Cold Chain Basics - Chill Factor
- Cold Chain: System maintaining vaccines at +2°C to +8°C (WHO standard) from manufacturer to recipient.
- Goal: Preserve vaccine potency; lost potency is irreversible.
- Chill Factor: The significant risk of accidental freezing of certain vaccines, especially at peripheral storage points.
- Freeze-sensitive vaccines: DPT, TT, HepB, IPV, Pentavalent, PCV, Rotavirus (liquid), liquid Hib.
- These contain adjuvants (e.g., aluminium salts) sensitive to freezing.
- Consequences of freezing:
- Irreversible ↓potency (adjuvant clumping/damage).
- ↑Local reactogenicity.
- Freeze-sensitive vaccines: DPT, TT, HepB, IPV, Pentavalent, PCV, Rotavirus (liquid), liquid Hib.
⭐ The "Shake Test" is crucial for detecting prior freezing in adsorbed vaccines (e.g., DPT, TT, HepB), as visual inspection alone is often insufficient to detect freeze damage.
Essential Equipment - The Cool Kit
- Walk-in Cold Rooms (WICR): Regional. Temp: +2°C to +8°C. Storage: 3 months.
- Deep Freezers (DF): Regional/District. Temp: -15°C to -25°C. Stores OPV, Measles etc. Makes ice packs.
- Ice-Lined Refrigerators (ILR): PHC. Temp: +2°C to +8°C. All vaccines. Holdover: 8-12 hrs.
- Cold Boxes: Transport. Temp: +2°C to +8°C. Duration: 5-7 days (frozen ice packs).
- Vaccine Carriers: Outreach. Holds 16-20 vials. Temp: +2°C to +8°C. Duration: 12-24 hrs (4 conditioned ice packs).
- Day Carriers: Smallest. Holds 6-8 vials. Duration: Few hours.
- Ice Packs: Water-filled, frozen. Condition before use with freeze-sensitive vaccines.

⭐ ILRs maintain temperature (+2°C to +8°C) for 8-12 hours (holdover time) during power outages, vital for vaccine viability at PHCs.
Temperature Tracking - Degrees of Defense
- Crucial for vaccine potency; maintains storage between +2°C to +8°C.
- Multiple "Degrees of Defense":
- Thermometers (Dial, Stem): Placed in ILR/DF; readings recorded twice daily.
- Vaccine Vial Monitor (VVM): Attached to vaccine vials; indicates cumulative heat exposure.
- Freeze Indicators (e.g., Freeze Watch) & Shake Test: Detect exposure to freezing temperatures (critical for freeze-sensitive vaccines like DPT, TT, HepB, IPV).
- Temperature Log Books/Charts: Continuous monitoring.
⭐ VVM: If the inner square is darker than or the same color as the outer reference circle, the vaccine has passed its heat exposure limit and must be discarded.
Vaccine Care & VVM - Vial Guardians
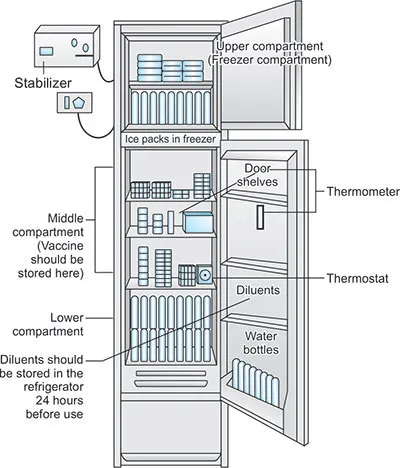
- Vaccine Storage (ILR/Refrigerator):
- Top/Basket 1 (Most Heat-Sensitive): OPV, Measles, MR, JE, Rotavirus, BCG.
- Bottom (Freeze-Sensitive, keep away from freezer): DPT, TT, Hep B, Pentavalent, IPV, PCV.
- Diluents: Store with respective vaccines or in door pockets (2-8°C); never freeze.
- Shake Test (for Freeze-Sensitive Vaccines):
- Suspect freezing for: DPT, TT, Hep B, Pentavalent, IPV, PCV.
- Compare sedimentation rate with a deliberately frozen then thawed vial (control).
- If test vial sediments slower than control: USE.
- If test vial sediments at same rate or faster: DISCARD.
- VVM (Vaccine Vial Monitor): Monitors cumulative heat exposure.
- Inner square (IS), outer circle (OC).
- Reading VVM & Action:
- IS lighter than OC: USE.
- IS same color as OC: DISCARD (Discard Point).
- IS darker than OC: DISCARD.
⭐ OPV VVM is most heat-sensitive, changing color fastest.
System Failures - Meltdown Management
- Causes: Power failure, equipment malfunction, human error (improper storage, poor monitoring).
- Consequences: Loss of vaccine potency, risk of VPI (Vaccine Preventable Illness) outbreak.
- Shake Test: Differentiates between frozen (damaged) T-series vaccines (DPT, TT, Hep B, Pentavalent) and usable ones. If sediment is granular & doesn't resuspend easily after shaking, vaccine is damaged.
⭐ VVM (Vaccine Vial Monitor) is the most reliable indicator for heat exposure of OPV, Measles, mumps, rubella, BCG, JE, Rotavirus, HPV vaccines. Discard if inner square is darker than or same color as outer circle (Stage 3 or 4).
High-Yield Points - ⚡ Biggest Takeaways
- Cold chain maintains vaccine potency from manufacturer to beneficiary at +2°C to +8°C.
- OPV is the most heat-sensitive vaccine; Hepatitis B, DPT, TT are key freeze-sensitive vaccines (never freeze).
- Vaccine Vial Monitor (VVM) indicates cumulative heat exposure; discard if inner square is darker/same as outer circle.
- The Shake Test identifies damage in freeze-sensitive vaccines (e.g., DPT, Hep B) if freezing is suspected.
- Ice-Lined Refrigerators (ILRs) at PHCs maintain temperature for extended periods during power outages.
- Cold boxes and vaccine carriers are essential for transporting vaccines to outreach immunization sessions securely.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

