Primary Structure: Definition & Basics - Amino Acid Chain Gang
- Definition: The linear sequence of amino acids in a polypeptide chain, determining the protein's identity.
- Peptide Bond: Amino acids are covalently linked by peptide bonds ($CONH$).
- Formed via a dehydration reaction between the α-carboxyl group of one amino acid and the α-amino group of the next.
- Termini & Directionality:
- N-terminus (Amino-terminus): The start of the chain, possessing a free amino group (-$NH_2$).
- C-terminus (Carboxyl-terminus): The end of the chain, possessing a free carboxyl group (-$COOH$).
- Read from N → C terminus.

⭐ The primary structure dictates all higher levels of protein structure (secondary, tertiary, quaternary) and thus its ultimate biological function. This sequence is genetically determined by DNA.
Peptide Bond: Characteristics - The Backbone's Blueprint
- Partial Double Bond Character:
- Due to resonance, the C-N peptide bond has ~40% double bond character.
- This makes it rigid and planar.

- Consequences of Rigidity:
- Restricted Rotation: No rotation around the peptide bond (C-N axis) itself.
- Trans Configuration Favored:
- Adjacent $\alpha$-carbons ($C_{\alpha}$) are usually trans to minimize steric hindrance between R-groups.
- Exception: Proline (X-Pro bonds) can adopt cis configuration more readily.
- Backbone Flexibility:
- Rotation is permitted around bonds connected to the $\alpha$-carbon ($C_{\alpha}$):
- N-$C_{\alpha}$ bond: Angle $\phi$ (phi).
- $C_{\alpha}$-C bond: Angle $\psi$ (psi).
- These $\phi$ and $\psi$ angles (Ramachandran angles) determine the polypeptide chain's conformation.
- Rotation is permitted around bonds connected to the $\alpha$-carbon ($C_{\alpha}$):
⭐ The peptide bond has approximately 40% double bond character due to resonance, making it rigid and planar.
Sequence Determination: Methods - Cracking the Protein Code
Crucial for protein function, evolution, & disease understanding.
- N-Terminal Analysis:
- Sanger's Reagent (FDNB): Identifies N-terminal AA. Forms DNP-AA. Destroys peptide.
- Edman Degradation (PITC): Sequential N-terminal AA ID. Phenylisothiocyanate (PITC) reacts with N-terminus, cleaved as PTH-AA. Peptide intact for next cycle.
⭐ Edman degradation allows sequential N-terminal AA removal and identification.
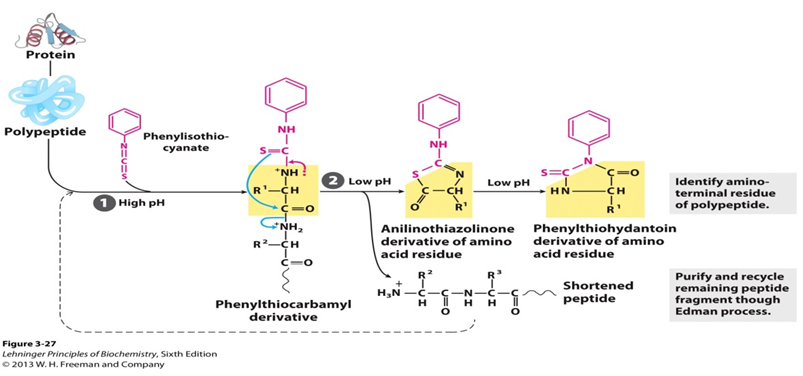
-
C-Terminal Analysis:
- Carboxypeptidases: Cleave C-terminal AAs.
- A: Most AAs (not Arg, Lys, Pro).
- B: Arg, Lys.
- Y: Most AAs.
- Carboxypeptidases: Cleave C-terminal AAs.
-
Specific Cleavage (Enzymatic): Fragments protein.
- Trypsin: Cleaves at C-side of Lys (K), Arg (R) (not if Pro follows). 📌 Try-K/R.
- Chymotrypsin: Cleaves at C-side of Phe (F), Tyr (Y), Trp (W) (aromatics) (not if Pro follows). 📌 Chymo-F/Y/W.
-
Mass Spectrometry (MS): Modern, rapid, sensitive for sequencing & ID.
Clinical Correlation: Primary Structure Defects - When Sequences Go Wrong
- Genetic mutations alter amino acid sequences, causing defective proteins and disease.
- Key Examples:
- Sickle Cell Anemia (HbS): $\beta$-globin gene mutation: Glu6Val (Glutamic acid $\rightarrow$ Valine at position 6).
⭐ Sickle cell anemia is caused by a single amino acid substitution (Glu$\rightarrow$Val at position 6) in the $\beta$-globin chain of hemoglobin.
- Cystic Fibrosis (CFTR): $\Delta$F508 (Phenylalanine deletion at 508) in CFTR protein.
- Phenylketonuria (PKU): Mutations in Phenylalanine Hydroxylase (PAH) enzyme.
- Sickle Cell Anemia (HbS): $\beta$-globin gene mutation: Glu6Val (Glutamic acid $\rightarrow$ Valine at position 6).
- Substitutions Impact:
- Conservative: Similar amino acid; minor functional change.
- Non-conservative: Dissimilar amino acid; major functional disruption.

High‑Yield Points - ⚡ Biggest Takeaways
- Primary structure: The linear sequence of amino acids linked by peptide bonds.
- Peptide bond: Exhibits partial double bond character, resulting in rigidity and planarity.
- Genetic determination: The amino acid sequence is encoded by DNA.
- Structural determinant: Dictates all higher levels of protein structure and thus function.
- Directionality: Defined by the N-terminus (amino) and C-terminus (carboxyl).
- Clinical significance: Sequence changes (e.g., sickle cell anemia: Glu⁶Val) can lead to disease.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

