Na⁺/K⁺ ATPase Basics - Pump It Up!
- Definition: An active transporter enzyme crucial for maintaining electrochemical gradients across the plasma membrane.
- Type: P-type ATPase (forms a phosphorylated intermediate).
- Location: Ubiquitous in animal cell plasma membranes.
- Stoichiometry & Energy: Pumps $3 Na^+{out} / 2 K^+{in}$ per ATP hydrolyzed.
⭐ The Na⁺/K⁺ ATPase is specifically inhibited by cardiac glycosides like digoxin and ouabain, which bind to the extracellular K⁺ site (E2-P form).
Pump Structure - The Molecular Machine
- Heterodimer: α and β subunits.
- α-subunit: Catalytic; binds 3 Na⁺, 2 K⁺, ATP. Has phosphorylation & cardiac glycoside (e.g., ouabain) sites.
- β-subunit: Glycosylated; aids α-subunit maturation & correct membrane insertion. Stabilizes α-subunit.
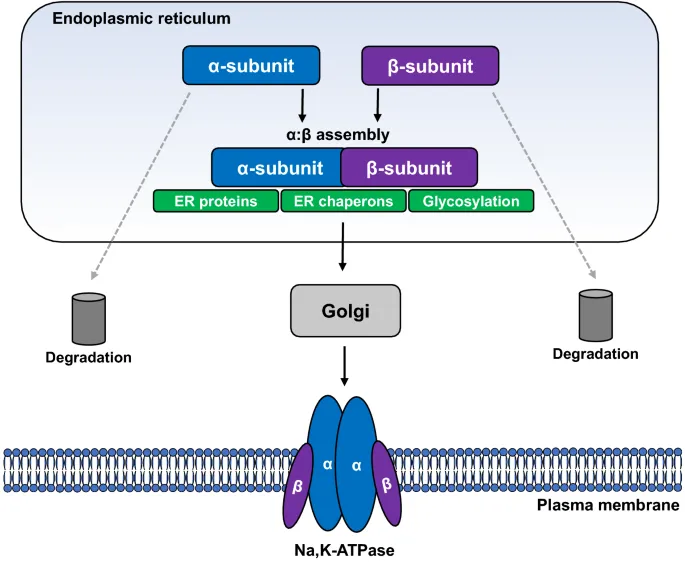
⭐ The α-subunit houses all key functional sites: ion (Na⁺, K⁺) and ATP binding, phosphorylation, and cardiac glycoside interaction.
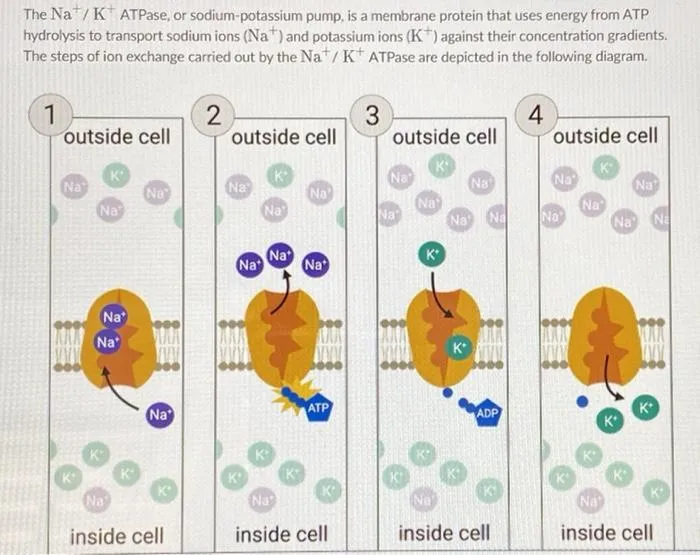
Mechanism Unveiled - The Pump Cycle
The Na⁺/K⁺-ATPase, a P-type ATPase, operates via the Post-Albers cycle, alternating between E1 (cytosolic) and E2 (extracellular-facing) conformations.
- E1 State (Cytosolic facing):
- High Na⁺ affinity; binds ATP & 3 Na⁺ from cytosol.
- $E1 + ATP + 3Na^+_{in} \rightarrow E1 \cdot ATP \cdot 3Na^+$
- Phosphorylation $\rightarrow$ E1~P:
- ATP hydrolysis: $E1 \cdot ATP \cdot 3Na^+ \rightarrow E1\text{\textasciitilde}P \cdot 3Na^+ + ADP$.
- Conformational change to E2-P (Na⁺ translocated out).
- $E1\text{\textasciitilde}P \cdot 3Na^+ \rightarrow E2\text{\textasciitilde}P \cdot 3Na^+$
- E2-P State (Extracellular facing):
- Releases 3 Na⁺ to extracellular space: $E2\text{\textasciitilde}P \cdot 3Na^+ \rightarrow E2\text{\textasciitilde}P + 3Na^+_{out}$.
- High K⁺ affinity; binds 2 K⁺ from extracellular space.
- $E2\text{\textasciitilde}P + 2K^+_{ext} \rightarrow E2\text{\textasciitilde}P \cdot 2K^+$
- Dephosphorylation (K⁺ dependent) $\rightarrow$ E2:
- $E2\text{\textasciitilde}P \cdot 2K^+ \rightarrow E2 \cdot 2K^+ + P_i$.
- Conformational change to E1 (K⁺ translocated in).
- $E2 \cdot 2K^+ \rightarrow E1 \cdot 2K^+$
- Releases 2 K⁺ to cytosol: $E1 \cdot 2K^+ \rightarrow E1 + 2K^+_{in}$.
📌 Mnemonic: KIN (K⁺ In), NAO (Na⁺ Out). Pumps 3 Na⁺ Out, 2 K⁺ In.

⭐ Cardiac glycosides (e.g., Digoxin) inhibit Na⁺/K⁺-ATPase by binding to the E2-P (phosphorylated) form on its extracellular side.
Pump's Purpose - Cellular Gatekeeper
- Maintains resting membrane potential (RMP), crucial for nerve and muscle cell excitability.
- Regulates cell volume by controlling intracellular solute concentration, preventing osmotic lysis or crenation.
- Drives secondary active transport for solutes like glucose and amino acids (e.g., Na⁺-glucose cotransport) and ions (e.g., Na⁺/Ca²⁺ exchange).
- Establishes and preserves steep electrochemical gradients for $Na⁺$ (low inside) and $K⁺$ (high inside).

⭐ The pump transports 3 $Na⁺$ ions out of the cell and 2 $K⁺$ ions into the cell for each molecule of ATP hydrolyzed, making the inside of the cell negative relative to the outside (electrogenic effect).
Pump Control - Who's the Boss?
- Short-term:
- Substrate availability: Intracellular $Na^+$ ([Na⁺]i), extracellular $K^+$ ([K⁺]o), ATP.
- Long-term:
- Hormonal control: Insulin ↑, Aldosterone ↑, Thyroid hormones ↑ activity/synthesis.
- Covalent modification: Primarily via phosphorylation by PKA/PKC, altering pump activity.
- Endogenous regulators: e.g., endogenous ouabain-like substances.
⭐ Insulin promotes $Na^+$-$K^+$ ATPase activity, driving $K^+$ into cells, which can cause hypokalemia.
Clinical Angles - When Pumps Fail
- Inhibitors: Cardiac glycosides (e.g., Digoxin).
- Bind E2-P form, block dephosphorylation → ↑ [Na⁺]i → reverses Na⁺/Ca²⁺ exchanger → ↑ [Ca²⁺]i → ↑ contractility.
- Digoxin:
- Uses: Heart failure, atrial fibrillation. Therapeutic range: 0.5-0.9 ng/mL.
- Toxicity: 📌 BEWS (Blurred vision/yellow halos, EKG changes, Weakness, Stomach upset).
- K⁺ Levels: Hypokalemia ↑ Digoxin toxicity; Hyperkalemia ↓ efficacy.
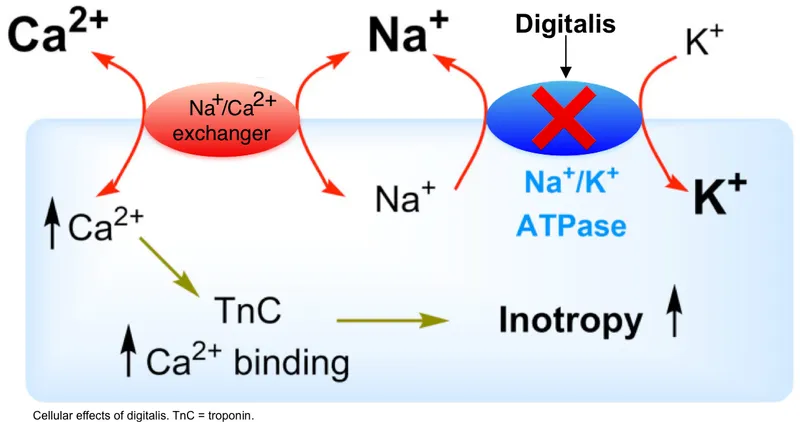
⭐ Hypokalemia potentiates Digoxin toxicity by increasing its binding to the Na⁺/K⁺-ATPase pump, leading to enhanced effects and risk of adverse events.
High‑Yield Points - ⚡ Biggest Takeaways
- A P-type ATPase crucial for active ion transport across plasma membranes.
- Establishes ion gradients by pumping 3 Na+ out and 2 K+ in.
- Utilizes one ATP molecule per transport cycle for energy.
- Maintains resting membrane potential, cell volume, and drives secondary active transport.
- Specifically inhibited by cardiac glycosides (e.g., Digoxin, Ouabain).
- Its activity is electrogenic, contributing to the negative intracellular potential an_average_of_-10mV_to_the_RMP_in_neurons
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

