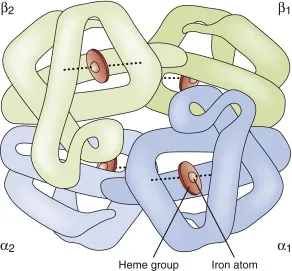
Hemoglobin Basics - Iron's O2 Taxi
- Structure: Tetrameric protein; 4 globin chains (e.g., 2$\alpha$, 2$\beta$ in HbA) + 4 heme groups.
- Heme: Iron-containing porphyrin; $Fe^{2+}$ (ferrous) at its core.
- Each $Fe^{2+}$ reversibly binds one $O_2$; 1 Hb molecule carries 4 $O_2$ molecules.
- Types (Adult %):
- HbA ($\alpha_2\beta_2$): >95%.
- HbA2 ($\alpha_2\delta_2$): 1.5-3.5%.
- HbF ($\alpha_2\gamma_2$): <2% (adults); higher $O_2$ affinity.
- Function: Primary: $O_2$ transport (lungs $\rightarrow$ tissues). Also $CO_2$ transport, blood buffer.
- Iron State for $O_2$ binding: Must be $Fe^{2+}$ (ferrous). $Fe^{3+}$ (ferric) in Methemoglobin (MetHb) cannot bind $O_2$.
⭐ HbF has higher oxygen affinity than HbA due to reduced binding of 2,3-BPG.
The O2-Hb Curve - Sigmoid Story
- Shape: Sigmoid (S-shaped) due to cooperative O2 binding.
- One O2 binding ↑ affinity for more O2.
- Axes: Y-axis: % Hb Saturation, X-axis: $pO_2$.
- P50 Value: $pO_2$ for 50% Hb saturation.
- Adult HbA: 26.6 mmHg.
- Significance:
- Steep part: Efficient O2 unloading in tissues.
- Plateau: Maintains O2 loading in lungs.
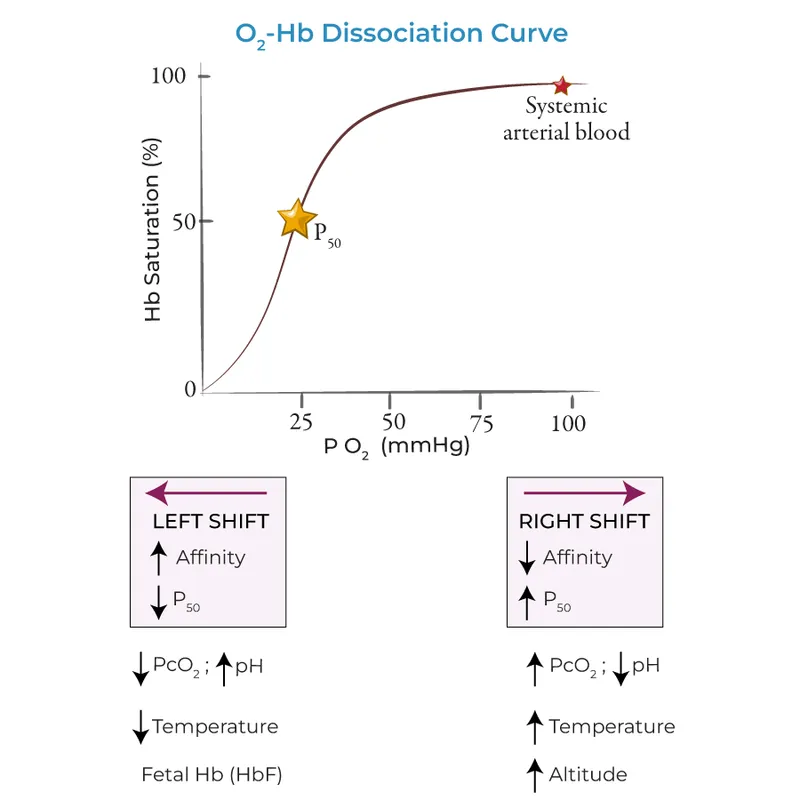
- Shifts:
- Right Shift (↑P50, ↓Affinity): 📌 CADET, face Right! (↑$CO_2$, ↑Acid/H+, ↑2,3-BPG, ↑Exercise, ↑Temp). Promotes O2 release.
- Left Shift (↓P50, ↑Affinity): Opposite factors (↓$CO_2$, etc.), HbF, CO, MetHb. Promotes O2 uptake.
⭐ Bohr effect: ↓ O2 affinity with ↑ $pCO_2$ or ↓pH, aiding O2 release in tissues.
Curve Shifters - Affinity Adjusters
Factors altering Hb-$O_2$ affinity. $P_{50}$: $O_2$ pressure for 50% Hb saturation (Normal: 26.6 mmHg). ↑ $P_{50}$ = ↓ affinity.
| Shift | $O_2$ Affinity | $P_{50}$ | Key Factors | Mnemonic |
|---|---|---|---|---|
| Right Shift | ↓ (unload $O_2$) | ↑ | ↑ $H^+$ (↓pH), ↑ $P_{\text{CO}_2}$ (Bohr), ↑ Temp, ↑ 2,3-BPG, Exercise | 📌 CADET, face Right! |
| Left Shift | ↑ (load $O_2$) | ↓ | ↓ $H^+$ (↑pH), ↓ $P_{\text{CO}_2}$, ↓ Temp, ↓ 2,3-BPG, HbF, CO, MetHb |
- 2,3-BPG: ↑ in hypoxia; stabilizes deoxyHb.
- CO: Binds Hb (~200x > $O_2$), forms COHb.
- HbF: ↑ affinity (poor 2,3-BPG binding).
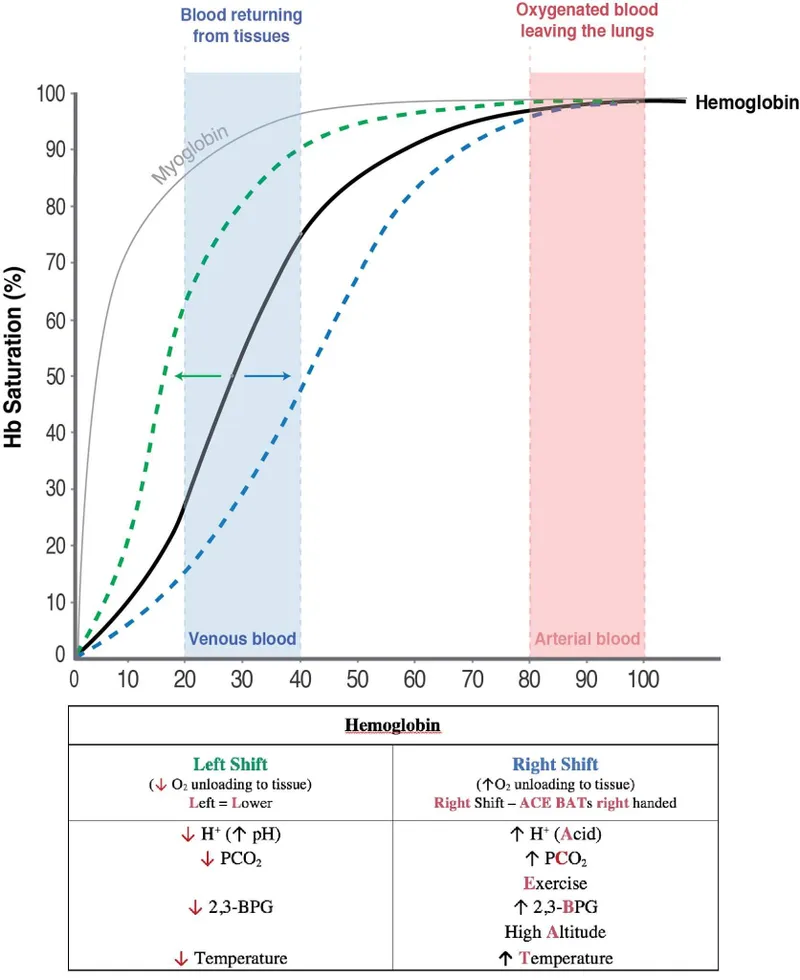
⭐ CO poisoning: left shifts ODC for available sites (↓ $O_2$ unloading) + ↓ $O_2$ carrying capacity.
Clinical Curves - Patho Pointers
- P₅₀: O₂ partial pressure for 50% Hb saturation (Normal: 26.6 mmHg). ↑P₅₀ = Right shift; ↓P₅₀ = Left shift.
- Right Shift (↓O₂ Affinity): Facilitates O₂ unloading. 📌 Mnemonic: "CADET, face Right!"
- Causes: ↑CO₂, ↑Acid (↓pH), ↑2,3-DPG, ↑Exercise, ↑Temperature.
- Clinical: Anemia, Chronic high altitude.
- Left Shift (↑O₂ Affinity): Impairs O₂ unloading.
- Causes: ↓CO₂, ↓Acid (↑pH), ↓2,3-DPG, ↓Temperature, HbF, CO, MetHb.
- CO Poisoning: Cherry-red skin. Normal PaO₂, unreliable SaO₂ (pulse ox).
- Methemoglobinemia (Fe³⁺): Chocolate blood. Tx: Methylene blue.
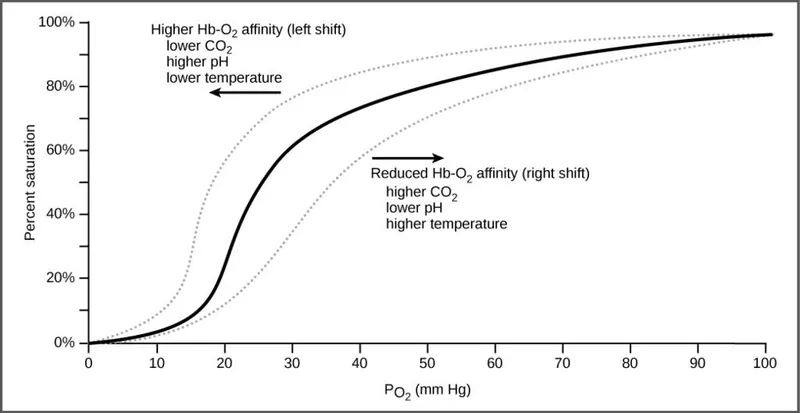
⭐ In CO poisoning, the O₂-Hb curve shifts left (impaired O₂ release) AND O₂ carrying capacity is reduced. Pulse oximetry is misleadingly normal.
- Hemoglobin's cooperative O2 binding creates a sigmoidal dissociation curve.
- P50 (~26.6 mmHg) indicates O2 affinity; ↑P50 means ↓affinity (right shift).
- Right shift factors (↑O2 release): ↑CO2, ↑H+ (↓pH), ↑2,3-BPG, ↑temperature. (CADET)
- Left shift factors (↓O2 release): ↓CO2, ↓H+ (↑pH), ↓2,3-BPG, ↓temperature, HbF, CO.
- Bohr effect: H+ and CO2 promote O2 release from Hb.
- CO poisoning: High Hb affinity for CO, left shift, ↓O2 carrying capacity.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

