PTMs Overview - Protein Polishers
Post-Translational Modifications (PTMs) are chemical alterations to proteins after translation. They are crucial for protein function and diversity.
- Definition: Covalent processing of proteins following synthesis.
- Sites: Occur in cytosol, endoplasmic reticulum (ER), and Golgi apparatus.
- Significance:
- Functional Diversity: Expands proteome complexity beyond genome.
- Regulation: Modulates enzyme activity, protein stability.
- Signaling: Key in signal transduction pathways (e.g., phosphorylation).
- Localization: Directs proteins to specific cellular compartments.
- Interactions: Influences protein-protein or protein-ligand binding.
- Degradation: Marks proteins for proteasomal breakdown (e.g., ubiquitination).
⭐ Exam Favourite: Phosphorylation, primarily on serine, threonine, and tyrosine residues, is one of the most common and critical PTMs for regulating cellular processes, including enzyme activity and signal transduction cascades. It's often reversible by phosphatases.
📌 Mnemonic: "Proteins Get Modified After Synthesis" (PTMs, Golgi, ER, Cytosol, Significance).
Phosphorylation & Glycosylation - Signal & Sugar Coats
-
Phosphorylation: Reversible addition of $PO_4^{3-}$ to Ser, Thr, or Tyr residues.
- Catalyzed by kinases (use ATP); reversed by phosphatases.
- Function: Regulates protein activity; vital for cell signaling (e.g., insulin receptor) and metabolic control (e.g., glycogen metabolism).
-
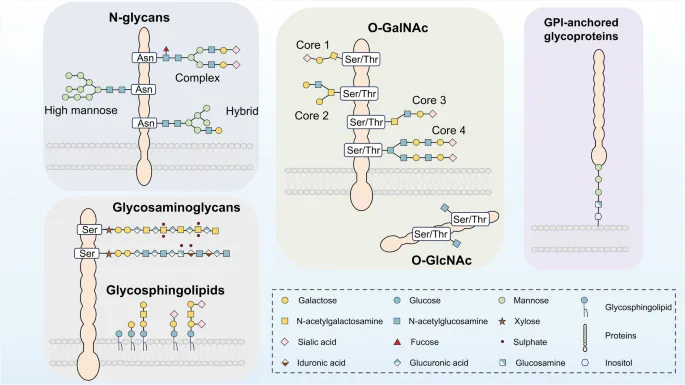
Glycosylation: Attachment of sugar chains (glycans) to proteins.
- N-linked: To Asparagine (Asn) in Asn-X-Ser/Thr sequence (X ≠ Pro).
- Site: Endoplasmic Reticulum (ER); involves lipid carrier dolichol phosphate.
- O-linked: To Serine (Ser) or Threonine (Thr) hydroxyl groups.
- Site: Golgi apparatus.
- Functions: Crucial for protein folding, ↑stability, targeting, cell recognition (e.g., ABO blood groups, antibodies). 📌 N-linked: Asn (ER); O-linked: Ser/Thr (Golgi).
- N-linked: To Asparagine (Asn) in Asn-X-Ser/Thr sequence (X ≠ Pro).
⭐ The ABO blood group system is determined by glycosyltransferases that add specific sugars to the H antigen, an example of O-linked glycosylation on red blood cell surface proteins and lipids.
Ubiquitination & Beyond - Tag, Regulate, Build
Error generating content for this concept group: Failed to process successful response
PTMs in Disease - Clinical Links
- I-cell Disease: Lysosomal enzyme targeting defect due to faulty Mannose-6-Phosphate (M6P) PTM.
- Alzheimer's Disease: Hyperphosphorylation of Tau protein forms neurofibrillary tangles.
- Cancer: Aberrant PTMs in signaling pathways (e.g., ↑EGFR phosphorylation).
- Scurvy: Defective collagen hydroxylation (requires Vitamin C) leads to weakened connective tissue.
- PTMs as Drug Targets:
- Kinase inhibitors (e.g., imatinib).
- HDAC inhibitors (e.g., vorinostat).
⭐ Faulty ubiquitination, a PTM, is implicated in Parkinson's disease, affecting protein degradation and Lewy body formation.
High‑Yield Points - ⚡ Biggest Takeaways
- Phosphorylation (Ser/Thr/Tyr) is a key reversible regulatory PTM (kinases/phosphatases).
- Glycosylation: N-linked (Asn, ER) for folding/sorting; O-linked (Ser/Thr, Golgi) for stability.
- Ubiquitination primarily targets proteins for proteasomal degradation.
- Histone acetylation/methylation are crucial for gene expression regulation.
- Collagen hydroxylation (Pro, Lys) requires Vitamin C for structural integrity.
- Gamma-carboxylation (Glu) in clotting factors (II,VII,IX,X) needs Vitamin K.
- Disulfide bonds (Cys-Cys) stabilize many extracellular proteins.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

