Cytochrome P450 System - Detox Dynamo
- Family of heme-containing monooxygenases; key players in Phase I detoxification.
- Location: Primarily smooth endoplasmic reticulum (SER) of hepatocytes; also extrahepatic (gut, lungs, kidney).
- Core Reaction: $RH + O_2 + NADPH + H^+ \rightarrow ROH + H_2O + NADP^+$
- Adds or unmasks polar groups (e.g., -OH, -NH2, -SH).
- Function: Metabolizes diverse xenobiotics (drugs, carcinogens) & endogenous molecules (steroids, prostaglandins).
- Named for 450 nm light absorption peak when CO-bound.

⭐ CYP3A4 is the most abundant CYP isoform in the human liver, responsible for metabolizing approximately 50% of clinically used drugs.
Cytochrome P450 System - Metabolic Makeover
- Location & Structure: Predominantly in liver smooth endoplasmic reticulum; heme-containing monooxygenases.
- Function: Metabolize diverse substrates: xenobiotics (drugs, carcinogens, pollutants) & endogenous compounds (steroids, fatty acids, prostaglandins).
- Nomenclature: CYP + Arabic numeral (family) + capital letter (subfamily) + Arabic numeral (isoform). E.g., CYP3A4 is a major drug-metabolizing enzyme.
Principal Reactions (Phase I):
- Oxidation (most common):
- Hydroxylation: $R-H + O_2 + NADPH + H^+ \rightarrow R-OH + H_2O + NADP^+$
- N-dealkylation, O-dealkylation, S-dealkylation
- Epoxidation, N-oxidation, S-oxidation
- Reduction (e.g., azo, nitro reduction)
- Hydrolysis (less frequent for P450s)
Catalytic Cycle:
⭐ The CYP450 catalytic cycle critically depends on NADPH-cytochrome P450 reductase for the transfer of electrons from NADPH to the P450 enzyme.
Cytochrome P450 System - Induction & Inhibition Show
- Enzyme Induction:
- Process: ↑ synthesis of CYP450 enzymes.
- Leads to: ↑ drug metabolism rate.
- Clinical effect: ↓ drug efficacy or ↑ production of toxic metabolites.
- Enzyme Inhibition:
- Process: ↓ activity of CYP450 enzymes (competitive or non-competitive).
- Leads to: ↓ drug metabolism rate.
- Clinical effect: ↑ plasma drug concentration → ↑ risk of drug toxicity.
| Feature | Inducers | Inhibitors |
|---|---|---|
| Mnemonic | 📌 CRAP GPS | 📌 SICKFACES.COM Group |
| Examples | Carbamazepine, Rifampicin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, St. John’s Wort | Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole, Grapefruit juice |
Cytochrome P450 System - Genes & Drugs
- Key Gene Families: CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4/5.
- Genetic Polymorphisms:
- Cause inter-individual variations in drug metabolism.
- Phenotypes: Poor Metabolizers (PMs), Intermediate (IMs), Extensive (EMs), Ultra-rapid Metabolizers (UMs).
- PMs: ↑ risk of drug toxicity; UMs: may experience therapeutic failure with standard doses.
- Common Inducers (↓ drug efficacy):
- Rifampicin, Carbamazepine, Phenytoin, Phenobarbital, St. John’s Wort, Chronic alcohol use, Smoking.
- Common Inhibitors (↑ drug toxicity):
- Azole antifungals (e.g., Ketoconazole), Macrolides (e.g., Erythromycin), Grapefruit juice, Cimetidine, SSRIs (e.g., Fluoxetine), Protease inhibitors.
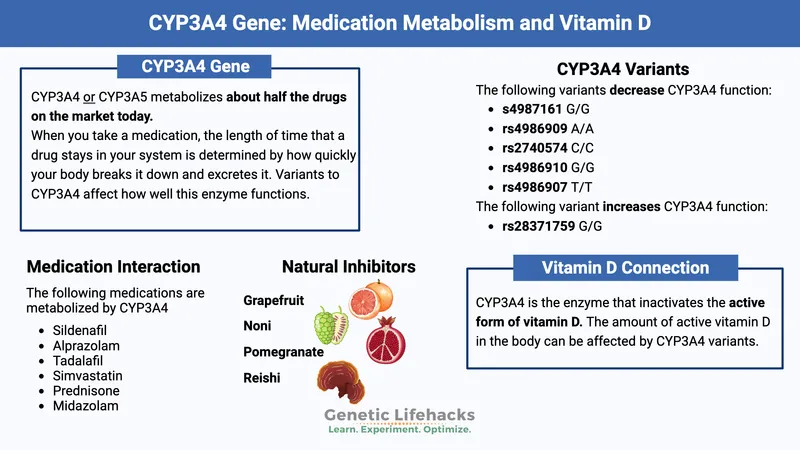
- Azole antifungals (e.g., Ketoconazole), Macrolides (e.g., Erythromycin), Grapefruit juice, Cimetidine, SSRIs (e.g., Fluoxetine), Protease inhibitors.
⭐ CYP2D6 polymorphism significantly affects the metabolism of codeine to morphine, impacting analgesic efficacy. Poor metabolizers may experience little to no pain relief, while ultra-rapid metabolizers may have increased adverse effects due to rapid morphine conversion.
High‑Yield Points - ⚡ Biggest Takeaways
- CYP450 enzymes: Located in liver smooth ER; are heme-containing monooxygenases.
- Primary role: Catalyze Phase I reactions (oxidation, reduction, hydrolysis) for drug metabolism.
- CYP3A4: Most abundant isoform, metabolizes ~50% of clinically used drugs.
- Inducers (e.g., Rifampicin, Phenobarbital) accelerate metabolism, potentially ↓ drug efficacy.
- Inhibitors (e.g., Ketoconazole, Grapefruit juice) slow metabolism, potentially ↑ drug toxicity.
- Genetic polymorphisms in CYP genes cause significant inter-individual variations in drug response.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

