Cholesterol: Basics - Essential Waxy Molecule
- Waxy, fat-like substance (sterol); essential structural component of animal cell membranes.
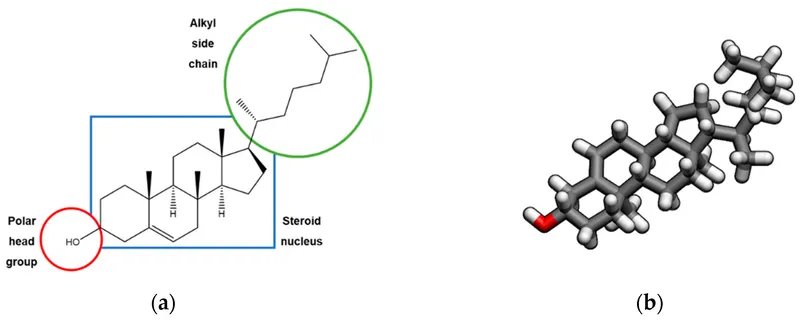
- Structure: 27-carbon molecule with a tetracyclic steroid nucleus (cyclopentanoperhydrophenanthrene ring).
- Amphipathic: Hydrophilic hydroxyl (-OH) group at C-3, hydrophobic body.
- Sources:
- Dietary (exogenous): Animal products (e.g., meat, eggs, dairy).
- De novo synthesis (endogenous): Primarily in liver; also intestine, adrenal cortex, gonads.
- Functions:
- Maintains cell membrane fluidity & integrity.
- Precursor for:
- Bile acids (aid fat digestion).
- Steroid hormones (e.g., cortisol, aldosterone, sex hormones).
- Vitamin D.
⭐ Most plasma cholesterol is esterified with a fatty acid, forming cholesteryl esters, for transport and storage; this is catalyzed by ACAT (intracellular) and LCAT (plasma).
Cholesterol: Synthesis - From Acetate to Sterol
- Location: Cytosol (early reactions), Endoplasmic Reticulum (ER) for HMG-CoA Reductase & later steps.
- Substrate: Acetyl-CoA (provides all carbon atoms).
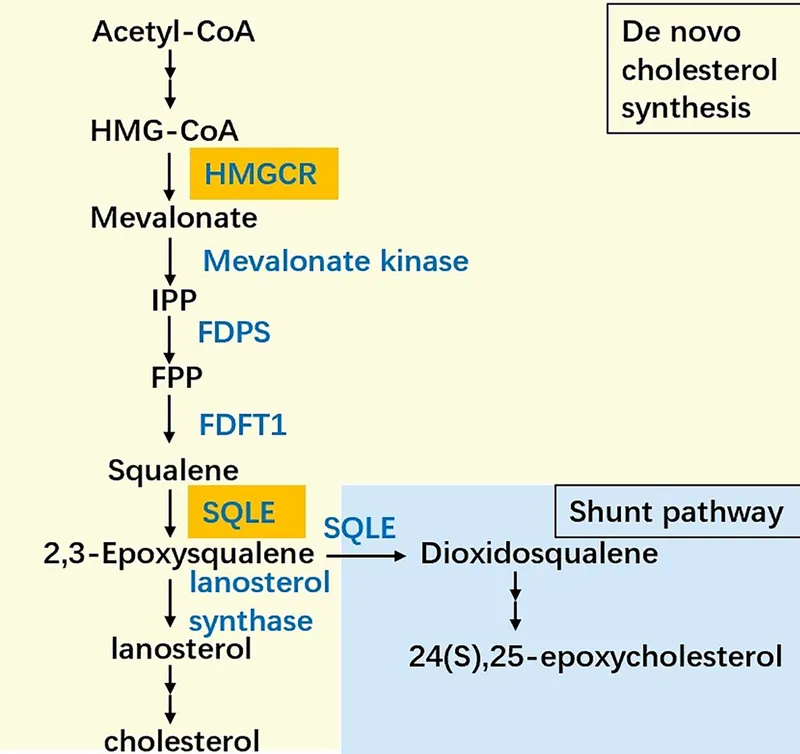
- Rate-Limiting Step (RLS): HMG-CoA conversion to Mevalonate by HMG-CoA Reductase (ER membrane, uses 2 NADPH).
⭐ HMG-CoA Reductase: Key regulatory point; target of statin drugs; inhibited by cholesterol, mevalonate; activated by insulin.
- Key Conversions:
- Mevalonate (6C) $\rightarrow$ Isopentenyl Pyrophosphate (IPP - 5C) (requires ATP, involves decarboxylation).
- IPP $\rightleftharpoons$ Dimethylallyl Pyrophosphate (DMAPP - 5C) (isomerization).
- IPP + DMAPP $\rightarrow$ Geranyl-PP (10C) $\rightarrow$ Farnesyl-PP (FPP - 15C).
- 2 FPP molecules condense to Squalene (30C) via Squalene Synthase (ER, requires NADPH).
- Squalene is converted to Lanosterol (30C, the first sterol) via Squalene Epoxidase (ER, requires O2, NADPH) and Lanosterol Cyclase.
- Lanosterol $\rightarrow$ Cholesterol (27C) through multiple (approx. 19) enzymatic steps in the ER (e.g., demethylations, double bond shifts).
Cholesterol: Regulation - The Control System
- Key Enzyme: HMG-CoA Reductase (HMGCR); ER-bound, rate-limiting.
- HMGCR Regulation:
- Feedback: ↑Cholesterol inhibits HMGCR activity & ↓SREBP-2 mediated transcription.
- Hormonal:
- Insulin, Thyroxine: ↑HMGCR (dephosphorylation, ↑transcription).
- Glucagon, Cortisol: ↓HMGCR (phosphorylation, ↓transcription).
- Transcriptional (SREBP-2):
- Low cholesterol → SREBP-2 active → ↑HMGCR gene expression.
- High cholesterol → SREBP-2 inactive → ↓HMGCR gene expression.
- Covalent: Dephosphorylated (Active) ↔ Phosphorylated (Inactive by AMPK).
- Degradation: ↑Sterols → ↑HMGCR proteolysis.
- Drugs: Statins (e.g., Atorvastatin) competitively inhibit HMGCR. 📌 "Statins Stop HMGCR".
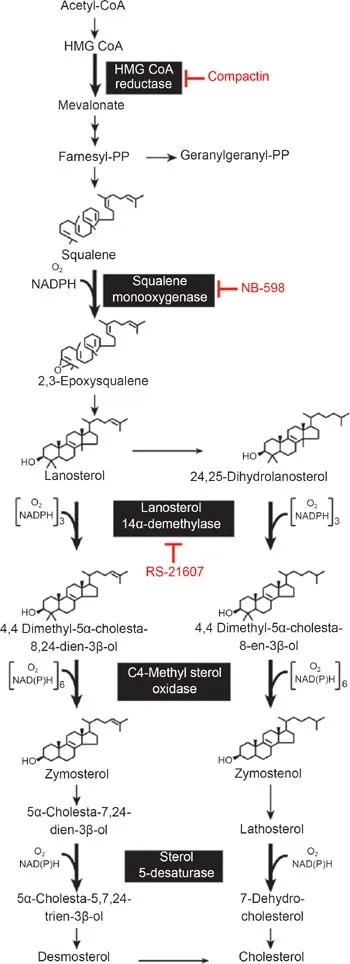
⭐ Statins competitively inhibit HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, effectively lowering plasma cholesterol.
Cholesterol: Transport & Fate - Delivery and Destiny
- Transport & Delivery: 📌 LDL: "Leaves Da Cholesterol"; HDL: "Helps Da Liver".
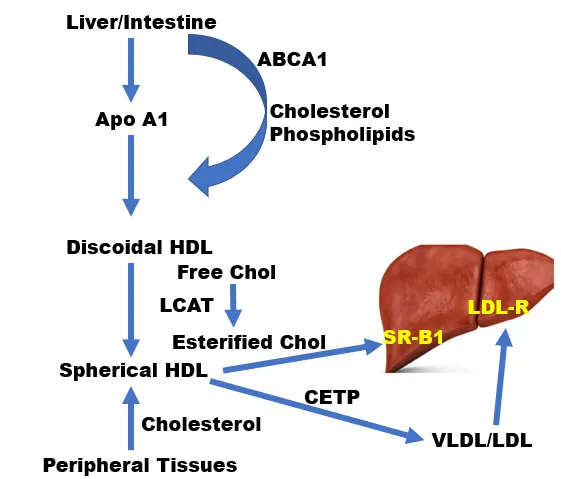
- LDL (Low-Density Lipoprotein): "Bad cholesterol"; delivers hepatic cholesterol to peripheral tissues. ApoB-100 mediated.
- HDL (High-Density Lipoprotein): "Good cholesterol"; Reverse Cholesterol Transport (RCT) to liver. Key: LCAT (Lecithin-Cholesterol Acyltransferase), ApoA-I (LCAT activator).
- CETP (Cholesteryl Ester Transfer Protein): Exchanges HDL's cholesteryl esters (CE) for VLDL/IDL's triglycerides (TG).
- Cellular Uptake & Storage:
- LDL receptor (LDL-R) mediated endocytosis.
- Stored as cholesteryl esters by ACAT (Acyl-CoA Cholesterol Acyltransferase) enzyme.
- Metabolic Fates (Utilization):
- Bile Acid Synthesis: Major elimination pathway in liver. Rate-limiting enzyme: Cholesterol 7α-hydroxylase (CYP7A1).
- Steroid Hormone Synthesis: Precursor for all steroid hormones (e.g., cortisol, aldosterone, sex hormones).
- Vitamin D Synthesis.
- Cell Membrane Component: Essential for structure and fluidity.
- Excretion:
- Primarily as bile acids and neutral sterols (unmodified cholesterol) in feces.
⭐ Familial Hypercholesterolemia (FH) is an autosomal dominant disorder, most commonly due to LDL receptor defects, leading to markedly ↑LDL-C levels and premature atherosclerosis.
High‑Yield Points - ⚡ Biggest Takeaways
- HMG-CoA reductase is the rate-limiting enzyme in cholesterol biosynthesis, targeted by statin drugs.
- All carbons of cholesterol are derived from Acetyl-CoA; synthesis occurs mainly in the liver.
- LDL delivers cholesterol to peripheral tissues; HDL mediates reverse cholesterol transport.
- Familial Hypercholesterolemia results from LDL receptor defects, causing ↑ plasma LDL.
- Cholesterol is the precursor for bile acids, steroid hormones, and Vitamin D.
- 7α-hydroxylase is the rate-limiting enzyme for bile acid synthesis from cholesterol.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

