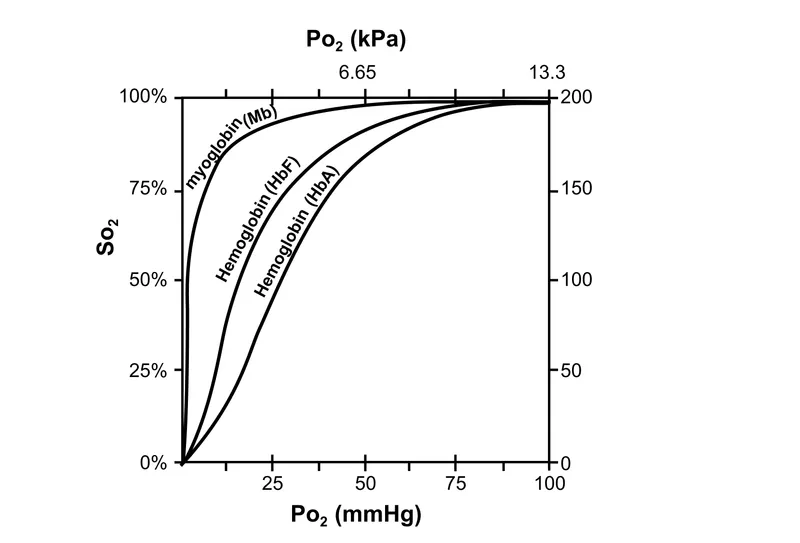
- Hemoglobin (Hb) & Myoglobin (Mb):
- Mb: Monomer, O2 storage, hyperbolic O2 binding curve.
- Hb: Tetramer (adult HbA is $\alpha_2\beta_2$), O2 transport, sigmoidal O2 binding curve (cooperative binding).
- Allosteric Regulation of Hb:
- O2: Positive homotropic effector.
- Heterotropic effectors (↓O2 affinity, stabilize T-state):
- H+ (Bohr effect: ↓pH → ↓O2 affinity).
- CO2 (forms carbaminohemoglobin, contributes to Bohr).
- 2,3-BPG (stabilizes T-state).
- 📌 Mnemonic: 'CADET, face Right' for factors shifting O2-Hb curve right (↑CO2, ↑Acid/↓pH, ↑2,3-BPG, ↑Exercise, ↑Temperature).
-
⭐ Fetal hemoglobin (HbF, with alpha-2 gamma-2 chains) has higher O2 affinity than adult hemoglobin (HbA, with alpha-2 beta-2 chains) as it binds 2,3-BPG less avidly.
- Enzymes:
- Active site (binding & catalytic sites) dictates substrate specificity (Lock-and-key vs. Induced fit) and catalytic efficiency by lowering activation energy ($E_a$).
- Collagen:
- Unique triple helix (Gly-X-Y repeats; X=Pro, Y=Hyp/Hyl).
- Post-translational modifications (hydroxylation requiring Vit C, glycosylation) & cross-linking are vital for tensile strength.
- Defects: Scurvy (Vit C deficiency), Osteogenesis Imperfecta.
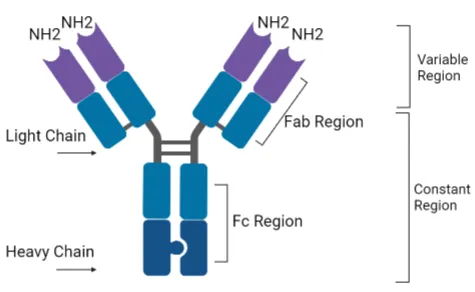
- Immunoglobulins (Antibodies):
- Modular: Variable regions (Fab, antigen binding) & Constant regions (Fc, effector functions).
- Composed of heavy & light chains linked by disulfide bonds.
Misfits & Maladies - When Proteins Go Rogue
- Protein misfolding: Can cause loss of normal function or gain of toxic function, often leading to aggregation (e.g., amyloid fibrils) and deposition.
- Key Proteinopathies & Associated Proteins:
- Alzheimer's Disease: Extracellular plaques of Amyloid-β (Aβ) (from APP); intracellular neurofibrillary tangles of hyperphosphorylated Tau protein.
- Parkinson's Disease: Intraneuronal aggregates of α-synuclein (Lewy bodies).
- Prion Diseases (e.g., CJD, Kuru): Conformational change of PrPC to pathogenic PrPSc; infectious, induces misfolding.
- Cellular defense: Chaperones attempt refolding; Ubiquitin-Proteasome System degrades misfolded proteins.
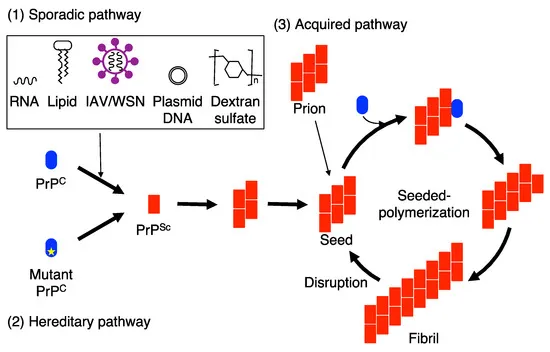
⭐ Prion diseases are unique as they can be infectious (e.g., Kuru), sporadic (e.g., sporadic CJD), or inherited (e.g., familial CJD), all involving PrPC → PrPSc conversion.
High‑Yield Points - ⚡ Biggest Takeaways
- Primary structure (amino acid sequence) dictates 3D conformation and function.
- Secondary structures (e.g., α-helix, β-sheet) are stabilized by backbone hydrogen bonds.
- Tertiary structure forms the active site; stabilized by disulfide bonds, hydrophobic interactions, and ionic bonds.
- Quaternary structure (e.g., Hemoglobin) involves multiple interacting polypeptide subunits.
- Protein misfolding leads to diseases like Alzheimer's disease, Parkinson's disease, and prion diseases.
- Chaperones (e.g., HSPs) assist in correct protein folding and prevent aggregation.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

