Skull Base Surgery

On this page
🏗️ Skull Base Surgery: The Cranial Foundation Mastery
The skull base forms a treacherous surgical frontier where millimeters separate success from catastrophe, demanding mastery of complex anatomy, diverse approaches, and split-second decision-making. You'll learn to navigate this three-dimensional labyrinth by understanding foundational architecture, selecting optimal surgical corridors for each pathology, recognizing tumor patterns through imaging, executing precise techniques that preserve critical neurovascular structures, and managing complications before they cascade. This lesson transforms the skull base from an intimidating no-go zone into conquerable territory through systematic frameworks that build your confidence and clinical judgment.
📌 Remember: ACE-PM for skull base regions - Anterior (frontal), Central (sphenoid), Ethmoid, Posterior (occipital), Middle (temporal) - Each zone demands specific surgical expertise with distinct complication profiles
The anterior skull base extends from the frontal bone to the sphenoid ridge, encompassing the cribriform plate and orbital roofs. This region houses the olfactory apparatus and provides access to frontal lobe pathology. Surgical violations here risk CSF leaks in 15-25% of cases and anosmia in 30-40% of extensive resections.
- Anterior Cranial Fossa Boundaries
- Superior: Frontal bone inner table
- Posterior: Sphenoid ridge and planum sphenoidale
- Lateral: Orbital roofs and frontal bone
- Critical thickness: Orbital roof 1-3mm average
- Danger zone: Cribriform plate 0.5-1mm thickness
- CSF leak risk: 20-30% with extensive cribriform work
The middle skull base represents the most complex surgical territory, housing the sella turcica, cavernous sinuses, and temporal bone structures. This region contains 60% of skull base pathology and requires multimodal approaches for optimal outcomes.
| Skull Base Region | Key Structures | Common Pathology | Surgical Risk | CSF Leak Rate | Functional Preservation |
|---|---|---|---|---|---|
| Anterior | Cribriform, orbital roof | Meningiomas, esthesioneurobloma | 15-20% morbidity | 20-25% | 70-80% olfaction |
| Central | Sella, planum sphenoidale | Pituitary adenomas, craniopharyngioma | 8-12% morbidity | 5-10% | 85-95% vision |
| Middle | Cavernous sinus, petrous apex | Trigeminal schwannomas, chordomas | 25-35% morbidity | 10-15% | 60-75% cranial nerves |
| Posterior | Clivus, foramen magnum | Chordomas, meningiomas | 20-30% morbidity | 15-20% | 70-85% brainstem function |
| Lateral | Petrous bone, jugular foramen | Acoustic neuromas, paragangliomas | 15-25% morbidity | 8-12% | 65-80% hearing |
The posterior skull base encompasses the clivus, foramen magnum, and cerebellopontine angle. This region presents unique challenges due to brainstem proximity and vertebrobasilar circulation. Surgical approaches here demand microsurgical precision with zero tolerance for vascular injury.
💡 Master This: Skull base surgery success depends on anatomical layer recognition - Each 1mm depth change alters risk profiles dramatically. Dural preservation reduces CSF leak rates from 25-30% to <5%, while bone reconstruction prevents pneumocephalus in >95% of cases
Modern skull base surgery integrates endoscopic visualization, image-guided navigation, and intraoperative monitoring to achieve maximal resection with minimal morbidity. Endoscopic approaches now account for 70-80% of anterior skull base procedures, offering superior visualization and reduced brain retraction compared to traditional craniotomies.
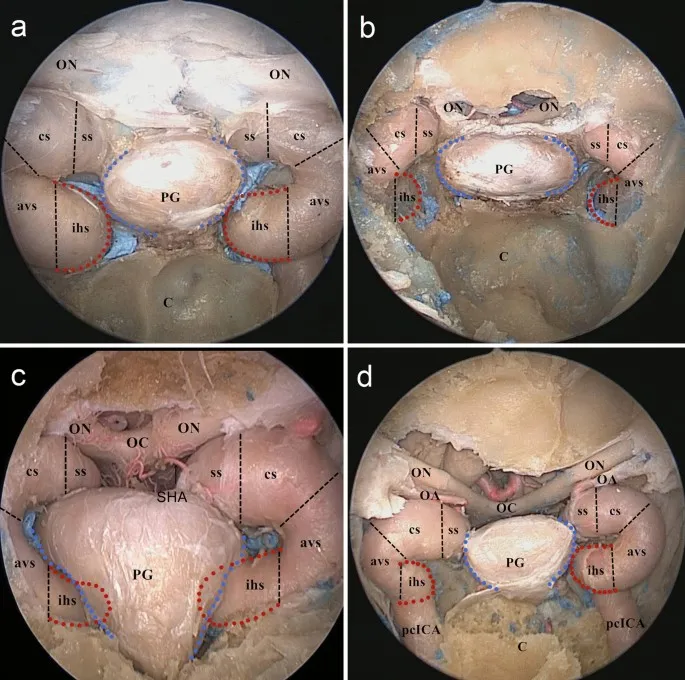
Understanding skull base surgery requires mastering the anatomical blueprint that guides every surgical decision, setting the foundation for exploring specific approaches and their clinical applications.
🏗️ Skull Base Surgery: The Cranial Foundation Mastery
🎯 Surgical Approach Arsenal: Navigation Strategies
📌 Remember: TALES for approach selection - Tumor location, Anatomical variants, Lesion size, Exposure needs, Surgeon experience - Optimal outcomes require matching approach to lesion demands with >90% accuracy
Endoscopic transsphenoidal approaches dominate sellar and parasellar pathology, providing direct midline access with minimal brain manipulation. This technique achieves gross total resection in 85-95% of pituitary adenomas while maintaining CSF leak rates below 5% in experienced hands.
- Endoscopic Advantages
- Superior visualization: 4K resolution with angled optics
- Reduced morbidity: No brain retraction required
- Faster recovery: 2-3 day average hospital stay
- Learning curve: 50-75 cases for proficiency
- Complication nadir: >100 cases experience
- GTR rates: 90-95% for microadenomas, 75-85% for macroadenomas
Transcranial approaches remain gold standard for lateral skull base pathology and complex anterior skull base lesions. These approaches provide wide surgical fields and multiple angle access but require brain retraction and longer operative times.
| Approach Type | Primary Indications | Operative Time | GTR Rate | Major Morbidity | Hospital Stay |
|---|---|---|---|---|---|
| Endoscopic Transsphenoidal | Pituitary adenomas, craniopharyngiomas | 2-4 hours | 85-95% | 3-8% | 2-3 days |
| Transcranial Anterior | Large meningiomas, esthesioneuroblastoma | 4-8 hours | 75-90% | 15-25% | 5-7 days |
| Lateral Skull Base | Acoustic neuromas, jugular paragangliomas | 6-12 hours | 80-95% | 20-35% | 7-10 days |
| Combined Approaches | Extensive chordomas, complex meningiomas | 8-16 hours | 70-85% | 25-40% | 10-14 days |
| Endoscopic Endonasal | Clival lesions, anterior cranial base | 3-6 hours | 80-90% | 10-20% | 4-6 days |
Lateral skull base approaches encompass multiple surgical corridors including retrosigmoid, translabyrinthine, and middle fossa routes. Approach selection depends on hearing preservation goals, tumor size, and anatomical considerations.
- Lateral Approach Selection Criteria
- Retrosigmoid: Hearing preservation priority, <3cm tumors
- Translabyrinthine: No useful hearing, >3cm tumors
- Middle fossa: Small tumors (<1.5cm), young patients
- Hearing preservation: 60-80% retrosigmoid, 0% translabyrinthine, 70-90% middle fossa
- Facial nerve preservation: >95% all approaches with monitoring
- GTR achievement: 90-95% translabyrinthine, 85-90% retrosigmoid
💡 Master This: Approach complexity correlates directly with surgeon experience requirements - Endoscopic approaches require >100 cases for optimal outcomes, while lateral skull base demands >50 cases and fellowship training for acceptable morbidity rates
Combined approaches address extensive pathology crossing multiple anatomical regions. These procedures require staged operations or team approaches with neurosurgical and otolaryngological expertise. Complication rates increase 2-3 fold but enable complete resection of otherwise inoperable lesions.
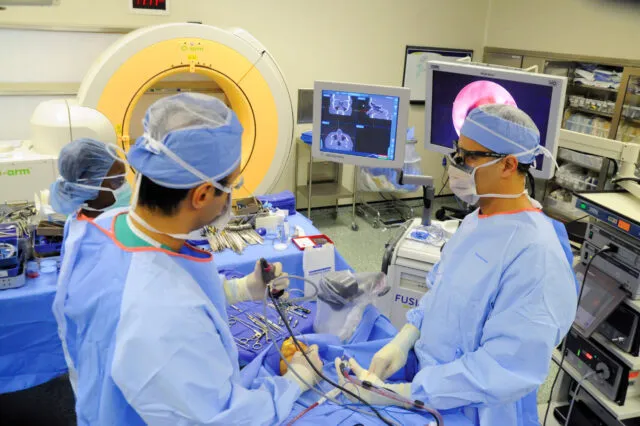
Image-guided navigation enhances surgical precision across all approaches, reducing critical structure injury by 30-50%. Real-time imaging with intraoperative CT or MRI enables immediate verification of resection completeness and complication detection.
Understanding approach selection principles enables optimal surgical planning, connecting anatomical access to specific pathology management and reconstruction requirements.
🎯 Surgical Approach Arsenal: Navigation Strategies
🧠 Pathology Spectrum: Tumor Territory Mastery
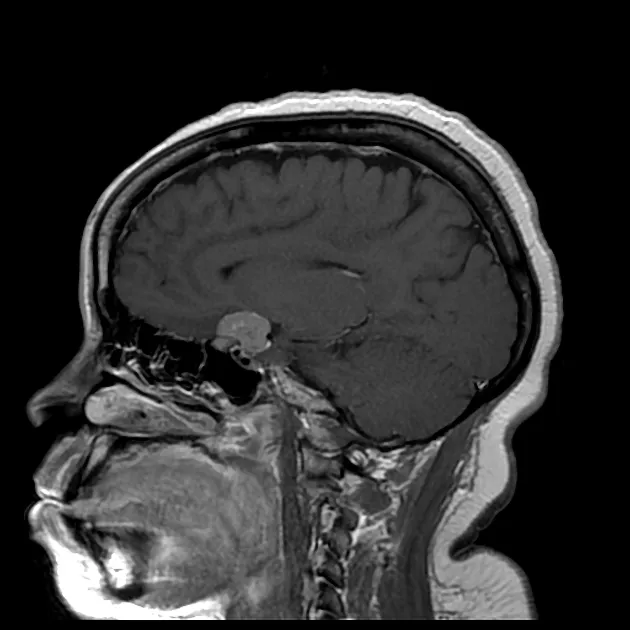
Skull base pathology encompasses diverse tumor types with distinct biological behaviors and management requirements. Tumor location, growth patterns, and invasion characteristics determine surgical approach, resection goals, and adjuvant therapy needs. Master clinicians recognize pathology-specific patterns enabling personalized treatment strategies.
📌 Remember: MAGIC for skull base tumors - Meningiomas (40%), Adenomas (25%), Gliomas (10%), Infections/inflammatory (10%), Chordomas/chondrosarcomas (15%) - Location predicts pathology with >80% accuracy
Pituitary adenomas represent 25-30% of skull base pathology, with microadenomas (<1cm) comprising 60-70% of cases. Functional adenomas present with hormonal syndromes while non-functional tumors cause mass effect symptoms. Endoscopic resection achieves biochemical cure in 80-95% of microadenomas and 60-80% of macroadenomas.
- Pituitary Adenoma Classification
- Microadenomas: <1cm, 90-95% cure rate
- Macroadenomas: >1cm, 70-85% cure rate
- Giant adenomas: >4cm, 50-70% cure rate
- Prolactinomas: Medical therapy first-line, >90% response
- Growth hormone: Biochemical cure 70-85% surgery
- Cushing's disease: Remission 80-95% microadenomas
Meningiomas account for 35-40% of skull base tumors, with anterior skull base and sphenoid wing locations predominating. WHO Grade I tumors comprise 80-85% of cases with excellent prognosis following gross total resection. Simpson Grade I-II resection achieves <5% recurrence rates at 10 years.
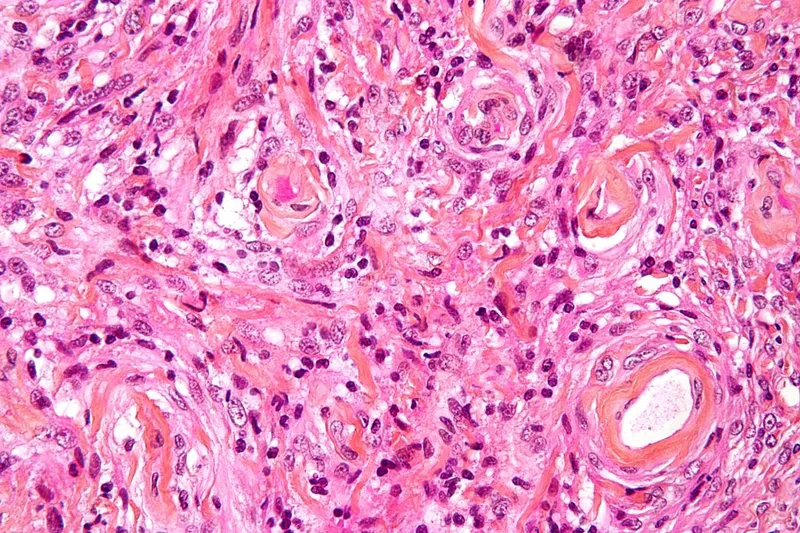
| Tumor Type | Skull Base Location | Growth Rate | Malignant Potential | 5-Year Survival | Recurrence Risk |
|---|---|---|---|---|---|
| Pituitary Adenoma | Sella turcica | Slow (2-3mm/year) | <1% | >98% | 5-15% |
| Meningioma Grade I | Multiple sites | Slow (1-2mm/year) | <5% | >95% | <10% GTR |
| Chordoma | Clivus, sacrum | Slow (3-5mm/year) | Low | 70-80% | 30-50% |
| Chondrosarcoma | Skull base, spine | Slow (2-4mm/year) | Moderate | 80-90% | 20-40% |
| Esthesioneuroblastoma | Nasal cavity, anterior skull base | Moderate (5-10mm/year) | High | 60-75% | 25-40% |
Chordomas arise from notochordal remnants, predominantly affecting the clivus (35-40%) and sacrococcygeal region (50-55%). These locally aggressive tumors demonstrate high recurrence rates (30-50%) following subtotal resection. Proton beam radiotherapy combined with maximal safe resection optimizes local control achieving 80-85% five-year progression-free survival.
- Chordoma Management Strategy
- Maximal safe resection: Primary goal with brainstem preservation
- Adjuvant radiotherapy: Proton beam preferred, >70 Gy total dose
- Long-term surveillance: Lifelong imaging every 6-12 months
- GTR achievement: <30% due to critical structure proximity
- Local control: 80-85% with combined therapy
- Distant metastasis: <10% but increases with recurrence
Esthesioneuroblastoma represents rare olfactory neoplasms (<5% skull base tumors) with bimodal age distribution and high metastatic potential. Kadish staging (A-D) correlates with prognosis: Stage A >90% five-year survival, Stage D <40% survival. Combined surgical resection and adjuvant radiotherapy achieve optimal outcomes.
💡 Master This: Skull base tumor biology determines treatment intensity - Benign lesions (pituitary adenomas, Grade I meningiomas) require complete resection for cure, locally aggressive tumors (chordomas) need multimodal therapy for control, malignant lesions (esthesioneuroblastoma) demand aggressive combined treatment for survival
Juvenile nasopharyngeal angiofibroma affects adolescent males exclusively, demonstrating characteristic vascular enhancement and aggressive local growth. Preoperative embolization reduces intraoperative blood loss from >2000mL to <500mL. Endoscopic resection achieves >95% cure rates with minimal morbidity in experienced centers.
Molecular markers increasingly guide treatment decisions - IDH mutations in skull base gliomas, BRAF mutations in craniopharyngiomas, and chromosomal alterations in chordomas provide prognostic information and targeted therapy opportunities.
Understanding pathology-specific characteristics enables tailored surgical planning, connecting tumor biology to optimal resection strategies and reconstruction requirements.
🧠 Pathology Spectrum: Tumor Territory Mastery
🔬 Diagnostic Precision: Pattern Recognition Mastery
Diagnostic precision in skull base pathology requires systematic integration of clinical symptoms, neurological findings, and advanced imaging. Pattern recognition enables accurate diagnosis in >90% of cases before tissue confirmation. Master clinicians utilize imaging signatures and clinical correlations to predict pathology and plan optimal management.
📌 Remember: IMAGES for skull base diagnosis - Intensity patterns, Mass effect, Anatomical location, Gadolinium enhancement, Edema patterns, Signal characteristics - Systematic analysis achieves >95% diagnostic accuracy
MRI sequences provide complementary information for tissue characterization and surgical planning. T1-weighted images demonstrate anatomy and gadolinium enhancement, T2-weighted sequences reveal tissue consistency and edema, while DWI differentiates cellular density and tumor grade.
- MRI Sequence Optimization
- T1 + Gadolinium: Enhancement patterns, vascular relationships
- T2-weighted: Tissue consistency, CSF spaces, edema
- FLAIR: Perilesional changes, infiltration patterns
- Pituitary protocol: Thin-section (1-2mm) coronal and sagittal
- Skull base protocol: Multiplanar with fat suppression
- Angiographic sequences: TOF or contrast-enhanced MRA
CT imaging remains essential for bone evaluation and surgical planning. High-resolution CT with bone algorithms demonstrates cortical destruction, calcification patterns, and pneumatization variants. CT angiography evaluates vascular encasement and collateral circulation.
| Imaging Finding | Pituitary Adenoma | Meningioma | Chordoma | Schwannoma | Chondrosarcoma |
|---|---|---|---|---|---|
| T1 Signal | Isointense | Isointense | Hypointense | Hypointense | Hypointense |
| T2 Signal | Variable | Variable | Hyperintense | Hyperintense | Hyperintense |
| Enhancement | Homogeneous | Intense homogeneous | Heterogeneous | Intense | Rings/septations |
| Bone Changes | Sellar expansion | Hyperostosis | Destruction | Expansion | Destruction |
| Calcification | Rare (<5%) | Common (20-25%) | Rare (<10%) | Rare (<5%) | Characteristic (>90%) |
Clinical presentation patterns correlate with anatomical involvement and tumor biology. Pituitary adenomas present with hormonal syndromes (60-70%) or visual field defects (40-50%). Skull base meningiomas cause cranial neuropathies (70-80%) and headaches (60-70%).
- Symptom-Location Correlations
- Anterior skull base: Anosmia (80-90%), frontal headaches (70-80%)
- Sellar region: Visual defects (60-70%), hormonal dysfunction (50-60%)
- Cavernous sinus: Diplopia (70-80%), facial numbness (60-70%)
- Bitemporal hemianopia: >90% specificity for chiasmal compression
- Cranial nerve III palsy: 85-90% indicates cavernous sinus involvement
- Trigeminal neuralgia: <5% associated with skull base pathology
Laboratory studies provide essential diagnostic information for functional pituitary adenomas. Hormonal evaluation includes prolactin, growth hormone, IGF-1, cortisol, and thyroid function. Dynamic testing with dexamethasone suppression or glucose tolerance confirms hormonal hypersecretion.
💡 Master This: Diagnostic accuracy improves systematically with multimodal integration - Clinical presentation provides 70-80% accuracy, adding MRI increases to 85-90%, incorporating CT achieves 90-95%, laboratory correlation reaches >95% diagnostic precision
Advanced imaging techniques enhance diagnostic precision and surgical planning. Diffusion tensor imaging maps white matter tracts, functional MRI localizes eloquent cortex, and perfusion imaging assesses tumor vascularity. Intraoperative imaging with CT or MRI enables real-time verification of resection completeness.
Molecular imaging with PET scanning differentiates tumor recurrence from radiation necrosis and assesses metabolic activity. 68Ga-DOTATATE PET demonstrates somatostatin receptor expression in pituitary adenomas and meningiomas, guiding medical therapy decisions.
Understanding diagnostic patterns enables accurate preoperative planning, connecting imaging characteristics to surgical approach selection and complication risk assessment.
🔬 Diagnostic Precision: Pattern Recognition Mastery
⚔️ Surgical Execution: Technical Mastery Framework
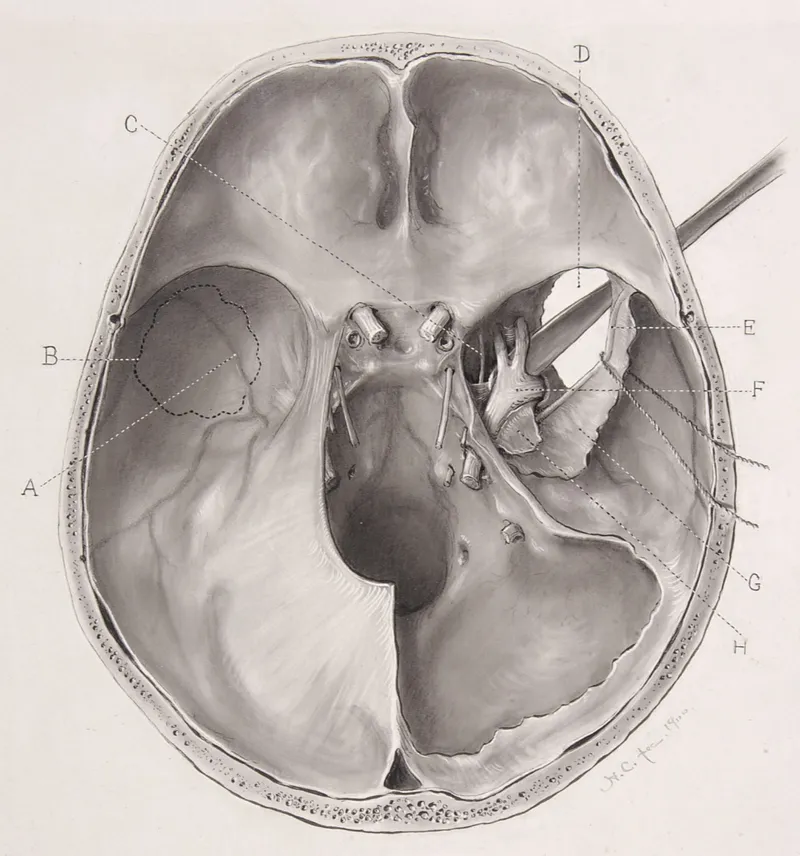
Surgical execution in skull base procedures requires systematic methodology integrating anatomical knowledge, technical skills, and decision-making algorithms. Master surgeons follow standardized protocols while adapting techniques based on intraoperative findings and patient-specific anatomy. Successful outcomes depend on meticulous planning, precise execution, and complication prevention.
📌 Remember: PRECISE for surgical execution - Planning, Recognition, Exposure, Control, Identification, Safety, Evaluation - Each step requires >95% accuracy for optimal outcomes
Preoperative planning establishes surgical strategy based on imaging analysis, patient factors, and surgeon experience. 3D reconstruction and virtual reality planning improve spatial understanding and reduce operative time by 15-25%. Multidisciplinary planning with anesthesia, otolaryngology, and radiation oncology optimizes perioperative care.
- Preoperative Planning Elements
- Imaging review: Multiplanar reconstruction, vascular mapping
- Approach selection: Risk-benefit analysis, surgeon expertise
- Equipment preparation: Specialized instruments, monitoring systems
- Navigation setup: Registration accuracy <2mm error
- Monitoring preparation: Cranial nerve and brainstem monitoring
- Blood product availability: Type and crossmatch for >1000mL loss
Exposure techniques vary by anatomical region and pathology characteristics. Endoscopic approaches provide superior visualization with 4K resolution and angled optics but require two-surgeon technique for optimal efficiency. Microscopic approaches offer stereoscopic vision and instrument stability for precise dissection.
| Surgical Phase | Critical Actions | Time Allocation | Complication Risk | Success Metrics | Monitoring Requirements |
|---|---|---|---|---|---|
| Exposure | Anatomical identification | 20-30% | Low (5-10%) | Complete visualization | Standard monitoring |
| Tumor Identification | Tissue characterization | 10-15% | Moderate (10-15%) | Clear tumor margins | Neuromonitoring |
| Devascularization | Vascular control | 15-25% | High (20-30%) | Hemostasis | Vascular monitoring |
| Tumor Removal | Systematic resection | 30-40% | Highest (25-40%) | GTR achievement | Continuous monitoring |
| Reconstruction | Defect repair | 15-20% | Moderate (15-20%) | Watertight closure | CSF pressure monitoring |
Intraoperative monitoring prevents neurological complications through real-time assessment of cranial nerve and brainstem function. Electromyography monitors facial nerve function with >95% sensitivity, while brainstem auditory evoked potentials assess hearing preservation during cerebellopontine angle surgery.
- Monitoring Modalities
- Cranial nerve EMG: Facial, trigeminal, hypoglossal nerves
- Brainstem monitoring: BAER, SSEP for posterior fossa surgery
- Visual monitoring: VEP for sellar and suprasellar lesions
- Facial nerve monitoring: >95% preservation with continuous EMG
- Hearing preservation: 70-80% with BAER monitoring
- Visual field preservation: 85-90% with careful chiasmal dissection
Hemostasis techniques prevent catastrophic bleeding and maintain visualization. Bipolar coagulation provides precise control for small vessels, while hemostatic agents and vascular clips manage larger bleeding. Endovascular techniques enable preoperative embolization reducing blood loss by 60-80%.
💡 Master This: Surgical success correlates directly with systematic execution - Deviation from protocol increases complication rates by 2-3 fold. Intraoperative decision-making requires real-time risk assessment balancing resection goals against functional preservation
Reconstruction principles prevent CSF leaks and restore anatomical integrity. Multilayer closure with autologous grafts achieves >95% success in preventing CSF leaks. Vascularized flaps provide robust reconstruction for large defects with <5% failure rates.
Quality metrics assess surgical performance including gross total resection rates, complication frequencies, and functional outcomes. Learning curves demonstrate improvement over 50-100 cases for complex procedures, emphasizing training requirements and volume-outcome relationships.
Understanding surgical execution principles enables optimal operative performance, connecting technical skills to patient outcomes and complication prevention strategies.
⚔️ Surgical Execution: Technical Mastery Framework
🛡️ Complication Management: Risk Mitigation Mastery
Complication management in skull base surgery demands proactive prevention, early recognition, and systematic intervention. Major complications occur in 15-25% of cases with permanent morbidity affecting 5-10% of patients. Master surgeons anticipate potential complications and implement prevention strategies while maintaining rescue protocols for immediate intervention.
📌 Remember: RESCUE for complication management - Recognition, Evaluation, Stabilization, Control, Understanding, Execution - Rapid response within <5 minutes prevents permanent injury in >80% of cases
CSF leaks represent the most common complication (10-25% incidence) with significant morbidity if unrecognized. Intraoperative identification enables immediate repair with >95% success, while delayed recognition increases meningitis risk to 15-20% and repair failure to 30-40%.
- CSF Leak Management Protocol
- Immediate recognition: Clear fluid drainage, glucose testing
- Intraoperative repair: Multilayer closure, tissue grafts
- Postoperative monitoring: Lumbar drainage, bed rest
- High-flow leaks: >100mL/day require surgical repair
- Low-flow leaks: <50mL/day may resolve with conservative management
- Repair success: >95% with immediate intervention, <70% with delayed repair
Vascular injuries occur in 3-8% of skull base procedures with catastrophic potential. Internal carotid artery injury carries 15-20% mortality and 30-40% stroke risk. Immediate recognition and vascular control prevent exsanguination and enable repair or bypass procedures.
| Complication Type | Incidence Rate | Recognition Time | Intervention Window | Permanent Morbidity | Prevention Strategy |
|---|---|---|---|---|---|
| CSF Leak | 10-25% | Immediate-24hrs | <48 hours | <5% | Multilayer closure |
| Cranial Nerve Injury | 15-30% | Immediate | <6 hours | 10-20% | Continuous monitoring |
| Vascular Injury | 3-8% | Immediate | <30 minutes | 30-50% | Careful dissection |
| Infection | 5-15% | 24-72 hours | <24 hours | <10% | Antibiotic prophylaxis |
| Pneumocephalus | 20-40% | Immediate-24hrs | <12 hours | <5% | Complete reconstruction |
Cranial nerve injuries affect 15-30% of patients with permanent deficits in 10-20%. Facial nerve injury causes cosmetic and functional deficits, while lower cranial nerve injuries affect swallowing and voice. Immediate recognition through electrophysiological monitoring enables protective measures and early intervention.
- Cranial Nerve Injury Management
- Facial nerve: Immediate repair vs delayed reconstruction
- Trigeminal nerve: Sensory rehabilitation, pain management
- Lower cranial nerves: Swallowing evaluation, aspiration prevention
- Facial nerve preservation: >95% with continuous monitoring
- Hearing preservation: 70-80% in acoustic neuroma surgery
- Swallowing function: Recovery in 80-90% within 6-12 months
Infection complications include meningitis (2-5%), brain abscess (<1%), and wound infections (5-10%). Antibiotic prophylaxis reduces infection rates by 50-70%, while sterile technique and minimal operative time provide additional protection.
💡 Master This: Complication prevention requires systematic risk assessment - Patient factors (diabetes, immunosuppression) increase infection risk 2-3 fold, operative factors (duration >6 hours, CSF exposure) increase leak risk 3-4 fold, surgeon factors (experience <50 cases) increase overall complications 2-fold
Postoperative monitoring protocols enable early complication detection and intervention. Neurological assessments every 2-4 hours detect >90% of neurological deterioration within 12 hours. Laboratory monitoring tracks electrolyte imbalances and hormonal dysfunction common after pituitary surgery.
Damage control strategies limit complication severity when prevention fails. Immediate surgical intervention for expanding hematomas, aggressive medical management for cerebral edema, and multidisciplinary care for complex complications optimize recovery potential.
Understanding complication management enables optimal patient safety, connecting risk recognition to intervention strategies and long-term outcome optimization.
🛡️ Complication Management: Risk Mitigation Mastery
🎯 Clinical Mastery Arsenal: Rapid Decision Framework
Clinical mastery in skull base surgery requires systematic decision frameworks enabling rapid assessment, accurate diagnosis, and optimal treatment selection. Expert surgeons utilize pattern recognition, evidence-based protocols, and experience-derived algorithms to navigate complex scenarios with >95% accuracy and minimal complications.
📌 Remember: MASTER framework - Multimodal assessment, Anatomical correlation, Systematic approach, Technical execution, Evidence integration, Risk stratification - Systematic application achieves optimal outcomes in >90% of cases
Essential Clinical Thresholds for immediate decision-making:
- Pituitary Surgery Thresholds
- Tumor size: <1cm (95% cure), >4cm (<70% cure)
- Cavernous sinus invasion: Knosp 0-2 (>90% GTR), 3-4 (<60% GTR)
- Visual field defects: >75% improvement with decompression <6 months
- Prolactin levels: >200 ng/mL suggests macroadenoma
- Growth hormone: >10 ng/mL post-glucose indicates acromegaly
- Cortisol: Midnight >50 μg/dL suggests Cushing's disease
Rapid Assessment Protocol for skull base emergencies:
| Clinical Scenario | Assessment Time | Key Decision Points | Intervention Window | Success Rate | Alternative Options |
|---|---|---|---|---|---|
| Pituitary Apoplexy | <30 minutes | Visual defects, consciousness | <24 hours | >90% | Medical management |
| CSF Leak | <15 minutes | Flow rate, glucose positive | <48 hours | >95% | Conservative management |
| Carotid Injury | <5 minutes | Bleeding control, neurological status | <30 minutes | 60-70% | Endovascular repair |
| Cranial Nerve Deficit | <10 minutes | Function assessment, monitoring changes | <6 hours | 70-80% | Delayed reconstruction |
| Postop Hematoma | <20 minutes | Mass effect, neurological decline | <2 hours | 80-90% | Medical management |
Pattern Recognition Mastery for common presentations:
- "See This, Think That" Correlations
- Bitemporal hemianopia + headaches = Pituitary macroadenoma (>95% specificity)
- Unilateral hearing loss + tinnitus = Acoustic neuroma (>90% specificity)
- Facial pain + diplopia = Cavernous sinus pathology (>85% specificity)
- Progressive visual loss: Urgent decompression within <24 hours
- Sudden hearing loss: Immediate audiometry and MRI within 48 hours
- New cranial nerve palsy: Emergency imaging and neurosurgical consultation
Evidence-Based Decision Trees for treatment selection:
💡 Master This: Clinical expertise develops through systematic pattern exposure - >100 cases required for basic competency, >300 cases for advanced skills, >500 cases for master-level decision-making. Continuous learning and outcome analysis maintain optimal performance
Critical Numbers Arsenal for immediate reference:
- Normal Values: Prolactin <25 ng/mL, GH <5 ng/mL, Cortisol 5-25 μg/dL
- Surgical Thresholds: GTR >95% resection, STR 80-95% resection
- Complication Rates: CSF leak 10-25%, permanent CN deficit 5-15%
- Outcome Metrics: Cure rates 80-95% functional adenomas, recurrence <10% at 5 years
Rapid Reference Tools for intraoperative decisions:
Understanding clinical mastery frameworks enables expert-level decision-making, connecting systematic assessment to optimal treatment selection and superior patient outcomes across all skull base pathology.
🎯 Clinical Mastery Arsenal: Rapid Decision Framework
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app












