Overview & Homeostasis - The Balancing Act
Blood glucose homeostasis: maintaining stable blood glucose levels. Essential for cellular energy.
- Normal Values:
- Fasting Plasma Glucose (FPG): 70-100 mg/dL
- Post-prandial (2h PPG): <140 mg/dL
- Glycated Hemoglobin (HbA1c): <5.7%
- Major Players:
- Organs: Pancreas (α & β cells), Liver, Skeletal Muscle, Adipose tissue.
- Hormones:
- Insulin (↓ glucose): Anabolic.
- Glucagon (↑ glucose): Catabolic.
- Others (↑): Adrenaline, Cortisol, Growth Hormone.
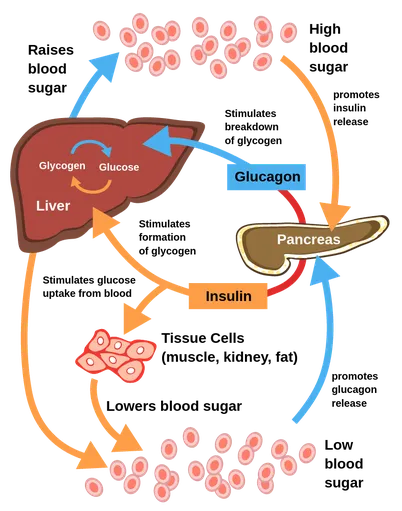
⭐ The liver is the primary organ for endogenous glucose production (gluconeogenesis & glycogenolysis).
Insulin Action - The Anabolic Architect
Pancreatic β-cell hormone; key for glucose homeostasis & anabolic processes.
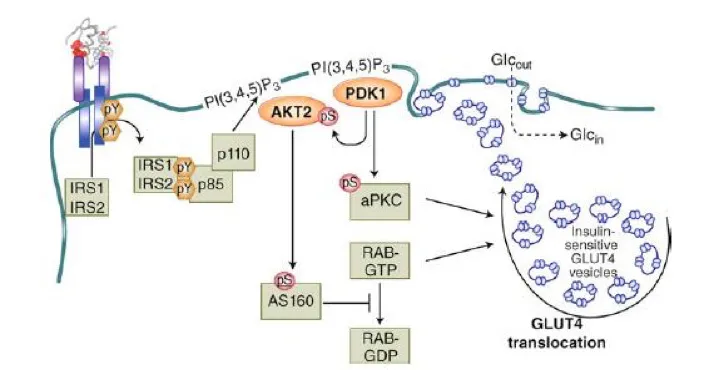
- Receptor & Pathway: Binds cell surface tyrosine kinase receptors → IRS phosphorylation → PI3K activation → Akt/PKB pathway.
- Glucose Uptake: ↑ GLUT4 translocation to cell membrane (muscle, adipose tissue) → ↑ cellular glucose entry.
- Metabolic Effects (Anabolic):
- Carbohydrates: ↑ Glycogenesis, ↑ Glycolysis (liver, muscle). ↓ Gluconeogenesis, ↓ Glycogenolysis.
- Lipids: ↑ Lipogenesis, ↑ Triglyceride storage. ↓ Lipolysis (inhibits Hormone Sensitive Lipase - HSL).
- Proteins: ↑ Amino acid uptake, ↑ Protein synthesis. ↓ Proteolysis.
- Ions: ↑ K+ uptake into cells. 📌 Mnemonic: Insulin drives K+ "IN"to cells.
⭐ Insulin is the most potent anabolic hormone; its deficiency (absolute or relative) leads to Diabetes Mellitus, characterized by hyperglycemia and catabolic states (e.g., DKA).
Glucagon & Co. - The Glucose Raisers
- Glucagon: Pancreatic α-cells. Major counter-regulatory hormone.
- Stimuli: Hypoglycemia (glucose < 70 mg/dL), amino acids.
- Actions (Liver): ↑Glycogenolysis (rapid), ↑Gluconeogenesis (sustained). ↑Lipolysis.
- Epinephrine: Adrenal medulla. Rapid glucose elevation.
- Stimuli: Hypoglycemia, stress.
- Actions: ↑Glycogenolysis (liver, muscle), ↑Gluconeogenesis, ↑Lipolysis.
- Cortisol: Adrenal cortex. Permissive, slow, prolonged.
- Stimuli: Stress (ACTH).
- Actions: ↑Gluconeogenesis (enzyme induction), ↓peripheral glucose use.
- Growth Hormone (GH): Anterior pituitary. Anti-insulin.
- Actions: ↓Peripheral glucose uptake, ↑Lipolysis, ↑Hepatic glucose output.
- Thyroid Hormones (T3/T4): ↑Gut glucose absorption, potentiate epinephrine.
- 📌 Mnemonic (Glucose Raisers): "GET C G" (Glucagon, Epinephrine, Thyroid, Cortisol, Growth Hormone).
⭐ Glucagonoma (α-cell tumor) presents with "4D" syndrome: Dermatitis (necrolytic migratory erythema), Diabetes, DVT, Depression.

GLUTs & Metabolic States - Gates & Shifts
Cellular glucose entry and metabolic adaptation depend on GLUT transporters and hormonal signals, especially insulin.
-
Key Glucose Transporters (GLUTs):
- GLUT1: Brain, RBCs (Basal, Insulin-Independent). $K_m \approx$ 1-2 mM.
- GLUT2: Liver (glucose uptake/release), Pancreas β-cells (glucose sensing), intestine, kidney. High $K_m \approx$ 15-20 mM. Insulin-Independent.
- GLUT3: Neurons, placenta. High affinity ($K_m <$ 1 mM). Insulin-Independent.
- GLUT4: Muscle, Adipose tissue (Insulin-Dependent). $K_m \approx$ 5 mM. 📌 4 = Insulin's Door.
-
Metabolic Shifts & Glucose Gates:
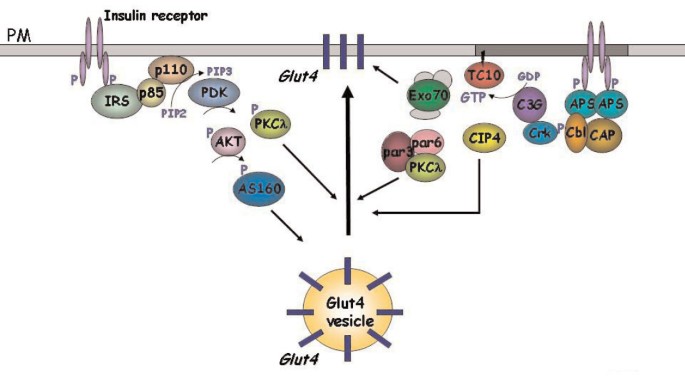
⭐ In the fed state, insulin promotes GLUT4 translocation from intracellular vesicles to the plasma membrane in muscle and adipose cells, increasing glucose uptake by 10-20 fold.
High‑Yield Points - ⚡ Biggest Takeaways
- Insulin is the primary hypoglycemic hormone; Glucagon is the main hyperglycemic hormone.
- GLUT4 (muscle, adipose tissue) is insulin-dependent for glucose uptake.
- GLUT2 (liver, pancreatic β-cells, kidney) is insulin-independent and bidirectional.
- Brain primarily uses glucose via insulin-independent GLUT1 and GLUT3.
- Normal fasting blood glucose: 70-100 mg/dL. HbA1c reflects long-term control (target < 7%).
- Von Gierke's disease (GSD Type Ia) causes severe fasting hypoglycemia due to glucose-6-phosphatase deficiency.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

