Thalassemias: Basics & Globin Genes - Chain Gang Genetics
- Quantitative genetic disorders: Reduced/absent synthesis of α or β globin chains, components of hemoglobin (e.g., $HbA (\alpha_2\beta_2)$).
- Causes globin chain imbalance → ineffective erythropoiesis, hemolysis, anemia.
- Globin Gene Details:
- α-globin cluster: Chromosome 16 (HBA1, HBA2) - 4 genes (αα/αα).
- β-globin cluster: Chromosome 11 (HBB) - 2 genes (β/β).
⭐ Normal adult Hb (HbA) is two α-chains & two β-chains. Humans: 4 α-globin genes (Chr 16, 2/chromosome) & 2 β-globin genes (Chr 11, 1/chromosome).
Alpha Thalassemias - Alpha‑Numeric Nightmares
- Defect: ↓ α-globin synthesis from gene deletions (HBA1/HBA2, Chr 16). Severity α number of deleted genes (1-4).

- HbH Disease: Excess β-chains → β₄ (HbH).
- Hb Bart's: Excess γ-chains → γ₄ (Hb Bart's) in fetuses.
⭐ Hb Bart's (γ₄), from 4 α-globin gene deletions, causes hydrops fetalis; lethal postnatally.
Beta Thalassemias - Beta Blockage Breakdown
- Defect in β-globin chain synthesis (HBB gene, Chr 11). Excess α-chains precipitate → ineffective erythropoiesis, hemolysis.
- Types:
- Minor (Trait): Genotype $β/β^+$ or $β/β^0$. Asymptomatic or mild microcytic anemia. ↑ HbA₂ >3.5%.
- Intermedia: Variable severity, later presentation.
- Major (Cooley's Anemia): Genotype $β^0/β^0$. Severe anemia by 4-6 months. Hepatosplenomegaly, bone deformities. Lifelong transfusions & iron chelation. ↑↑ HbF.
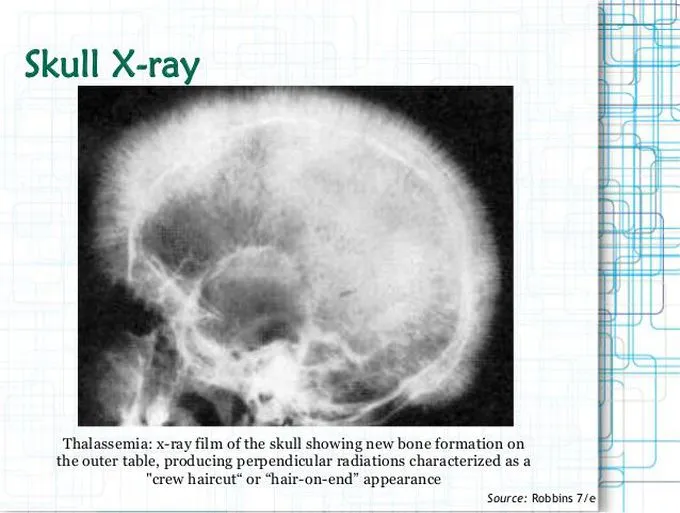
⭐ The characteristic "crew-cut" or "hair-on-end" appearance on skull X-ray in β-thalassemia major is due to massive expansion of erythroid marrow.
- 📌 Beta = Bone marrow expansion.
Clinical Features & Complications - Systemic Storm
- Severe anemia: Pallor, poor growth, FTT.
- Extramedullary hematopoiesis (EMH):
- Massive hepatosplenomegaly.
- Bone marrow expansion: "Chipmunk facies", frontal bossing, fractures.
- Iron overload (transfusions, ↑GIT absorption):
- Cardiac: Cardiomyopathy (major cause of death).
- Hepatic: Cirrhosis.
- Endocrine: Diabetes, hypogonadism.
- Others: Gallstones, infections.

⭐ Iron overload, a consequence of repeated blood transfusions and increased gastrointestinal absorption, is the primary cause of mortality and morbidity in transfusion-dependent thalassemias, particularly affecting the heart, liver, and endocrine organs.
Diagnosis & Management - Decoding & Defending
-
Investigations:
- CBC: Microcytic hypochromic anemia, ↑RBC count (thal trait), ↑RDW.
- Peripheral Smear: Target cells, anisopoikilocytosis, basophilic stippling.

- Iron studies: Normal/↑ serum iron & ferritin (crucial to differentiate from IDA).
- Hb Electrophoresis/HPLC: Definitive.
⭐ An HbA2 level greater than 3.5% on hemoglobin electrophoresis or HPLC is a key diagnostic marker for β-thalassemia trait.
- β-thal major: Markedly ↑HbF (often >90%), ↓/absent HbA.
- α-thal (HbH disease): HbH (β4) detected.
- DNA analysis: Confirmatory, carrier screening, prenatal diagnosis.
-
Management Principles:
- Thalassemia Minor/Trait: Usually no treatment; genetic counseling vital.
- Thalassemia Major & Intermedia (symptomatic):
- Regular packed RBC transfusions (maintain pre-transfusion Hb 9-10.5 g/dL).
- Iron chelation therapy (e.g., Deferasirox, Deferoxamine) to prevent hemosiderosis.
- Folic acid supplementation.
- Splenectomy: For hypersplenism, ↑transfusion needs.
- Allogeneic HSCT: Only curative option.
High‑Yield Points - ⚡ Biggest Takeaways
- Thalassemias: Quantitative defects in globin synthesis (α or β chains).
- α-Thalassemia: Caused by gene deletions (chromosome 16). Hb Bart's (γ4) in severe forms (hydrops fetalis).
- HbH disease (β4): Three α-gene deletions; chronic hemolysis.
- β-Thalassemia: Caused by point mutations (chromosome 11). Excess α-chains precipitate.
- β-Thalassemia Major (Cooley's Anemia): Severe anemia, hepatosplenomegaly, "crew-cut" skull X-ray, requires transfusions.
- β-Thalassemia Minor: Often asymptomatic; ↑ HbA2 is characteristic.
- Peripheral smear: Microcytic, hypochromic anemia with target cells and basophilic stippling common findings across types.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

